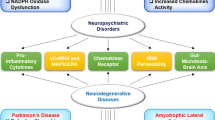
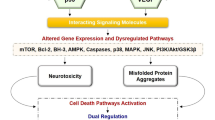
Abstract
The word mad has historically been associated with the psyche, emotions, and abnormal behavior. Dementia is a common symptom among psychiatric disorders or mad (schizophrenia, depression, bipolar disorder) patients. Autophagy/mitophagy is a protective mechanism used by cells to get rid of dysfunctional cellular organelles or mitochondria. Autophagosome/mitophagosome abundance in autophagy depends on microtubule-associated protein light chain 3B (LC3B-II) and autophagy-triggering gene (ATG) which functions as an autophagic biomarker for phagophore production and quick mRNA disintegration. Defects in either LC3B-II or the ATG lead to dysregulated mitophagy-and-autophagy–linked dementia (MAD). The impaired MAD is closely associated with schizophrenia, depression, and bipolar disorder. The pathomechanism of psychosis is not entirely known, which is the severe limitation of today’s antipsychotic drugs. However, the reviewed circuit identifies new insights that may be especially helpful in targeting biomarkers of dementia. Neuro-theranostics can also be achieved by manufacturing either bioengineered bacterial and mammalian cells or nanocarriers (liposomes, polymers, and nanogels) loaded with both imaging and therapeutic materials. The nanocarriers must cross the BBB and should release both diagnostic agents and therapeutic agents in a controlled manner to prove their effectiveness against psychiatric disorders. In this review, we highlighted the potential of microRNAs (miRs) as neuro-theranostics in the treatment of dementia by targeting autophagic biomarkers LC3B-II and ATG. Focus was also placed on the potential for neuro-theranostic nanocells/nanocarriers to traverse the BBB and induce action against psychiatric disorders. The neuro-theranostic approach can provide targeted treatment for mental disorders by creating theranostic nanocarriers.





Similar content being viewed by others
Data Availability
Data sharing is not applicable (NA).
References
Pickard H (2015) Choice, deliberation, violence: mental capacity and criminal responsibility in personality disorder. Int J Law Psychiatry 40:15. https://doi.org/10.1016/J.IJLP.2015.04.008
Kiran C, Chaudhury S (2009) Understanding delusions Ind Psychiatry J 18:3. https://doi.org/10.4103/0972-6748.57851
Arvanitakis Z, Shah RC, Bennett DA (2019) Diagnosis and management of dementia: a review. J Am Med Assoc 322(16):1589–1599. https://doi.org/10.1001/jama.2019.4782
Ricci G (2019). Social aspects of dementia prevention from a worldwide to national perspective: a review on the international situation and the example of Italy. https://doi.org/10.1155/2019/8720904
Tori K, Kalligeros M, Nanda A et al (2020). Association between dementia and psychiatric disorders in long-term care residents an observational clinical study. https://doi.org/10.1097/MD.0000000000021412
Zilkens RR, Bruce DG, Duke J et al (2014) Send orders for reprints to reprints@benthamscience.net Severe psychiatric disorders in mid-life and risk of dementia in late-life (age 65–84 years): a population based case-control study. Curr Alzheimer Res 11:681–693
Luvsannyam E, Jain MS, Pormento MKL et al (2022) Neurobiology of schizophrenia: a comprehensive review. Cureus 14(4):23959. https://doi.org/10.7759/CUREUS.23959
Stępnicki P, Kondej M, Kaczor AA (2018) Current concepts and treatments of schizophrenia. Mol A J Synth Chem Nat Prod Chem 23(8):2087. https://doi.org/10.3390/MOLECULES23082087
Fink M, Shorter E, Taylor MA (2010) Catatonia Is not schizophrenia: Kraepelin’s error and the need to recognize catatonia as an independent syndrome in medical nomenclature. Schizophr Bull 36:314. https://doi.org/10.1093/SCHBUL/SBP059
Tripathi M, Vibha D (2009) Reversible dementias. Indian J Psychiatry 51:S52
Fountoulakis KN (2010) The emerging modern face of mood disorders: a didactic editorial with a detailed presentation of data and definitions. Ann Gen Psychiatry 9(14):1–22. https://doi.org/10.1186/1744-859X-9-14
Ganguli M (2009) Depression, cognitive impairment and dementia: why should clinicians care about the web of causation? Indian J Psychiatry 51:S29
Banga A, Gyurmey T, Matuskey D et al (2013) Late-life onset bipolar disorder presenting as a case ofpseudo-dementia: a case discussion and review of literature. Yale J Biol Med 86:235
Leung CCY, Gadelrab R, Ntephe CU et al (2019) Clinical course, neurobiology and therapeutic approaches to treatment resistant schizophrenia. Toward an integrated view. Front Psych 10:601. https://doi.org/10.3389/FPSYT.2019.00601/BIBTEX
Cerejeira J, Lagarto L, Mukaetova-Ladinska EB (2012) Behavioral and psychological symptoms of dementia. Front Neurol 3(73):1–21. https://doi.org/10.3389/FNEUR.2012.00073
Kitching D (2015) Depression in dementia. . Aust Prescr 38:209. https://doi.org/10.18773/AUSTPRESCR.2015.071
Parzych KR, Klionsky DJ (2014) An overview of autophagy: morphology, mechanism, and regulation. Antioxid Redox Signal 20:460. https://doi.org/10.1089/ARS.2013.5371
Khandia R, Dadar M, Munjal A et al (2019) A comprehensive review of autophagy and its various roles in infectious, non-infectious, and lifestyle diseases: current knowledge and prospects for disease prevention, novel drug design, and therapy. Cells 8(7):674. https://doi.org/10.3390/CELLS8070674
Yang Z, Klionsky DJ (2009) An overview of the molecular mechanism of autophagy. Curr Top Microbiol Immunol 335:1. https://doi.org/10.1007/978-3-642-00302-8_1
Green DR, Galluzzi L, Kroemer G (2011) Mitochondria and the autophagy-inflammation-cell death axis in organismal aging. Science 333:1109. https://doi.org/10.1126/SCIENCE.1201940
Daniels TE, Olsen EM, Tyrka AR (2020) Stress and psychiatric disorders: the role of mitochondria. Annu Rev Clin Psychol 16:165. https://doi.org/10.1146/ANNUREV-CLINPSY-082719-104030
Picard M, McEwen BS (2018) Psychological stress and mitochondria: a conceptual framework. Psychosom Med 80:126. https://doi.org/10.1097/PSY.0000000000000544
Picard M, McEwen BS (2018) Psychological stress and mitochondria: a systematic review. Psychosom Med 80:141. https://doi.org/10.1097/PSY.0000000000000545
Eckl E-M, Ziegemann O, Krumwiede L et al (2021) Sensing, signaling and surviving mitochondrial stress. Cell Mol Life Sci 78:5925–5951. https://doi.org/10.1007/s00018-021-03887-7
Cai Q, Jeong YY (2020) Mitophagy in Alzheimer’s disease and other age-related neurodegenerative diseases. Cells 9(1):150. https://doi.org/10.3390/CELLS9010150
Bernstein HG, Keilhoff G, Dobrowolny H, Steiner J (2020) Enhanced mitochondrial autophagy (mitophagy) in oligodendrocytes might play a role in white matter pathology in schizophrenia. Med Hypotheses 134:109443. https://doi.org/10.1016/J.MEHY.2019.109443
Di Meo S, Reed TT, Venditti P, Victor VM (2016) Role of ROS and RNS sources in physiological and pathological conditions. Oxid Med Cell Longev 2016:1245049. https://doi.org/10.1155/2016/1245049
Zorov DB, Juhaszova M, Sollott SJ (2014) Mitochondrial reactive oxygen species (ROS) and ROS-induced ROS release. Physiol Rev 94:909. https://doi.org/10.1152/PHYSREV.00026.2013
Jung J, Huh Y, Shefa U et al (2019). Mitophagy links oxidative stress conditions and neurodegenerative diseases. https://doi.org/10.4103/1673-5374.249218
Li S, Zhang J, Liu C et al (2021) The role of mitophagy in regulating cell death. Oxid Med Cell Longev 2021:6617256. https://doi.org/10.1155/2021/6617256
Panda SP, Dhurandhar Y, Agrawal M (2022) The interplay of epilepsy with impaired mitophagy and autophagy linked dementia (MAD): a review of therapeutic approaches. Mitochondrion 66:27–37. https://doi.org/10.1016/J.MITO.2022.07.002
Doblado L, Lueck C, Rey C et al (2021) Mitophagy in human diseases. Int J Mol Sci 22:3903. https://doi.org/10.3390/IJMS22083903
Mailman RB, Murthy V (2010) Third generation antipsychotic drugs: partial agonism or receptor functional selectivity? Curr Pharm Des 16:488–501
Boczek T, Mackiewicz J, Sobolczyk M et al (2021) The role of G protein-coupled receptors (GPCRs) and calcium signaling in schizophrenia. Focus on GPCRs activated by neurotransmitters and chemokines. Cells 10:1228. https://doi.org/10.3390/CELLS10051228
Read J, Williams J (2019) Current Drug Safety Current Drug Safety Send Orders for Reprints to reprints@benthamscience.net Positive and negative effects of antipsychotic medication: an interna-tional online survey of 832 recipients. Curr Drug Saf 14:173–181. https://doi.org/10.2174/1574886314666190301152734
Allen JD, Bishop JR (2019) A systematic review of genome-wide association studies of antipsychotic response. Pharmacogenomics 20:291–306. https://doi.org/10.2217/pgs-2018-0163
Wahbeh MH, Avramopoulos D (2021) Gene-environment interactions in schizophrenia: a literature review. Genes (Basel) 12:1–13. https://doi.org/10.3390/GENES12121850
Hong J, Bang M (2020) Anti-inflammatory strategies for schizophrenia: a review of evidence for therapeutic applications and drug repurposing. Clin Psychopharmacol Neurosci 18:10. https://doi.org/10.9758/CPN.2020.18.1.10
Zhuo C, Xu Y, Hou W et al (2022) Mechanistic/mammalian target of rapamycin and side effects of antipsychotics: insights into mechanisms and implications for therapy. Transl Psychiatry 12(1):1–8. https://doi.org/10.1038/S41398-021-01778-W
Bar-Yosef T, Damri O, Agam G (2019) Dual role of autophagy in diseases of the central nervous system. Front Cell Neurosci 13(196):1–14. https://doi.org/10.3389/FNCEL.2019.00196
Sekiguchi A, Kanno H, Ozawa H et al (2012) Rapamycin promotes autophagy and reduces neural tissue damage and locomotor impairment after spinal cord injury in mice. J Neurotrauma 29:946–956. https://doi.org/10.1089/NEU.2011.1919
Gros F, Muller S (2014) Pharmacological regulators of autophagy and their link with modulators of lupus disease. Br J Pharmacol 171:4337. https://doi.org/10.1111/BPH.12792
Sciarretta S, Zhai P, Volpe M, Sadoshima J (2012) Pharmacological modulation of autophagy during cardiac stress. J Cardiovasc Pharmacol 60:235–241. https://doi.org/10.1097/FJC.0B013E3182575F61
Chen MH, Wang FX, Cao JJ et al (2017) Light-up mitophagy in live cells with dual-functional theranostic phosphorescent iridium(III) complexes. ACS Appl Mater Interfaces 9:13304–13314. https://doi.org/10.1021/ACSAMI.7B01735
Dayem AA, Hossain MK, Lee S Bin et al (2017) The role of reactive oxygen species (ROS) in the biological activities of metallic nanoparticles. Int J Mol Sci 18(1):1–21. https://doi.org/10.3390/IJMS18010120
Lunova M, Smolková B, Lynnyk A et al (2019) Targeting the mTOR signaling pathway utilizing nanoparticles: a critical overview. Cancers (Basel) 11(1):1–18. https://doi.org/10.3390/CANCERS11010082
**e J, Lee S, Chen X (2010). Nanoparticle-based theranostic agents. https://doi.org/10.1016/j.addr.2010.07.009
Zare H, Ahmadi S, Ghasemi A et al (2021) Carbon nanotubes: smart drug/gene delivery carriers. Int J Nanomedicine 16:1681. https://doi.org/10.2147/IJN.S299448
Yu Z, Li Q, Wang J et al (2020) Reactive oxygen species-related nanoparticle toxicity in the biomedical field. Nanoscale Res Lett 151(15):1–14. https://doi.org/10.1186/S11671-020-03344-7
Luk BT, Zhang L (2014) Currentadvances in polymer-based nanotheranosticsfor cancer treatment and diagnosis. ACS Appl Mater Interfaces 6:21859. https://doi.org/10.1021/AM5036225
Gheorghita R, Anchidin-Norocel L, Filip R et al (2021) Applications of biopolymers for drugs and probiotics delivery. Polymers (Basel) 13(16):2729. https://doi.org/10.3390/POLYM13162729
Begines B, Ortiz T, Pérez-Aranda M et al (2020) Polymeric nanoparticles for drug delivery: recent developments and future prospects. Nanomaterials 10:1–41. https://doi.org/10.3390/NANO10071403
Attia MF, Anton N, Wallyn J et al (2019) An overview of active and passive targeting strategies to improve the nanocarriers efficiency to tumour sites. J Pharm Pharmacol 71:1185–1198. https://doi.org/10.1111/JPHP.13098
Steichen SD, Caldorera-Moore M, Peppas NA (2012). A review of current nanoparticle and targeting moieties for the delivery of cancer therapeutics. https://doi.org/10.1016/j.ejps.2012.12.006
Subhan MA, Yalamarty SSK, Filipczak N et al (2021) Recent advances in tumor targeting via EPR effect for cancer treatment. J Pers Med 11:571. https://doi.org/10.3390/JPM11060571
Kraft LJ, Nguyen TA, Vogel SS, Kenworthy AK (2014) Size, stoichiometry, and organization of soluble LC3-associated complexes. Autophagy 10:861. https://doi.org/10.4161/AUTO.28175
Tanida I, Ueno T, Kominami E (2008) LC3 and autophagy. Methods Mol Biol 445:77–88. https://doi.org/10.1007/978-1-59745-157-4_4
Koukourakis MI, Kalamida D, Giatromanolaki A et al (2015) Autophagosome proteins LC3A, LC3B and LC3C have distinct subcellular distribution kinetics and expression in cancer cell lines. PLoS One 10(9):1–13. https://doi.org/10.1371/JOURNAL.PONE.0137675
Tanida I, Ueno T, Kominami E (2004) LC3 conjugation system in mammalian autophagy. Int J Biochem Cell Biol 36:2503. https://doi.org/10.1016/J.BIOCEL.2004.05.009
Dancourt J, Melia TJ (2014) Lipidation of the autophagy proteins LC3 and GABARAP is a membrane-curvature dependent process. Autophagy 10:1470. https://doi.org/10.4161/AUTO.29468
Sharifi MN, Mowers EE, Drake LE, Macleod KF (2015) Measuring autophagy in stressed cells. Methods Mol Biol 1292:129. https://doi.org/10.1007/978-1-4939-2522-3_10
Mizushima N, Yoshimori T, Levine B (2010) Methods in mammalian autophagy research. Cell 140:313. https://doi.org/10.1016/J.CELL.2010.01.028
Agrotis A, Pengo N, Burden JJ, Ketteler R (2019) Redundancy of human ATG4 protease isoforms in autophagy and LC3/GABARAP processing revealed in cells. Autophagy 15:976. https://doi.org/10.1080/15548627.2019.1569925
Lee Y, Jun Y, Choi H et al (2017) Development of LC3/GABARAP sensors containing a LIR and a hydrophobic domain to monitor autophagy. EMBO J 36:1100. https://doi.org/10.15252/EMBJ.201696315
Das G, Shravage BV, Baehrecke EH (2012) Regulation and function of autophagy during cell survival and cell death. Cold Spring Harb Perspect Biol 4:1–14. https://doi.org/10.1101/CSHPERSPECT.A008813
Johansen T, Lamark T (2011) Selective autophagy mediated by autophagic adapter proteins. Autophagy 7:279. https://doi.org/10.4161/AUTO.7.3.14487
Mizushima N, Yoshimori T (2007) How to interpret LC3 immunoblotting. Autophagy 3:542–545. https://doi.org/10.4161/AUTO.4600
Yang Z, Wilkie-Grantham RP, Yanagi T et al (2015) ATG4B (Autophagin-1) phosphorylation modulates autophagy. J Biol Chem 290:26549. https://doi.org/10.1074/JBC.M115.658088
Goldsmith J, Levine B, Debnath J (2014) Autophagy and cancer metabolism. Methods Enzymol 542:25–57. https://doi.org/10.1016/B978-0-12-416618-9.00002-9
Vujić N, Bradić I, Goeritzer M et al (2021) ATG7 is dispensable for LC3-PE conjugation in thioglycolate-elicited mouse peritoneal macrophages. Autophagy 17:3402–3407. https://doi.org/10.1080/15548627.2021.1874132
Walczak M, Martens S (2013) Dissecting the role of the Atg12–Atg5-Atg16 complex during autophagosome formation. Autophagy 9:424. https://doi.org/10.4161/AUTO.22931
Otomo C, Metlagel Z, Takaesu G, Otomo T (2013) Structure of the human ATG12~ATG5 conjugate required for LC3 lipidation in autophagy. Nat Struct Mol Biol 20:59. https://doi.org/10.1038/NSMB.2431
Pankiv S, Clausen TH, Lamark T et al (2007) p62/SQSTM1 binds directly to Atg8/LC3 to facilitate degradation of ubiquitinated protein aggregates by autophagy. J Biol Chem 282:24131–24145. https://doi.org/10.1074/JBC.M702824200
Hwang HJ, Ha H, Lee BS et al (2022) LC3B is an RNA-binding protein to trigger rapid mRNA degradation during autophagy. Nat Commun 13(1):1–17. https://doi.org/10.1038/S41467-022-29139-1
Hwang HJ, Kim YK (2023) The role of LC3B in autophagy as an RNA-binding protein. Autophagy 19(3):1028–1030. https://doi.org/10.1080/15548627.2022.2111083
Kim H, Seong J (2021) Fluorescent protein-based autophagy biosensors. Materials (Basel) 14(11):3019. https://doi.org/10.3390/MA14113019
Kost TA, Condreay JP, Jarvis DL (2005) Baculovirus as versatile vectors for protein expression in insect and mammalian cells. Nat Biotechnol 23:567. https://doi.org/10.1038/NBT1095
O’Grady M, Batchelor RH, Scheyhing K et al (2011) BacMam-mediated gene delivery into multipotent mesenchymal stromal cells. Methods Mol Biol 698:485–504. https://doi.org/10.1007/978-1-60761-999-4_34
Chen CY, Lin CY, Chen GY, Hu YC (2011) Baculovirus as a gene delivery vector: recent understandings of molecular alterations in transduced cells and latest applications. Biotechnol Adv 29:618. https://doi.org/10.1016/J.BIOTECHADV.2011.04.004
Levin E, Diekmann H, Fischer D (2016) Highly efficient transduction of primary adult CNS and PNS neurons. Sci Rep 6:38928. https://doi.org/10.1038/SREP38928
Kwon DH, Kim S, Jung YO et al (2017) The 1:2 complex between RavZ and LC3 reveals a mechanism for deconjugation of LC3 on the phagophore membrane. Autophagy 13:70. https://doi.org/10.1080/15548627.2016.1243199
Tsukahara T, Haniu H, Matsuda Y (2015) The PTB-associated splicing factor/peroxisome proliferator-activated receptor gamma axis regulates autophagosome formation in human pancreatic cancer cells. Biores Open Access 4:319. https://doi.org/10.1089/BIORES.2015.0018
Mauthe M, Orhon I, Rocchi C et al (2018) Chloroquine inhibits autophagic flux by decreasing autophagosome-lysosome fusion. Autophagy 14:1435. https://doi.org/10.1080/15548627.2018.1474314
Frost LS, Dhingra A, Reyes-Reveles J, Boesze-Battaglia K (2017) The use of DQ-BSA to monitor the turnover of autophagy-associated cargo. Methods Enzymol 587:43–54. https://doi.org/10.1016/BS.MIE.2016.09.052
Zhang Z, Singh R, Aschner M (2016) Methods for the detection of autophagy in mammalian cells. Curr Protoc Toxicol 69:1–20. https://doi.org/10.1002/CPTX.11
Fivenson EM, Lautrup S, Sun N et al (2017) Mitophagy in neurodegeneration and aging. Neurochem Int 109:202. https://doi.org/10.1016/J.NEUINT.2017.02.007
Tran M, Reddy PH (2020) Defective autophagy and mitophagy in aging and Alzheimer’s disease. Front Neurosci 14:612757. https://doi.org/10.3389/FNINS.2020.612757
Rao RV, Bredesen DE (2004) Misfolded proteins, endoplasmic reticulum stress and neurodegeneration. Curr Opin Cell Biol 16:653. https://doi.org/10.1016/J.CEB.2004.09.012
Li L, Acioglu C, Heary RF, Elkabes S (2021) Role of astroglial toll-like receptors (TLRs) in central nervous system infections, injury and neurodegenerative diseases. Brain Behav Immun 91:740. https://doi.org/10.1016/J.BBI.2020.10.007
Hanke ML, Kielian T (2011) Toll-like receptors in health and disease in the brain: mechanisms and therapeutic potential. Clin Sci (Lond) 121:367. https://doi.org/10.1042/CS20110164
Akabane S, Matsuzaki K, Yamashita SI et al (2016) Constitutive activation of PINK1 protein leads to proteasome-mediated and non-apoptotic cell death independently of mitochondrial autophagy. J Biol Chem 291:16162. https://doi.org/10.1074/JBC.M116.714923
McLelland GL, Goiran T, Yi W et al (2018) Mfn2 ubiquitination by PINK1/parkin gates the p97-dependent release of ER from mitochondria to drive mitophagy. Elife 7:1–35. https://doi.org/10.7554/ELIFE.32866
Gegg ME, Schapira AHV (2011) PINK1-parkin-dependent mitophagy involves ubiquitination of mitofusins 1 and 2: implications for Parkinson disease pathogenesis. Autophagy 7:243. https://doi.org/10.4161/AUTO.7.2.14332
Terešak P, Lapao A, Subic N et al (2022) Regulation of PRKN-independent mitophagy. Autophagy 18:24. https://doi.org/10.1080/15548627.2021.1888244
Park SY, Koh HC (2020) FUNDC1 regulates receptor-mediated mitophagy independently of the PINK1/Parkin-dependent pathway in rotenone-treated SH-SY5Y cells. Food Chem Toxicol 137:111163. https://doi.org/10.1016/J.FCT.2020.111163
Tripathi A, Sciani G, Barichello T et al (2021) Mitophagy in depression: pathophysiology and treatment targets. Mitochondrion 61:1–10. https://doi.org/10.1016/J.MITO.2021.08.016
Bottani E, Lamperti C, Prigione A et al (2020) Therapeutic approaches to treat mitochondrial diseases: “one-size-fits-all” and “precision medicine” strategies. Pharmaceutics 12:1–63. https://doi.org/10.3390/PHARMACEUTICS12111083
Iwashita H, Torii S, Nagahora N et al (2017) Live cell imaging of mitochondrial autophagy with a novel fluorescent small molecule. ACS Chem Biol 12:2546–2551. https://doi.org/10.1021/ACSCHEMBIO.7B00647
Glancy B (2020) Visualizing mitochondrial form and function within the cell. Trends Mol Med 26:58. https://doi.org/10.1016/J.MOLMED.2019.09.009
Zhu J, Dagda RK, Chu CT (2011) Monitoring mitophagy in neuronal cell cultures. Methods Mol Biol 793:325. https://doi.org/10.1007/978-1-61779-328-8_21
Han S, Zhang M, Jeong YY et al (2021) The role of mitophagy in the regulation of mitochondrial energetic status in neurons. Autophagy 17:4182. https://doi.org/10.1080/15548627.2021.1907167
Jouan E, Le Vee M, Denizot C et al (2014) The mitochondrial fluorescent dye rhodamine 123 is a high-affinity substrate for organic cation transporters (OCTs) 1 and 2. Fundam Clin Pharmacol 28:65–77. https://doi.org/10.1111/J.1472-8206.2012.01071.X
Mitra K, Lippincott-Schwartz J (2010) Analysis of mitochondrial dynamics and functions using imaging approaches. Curr Protoc Cell Biol 46(1):4–25. https://doi.org/10.1002/0471143030.CB0425S46
Chazotte B (2011) Labeling mitochondria with MitoTracker dyes. Cold Spring Harb Protoc 2011:990–992. https://doi.org/10.1101/PDB.PROT5648
Jacotot E, Ferri KF, El Hamel C et al (2001) Control of mitochondrial membrane permeabilization by adenine nucleotide translocator interacting with HIV-1 viral protein R and Bcl-2. J Exp Med 193:509. https://doi.org/10.1084/JEM.193.4.509
Schneider A, Kurz S, Manske K et al (2019) Single organelle analysis to characterize mitochondrial function and crosstalk during viral infection. Sci Rep 9(1):1–11. https://doi.org/10.1038/S41598-019-44922-9
Lee S, Chen X (2011) Selective imaging of mitochondrial surfaces with novel fluorescent probes. ChemBioChem 12:2120. https://doi.org/10.1002/CBIC.201100365
Cottet-Rousselle C, Ronot X, Leverve X, Mayol JF (2011) Cytometric assessment of mitochondria using fluorescent probes. Cytom Part A 79A:405–425. https://doi.org/10.1002/CYTO.A.21061
Hallap T, Nagy S, Jaakma Ü et al (2005) Mitochondrial activity of frozen-thawed spermatozoa assessed by MitoTracker Deep Red 633. Theriogenology 63:2311–2322. https://doi.org/10.1016/J.THERIOGENOLOGY.2004.10.010
Qin Y, Jiang W, Li A et al (2021) The combination of paraformaldehyde and glutaraldehyde is a potential fixative for mitochondria. Biomolecules 11(5):711. https://doi.org/10.3390/BIOM11050711/S1
Gautam N, Sankaran S, Yason JA et al (2018) A high content imaging flow cytometry approach to study mitochondria in T cells: MitoTracker Green FM dye concentration optimization. Methods 134–135:11–19. https://doi.org/10.1016/J.YMETH.2017.11.015
Soliman E, Elhassanny AEM, Malur A et al (2020) Impaired mitochondrial function of alveolar macrophages in carbon nanotube-induced chronic pulmonary granulomatous disease. Toxicology 445:152598. https://doi.org/10.1016/J.TOX.2020.152598
Kholmukhamedov A, Schwartz JM, Lemasters JJ (2013) MitoTracker probes and mitochondrial membrane potential. Shock 39:543. https://doi.org/10.1097/SHK.0B013E318292300D
Han J, Han MS, Tung CH (2013) A non-toxic fluorogenic dye for mitochondria labeling. Biochim Biophys Acta 1830:5130. https://doi.org/10.1016/J.BBAGEN.2013.07.001
Presley AD, Fuller KM, Arriaga EA (2003) MitoTracker Green labeling of mitochondrial proteins and their subsequent analysis by capillary electrophoresis with laser-induced fluorescence detection. J Chromatogr B Anal Technol Biomed Life Sci 793:141–150. https://doi.org/10.1016/S1570-0232(03)00371-4
Buravkov SV, Pogodina MV, Buravkova LB (2014) Comparison of mitochondrial fluorescent dyes in stromal cells. Bull Exp Biol Med 157:654–658. https://doi.org/10.1007/S10517-014-2637-3
Minamikawa T, Sriratana A, Williams DA et al (1999) Chloromethyl-X-rosamine (MitoTracker Red) photosensitises mitochondria and induces apoptosis in intact human cells. J Cell Sci 112 (14):2419–2430. https://doi.org/10.1242/JCS.112.14.2419
Buckman JF, Hernández H, Kress GJ et al (2001) MitoTracker labeling in primary neuronal and astrocytic cultures: influence of mitochondrial membrane potential and oxidants. J Neurosci Methods 104:165–176. https://doi.org/10.1016/S0165-0270(00)00340-X
Marvel CL, Paradiso S (2004) Cognitive and neurological impairment in mood disorders. Psychiatr Clin North Am 27:19. https://doi.org/10.1016/S0193-953X(03)00106-0
Penninx BWJH, Lange SMM (2018) Metabolic syndrome in psychiatric patients: overview, mechanisms, and implications. Dialogues Clin Neurosci 20:63. https://doi.org/10.31887/DCNS.2018.20.1/BPENNINX
Patel KR, Cherian J, Gohil K, Atkinson D (2014) Schizophrenia: overview and treatment options. Pharm Ther 39:638
Murphy D (2011) Conceptual foundations of biological psychiatry. Philos Med 16:425–451. https://doi.org/10.1016/B978-0-444-51787-6.50013-1
Perneczky R (2019) Dementia prevention and reserve against neurodegenerative disease. Dialogues Clin Neurosci 21:53. https://doi.org/10.31887/DCNS.2019.21.1/RPERNECZKY2
Cai L, Huang J (2018) Schizophrenia and risk of dementia: a meta-analysis study. Neuropsychiatr Dis Treat 14:2047. https://doi.org/10.2147/NDT.S172933
Perkovic MN, Erjavec GN, Strac DS et al (2017) Theranostic biomarkers for schizophrenia. Int J Mol Sci 18(4):733. https://doi.org/10.3390/IJMS18040733
Merenlender-Wagner A, Malishkevich A, Shemer Z et al (2015) Autophagy has a key role in the pathophysiology of schizophrenia. Mol Psychiatry 20:126. https://doi.org/10.1038/MP.2013.174
Grossman M, Irwin DJ (2016) The mental status examination in patients with suspected dementia. Contin Lifelong Learn Neurol 22(2):385–403. https://doi.org/10.1212/CON.0000000000000298
Byrne P (2007) Managing the acute psychotic episode. BMJ Br Med J 334:686. https://doi.org/10.1136/BMJ.39148.668160.80
Kennedy SH (2008) Core symptoms of major depressive disorder: relevance to diagnosis and treatment. Dialogues Clin Neurosci 10:271. https://doi.org/10.31887/DCNS.2008.10.3/SHKENNEDY
Mei Y, Thompson MD, Cohen RA, Tong XY (2015) Autophagy and oxidative stress in cardiovascular diseases. Biochim Biophys Acta 1852:243. https://doi.org/10.1016/J.BBADIS.2014.05.005
Fleming A, Bourdenx M, Fujimaki M et al (2022) The different autophagy degradation pathways and neurodegeneration. Neuron 110:935–966. https://doi.org/10.1016/J.NEURON.2022.01.017
Abi-Dargham A, Rodenhiser J, Printz D et al (2000) Increased baseline occupancy of D2 receptors by dopamine in schizophrenia. Proc Natl Acad Sci U S A 97:8104–8109. https://doi.org/10.1073/PNAS.97.14.8104
Steeds H, Carhart-Harris RL, Stone JM (2015) Drug models of schizophrenia. Ther Adv Psychopharmacol 5:43. https://doi.org/10.1177/2045125314557797
Zhou Y, Danbolt NC (2014) Glutamate as a neurotransmitter in the healthy brain. J Neural Transm 121:799. https://doi.org/10.1007/S00702-014-1180-8
McCutcheon RA, Krystal JH, Howes OD (2020) Dopamine and glutamate in schizophrenia: biology, symptoms and treatment. World Psychiatry 19:15. https://doi.org/10.1002/WPS.20693
Tropea D, Hardingham N, Millar K, Fox K (2018) Mechanisms underlying the role of DISC1 in synaptic plasticity. J Physiol 596:2747. https://doi.org/10.1113/JP274330
Quintero H, Shiga Y, Belforte N et al (2022) Restoration of mitochondria axonal transport by adaptor Disc1 supplementation prevents neurodegeneration and rescues visual function. Cell Rep. 40:111324. https://doi.org/10.1016/J.CELREP.2022.111324
Lahiri V, Klionsky DJ (2017) PHB2/prohibitin 2: an inner membrane mitophagy receptor. Cell Res 27:311–312. https://doi.org/10.1038/CR.2017.23
Thomson PA, Malavasi ELV, Grünewald E et al (2013) DISC1 genetics, biology and psychiatric illness. Front Biol (Bei**g) 8:1. https://doi.org/10.1007/S11515-012-1254-7
Flippo KH, Strack S (2017) An emerging role for mitochondrial dynamics in schizophrenia. Schizophr Res 187:26. https://doi.org/10.1016/J.SCHRES.2017.05.003
Baldessarini RJ, Vázquez GH, Tondo L (2020) Bipolar depression: a major unsolved challenge. Int J Bipolar Disord 8(1):1. https://doi.org/10.1186/S40345-019-0160-1
Cataldo AM, McPhie DL, Lange NT et al (2010) Abnormalities in mitochondrial structure in cells from patients with bipolar disorder. Am J Pathol 177:575–585. https://doi.org/10.2353/AJPATH.2010.081068
Reinecke F, Smeitink JAM, van der Westhuizen FH (2009) OXPHOS gene expression and control in mitochondrial disorders. Biochim Biophys Acta - Mol Basis Dis 1792:1113–1121. https://doi.org/10.1016/J.BBADIS.2009.04.003
Roberts RC (2017) Postmortem studies on mitochondria in schizophrenia. Schizophr Res 187:17. https://doi.org/10.1016/J.SCHRES.2017.01.056
Guo CY, Sun L, Chen XP, Zhang DS (2013) Oxidative stress, mitochondrial damage and neurodegenerative diseases. Neural Regen Res 8:2003. https://doi.org/10.3969/J.ISSN.1673-5374.2013.21.009
Liu J, Liu L, Han YS et al (2021) The molecular mechanism underlying mitophagy-mediated hippocampal neuron apoptosis in diabetes-related depression. J Cell Mol Med 25:7342–7353. https://doi.org/10.1111/JCMM.16763
Scaini G, Barichello T, Fries GR et al (2019) TSPO upregulation in bipolar disorder and concomitant downregulation of mitophagic proteins and NLRP3 inflammasome activation. Neuropsychopharmacology 44:1291. https://doi.org/10.1038/S41386-018-0293-4
Gatliff J, East D, Crosby J et al (2014) TSPO interacts with VDAC1 and triggers a ROS-mediated inhibition of mitochondrial quality control. Autophagy 10:2279. https://doi.org/10.4161/15548627.2014.991665
Tsuang MT, Bar JL, Stone WS, Faraone SV (2004) Gene-environment interactions in mental disorders. World Psychiatry 3:73
Ogundele MO (2018) Behavioural and emotional disorders in childhood: a brief overview for paediatricians. World J Clin Pediatr 7(1):9. https://doi.org/10.5409/WJCP.V7.I1.9
Feigin VL, Nichols E, Alam T et al (2019) Global, regional, and national burden of neurological disorders, 1990–2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol 18:459. https://doi.org/10.1016/S1474-4422(18)30499-X
Allen J, Romay-Tallon R, Brymer KJ et al (2018) Mitochondria and mood: mitochondrial dysfunction as a key player in the manifestation of depression. Front Neurosci 12:386. https://doi.org/10.3389/FNINS.2018.00386
Chan ST, McCarthy MJ, Vawter MP (2020) Psychiatric drugs impact mitochondrial function in brain and other tissues. Schizophr Res 217:136–147. https://doi.org/10.1016/J.SCHRES.2019.09.007
Lautrup S, Lou G, Aman Y et al (2019) Microglial mitophagy mitigates neuroinflammation in Alzheimer’s disease. Neurochem Int 129:104469. https://doi.org/10.1016/J.NEUINT.2019.104469
Goode A, Butler K, Long J et al (2016) Defective recognition of LC3B by mutant SQSTM1/p62 implicates impairment of autophagy as a pathogenic mechanism in ALS-FTLD. Autophagy 12:1094. https://doi.org/10.1080/15548627.2016.1170257
Dagda RK, Rice M (2017) Protocols for assessing mitophagy in neuronal cell lines and primary neurons. NeuroMethods 123:249. https://doi.org/10.1007/978-1-4939-6890-9_13
Ebrahimi-Fakhari D, Saffari A, Wahlster L et al (2016) Congenital disorders of autophagy: an emerging novel class of inborn errors of neuro-metabolism. Brain 139:317. https://doi.org/10.1093/BRAIN/AWV371
Jeelani S, Jagat Reddy RC, Maheswaran T et al (2014) Theranostics: a treasured tailor for tomorrow. J Pharm Bioallied Sci 6:S6. https://doi.org/10.4103/0975-7406.137249
Zavaleta C, Ho D, Chung EJ (2018) Theranostic nanoparticles for tracking and monitoring disease state. SLAS Technol 23:281. https://doi.org/10.1177/2472630317738699
Cen P, Zhou Y, Cui C et al (2022) Optical molecular imaging and theranostics in neurological diseases based on aggregation-induced emission luminogens. Eur J Nucl Med Mol Imaging 4913(49):4529–4550. https://doi.org/10.1007/S00259-022-05894-7
Patra JK, Das G, Fraceto LF et al (2018) Nano based drug delivery systems: recent developments and future prospects. J Nanobiotechnology 161(16):1–33. https://doi.org/10.1186/S12951-018-0392-8
Wen H, Jung H, Li X (2015) Drug delivery approaches in addressing clinical pharmacology-related issues: opportunities and challenges. AAPS J 17:1327. https://doi.org/10.1208/S12248-015-9814-9
Murthy SK (2007) Nanoparticles in modern medicine: state of the art and future challenges. Int J Nanomedicine 2:129
Sharma D, Kanchi S, Bisetty K (2019) Biogenic synthesis of nanoparticles: a review. Arab J Chem 12:3576–3600. https://doi.org/10.1016/J.ARABJC.2015.11.002
Madamsetty VS, Mukherjee A, Mukherjee S (2019) Recent trends of the bio-inspired nanoparticles in cancer theranostics. Front Pharmacol 10:1264. https://doi.org/10.3389/FPHAR.2019.01264
Yang W, Veroniaina H, Qi X et al (2020) Soft and condensed nanoparticles and nanoformulations for cancer drug delivery and repurpose. Adv Ther 3:1900102. https://doi.org/10.1002/ADTP.201900102
Siafaka PI, Okur NÜ, Karantas ID et al (2021) Current update on nanoplatforms as therapeutic and diagnostic tools: a review for the materials used as nanotheranostics and imaging modalities. Asian J Pharm Sci 16:24–46. https://doi.org/10.1016/J.AJPS.2020.03.003
Gaur M, Misra C, Yadav AB et al (2021) Biomedical applications of carbon nanomaterials: Fullerenes, quantum dots, nanotubes, nanofibers, and graphene. Materials (Basel) 14(20):1–35. https://doi.org/10.3390/MA14205978
Battigelli A, Russier J, Venturelli E et al (2013) Peptide-based carbon nanotubes for mitochondrial targeting. Nanoscale 5:9110–9117. https://doi.org/10.1039/C3NR02694A
Rozhina E, Batasheva S, Miftakhova R et al (2021) Comparative cytotoxicity of kaolinite, halloysite, multiwalled carbon nanotubes and graphene oxide. Appl Clay Sci 205:106041. https://doi.org/10.1016/J.CLAY.2021.106041
Miyashita T (2015) Confocal microscopy for intracellular co-localization of proteins. Methods Mol Biol 1278:515–526. https://doi.org/10.1007/978-1-4939-2425-7_34
Esfahani DR, Tangen KM, Sadeh M et al (2018) Systems engineers’ role in biomedical research. Convection-enhanced drug delivery. Comput Aided Chem Eng 42:271–302. https://doi.org/10.1016/B978-0-444-63964-6.00009-X
Biswas S, Dodwadkar NS, Deshpande PP, Torchilin VP (2012) Liposomes loaded with paclitaxel and modified with novel triphenylphosphonium-PEG-PE conjugate possess low toxicity, target mitochondria and demonstrate enhanced antitumor effects in vitro and in vivo. J Control Release 159:393. https://doi.org/10.1016/J.JCONREL.2012.01.009
Chen T, Chen H, Jiang Y et al (2022) Co-Delivery of 5-Fluorouracil and Paclitaxel in mitochondria-targeted KLA-modified liposomes to improve triple-negative breast cancer treatment. Pharm 15(7):881. https://doi.org/10.3390/PH15070881
Leucht S, Pitschel-Walz G, Engel RR, Kissling W (2002) Amisulpride, an unusual “atypical” antipsychotic: a meta-analysis of randomized controlled trials. Am J Psychiatry 159:180–190. https://doi.org/10.1176/APPI.AJP.159.2.180
Zielinska A, Carreiró F, Oliveira AM et al (2020) Polymeric nanoparticles: Production, characterization, toxicology and ecotoxicology. Molecules 25(16):3731. https://doi.org/10.3390/MOLECULES25163731
Fan D, Li H, Shi S, Chen X (2016) Hollow molecular imprinted polymers towards rapid, effective and selective extraction of caffeic acid from fruits. J Chromatogr A 1470:27–32. https://doi.org/10.1016/J.CHROMA.2016.10.006
Liechty WB, Kryscio DR, Slaughter BV, Peppas NA (2010) Polymers for drug delivery systems. Annu Rev Chem Biomol Eng 1:149. https://doi.org/10.1146/ANNUREV-CHEMBIOENG-073009-100847
Makadia HK, Siegel SJ (2011) Poly Lactic-co-glycolic acid (PLGA) as biodegradable controlled drug delivery carrier. Polym 3(3):1377–1397. https://doi.org/10.3390/POLYM3031377
Rukmangathen R, Yallamalli IM, Yalavarthi PR (2019) Formulation and biopharmaceutical evaluation of risperidone-loaded chitosan nanoparticles for intranasal delivery. Drug Dev Ind Pharm 45:1342–1350. https://doi.org/10.1080/03639045.2019.1619759
Panda A, Meena J, Katara R, Majumdar DK (2016) Formulation and characterization of clozapine and risperidone co-entrapped spray-dried PLGA nanoparticles. Pharm Dev Technol 21:43–53. https://doi.org/10.3109/10837450.2014.965324
Coupland JN, Hayes JE (2014) Physical Approaches to masking bitter taste: Lessons from food and pharmaceuticals. Pharm Res 31(11):2921. https://doi.org/10.1007/S11095-014-1480-6
Komossa K, Rummel-Kluge C, Schmid F et al (2009) Aripiprazole versus other atypical antipsychotics for schizophrenia. Cochrane database Syst Rev 2(4):CD006569
Masoumi HRF, Basri M, Samiun WS et al (2015) Enhancement of encapsulation efficiency of nanoemulsion-containing aripiprazole for the treatment of schizophrenia using mixture experimental design. Int J Nanomedicine 10:6469–6476. https://doi.org/10.2147/IJN.S89364
Aderibigbe BA, Naki T (2018) Design and efficacy of nanogels formulations for intranasal administration. Mol A J Synth Chem Nat Prod Chem 23(6):1241. https://doi.org/10.3390/MOLECULES23061241
Soni KS, Desale SS, Bronich TK (2016) Nanogels: an overview of properties, biomedical applications and obstacles to clinical translation. J Control Release 240:109. https://doi.org/10.1016/J.JCONREL.2015.11.009
Khan KU, Akhtar N, Minhas MU (2020) Poloxamer-407-co-poly (2-acrylamido-2-methylpropane sulfonic acid) cross-linked nanogels for solubility enhancement of olanzapine: synthesis, characterization, and toxicity evaluation. AAPS PharmSciTech 21:1–15. https://doi.org/10.1208/S12249-020-01694-0/METRICS
Daneman R, Prat A (2015) The blood–brain barrier. Cold Spring Harb Perspect Biol 7(1):a020412. https://doi.org/10.1101/CSHPERSPECT.A020412
Pardridge WM (2012) Drug transport across the blood–brain barrier. J Cereb Blood Flow Metab 32:1959. https://doi.org/10.1038/JCBFM.2012.126
Ayloo S, Gu C (2019) Transcytosis at the blood-brain barrier. Curr Opin Neurobiol 57:32. https://doi.org/10.1016/J.CONB.2018.12.014
Lei W, Yang C, Wu Y et al (2022) Nanocarriers surface engineered with cell membranes for cancer targeted chemotherapy. J Nanobiotechnology 20:45. https://doi.org/10.1186/S12951-022-01251-W
He Q, Liu J, Liang J et al (2018) Towards improvements for penetrating the blood–brain barrier—recent progress from a material and pharmaceutical perspective. Cells 7(4):24. https://doi.org/10.3390/CELLS7040024
Barar J, Rafi MA, Pourseif MM, Omidi Y (2016) Blood-brain barrier transport machineries and targeted therapy of brain diseases. Bioimpacts 6:225. https://doi.org/10.15171/BI.2016.30
Prades C, Arnould I, Annilo T et al (2002) The human ATP binding cassette gene ABCA13, located on chromosome 7p12.3, encodes a 5058 amino acid protein with an extracellular domain encoded in part by a 4.8-kb conserved exon. Cytogenet Genome Res 98:160–168. https://doi.org/10.1159/000069852
Miller DS (2015) Regulation of ABC transporters at the blood-brain barrier. Clin Pharmacol Ther 97:395. https://doi.org/10.1002/CPT.64
Knight HM, Pickard BS, Maclean A et al (2009) A cytogenetic abnormality and rare coding variants identify ABCA13 as a candidate gene in schizophrenia, bipolar disorder, and depression. Am J Hum Genet 85:833–846. https://doi.org/10.1016/J.AJHG.2009.11.003
Tomioka M, Toda Y, Kurisu J et al (2012) The effects of neurological disorder-related codon variations of ABCA13 on the function of the ABC protein. Biosci Biotechnol Biochem 76:2289–2293. https://doi.org/10.1271/BBB.120563
Chen J, Khan RAW, Wang M et al (2017) Association between the variability of the ABCA13 gene and the risk of major depressive disorder and schizophrenia in the Han Chinese population. World J Biol Psychiatry 18:550–556. https://doi.org/10.1080/15622975.2016.1245442
Qian L, Qin Y, Chen X et al (2020) ATP-binding cassette transporter 13 mRNA expression level in schizophrenia patients. Sci Reports 101(10):1–5. https://doi.org/10.1038/s41598-020-78530-9
Koepsell H (2020) Glucose transporters in brain in health and disease. Pflugers Arch 472:1299. https://doi.org/10.1007/S00424-020-02441-X
Morgello S, Uson RR, Schwartz EJ, Haber RS (1995) The human blood-brain barrier glucose transporter (GLUT1) is a glucose transporter of gray matter astrocytes. Glia 14:43–54. https://doi.org/10.1002/GLIA.440140107
Bryll A, Skrzypek J, Krzyściak W et al (2020) Oxidative-antioxidant imbalance and impaired glucose metabolism in schizophrenia. Biomolecules 10:384. https://doi.org/10.3390/BIOM10030384
Maas MB, Furie KL (2009) Molecular biomarkers in stroke diagnosis and prognosis. Biomark Med 3:363. https://doi.org/10.2217/BMM.09.30
Greene C, Kealy J, Humphries MM et al (2018) Dose-dependent expression of claudin-5 is a modifying factor in schizophrenia. Mol Psychiatry 23:2156. https://doi.org/10.1038/MP.2017.156
Yu M, Sun N, Buajieerguli M et al (2018) Glucose transporter type 1 deficiency syndrome. Chinese J Neurol 53:138–142. https://doi.org/10.3760/cma.j.issn.1006-7876.2020.02.012
Zaragozá R (2020) Transport of amino acids across the Blood-Brain Barrier. Front Physiol 11:973. https://doi.org/10.3389/FPHYS.2020.00973
Combs JA, Denicola GM (2019) The non-essential amino acid cysteine becomes essential for tumor proliferation and survival. Cancers (Basel) 11(5):678. https://doi.org/10.3390/CANCERS11050678
Nguyen HTT, Merlin D (2012) Homeostatic and innate immune responses: role of the transmembrane glycoprotein CD98. Cell Mol Life Sci 69:3015. https://doi.org/10.1007/S00018-012-0963-Z
Zafra F, Aragon C, Olivares L et al (1995) Glycine transporters are differentially expressed among CNS cells. J Neurosci 15:3952–3969. https://doi.org/10.1523/JNEUROSCI.15-05-03952.1995
Barsch L, Werdehausen R, Leffler A, Eulenburg V (2021) Modulation of glycinergic neurotransmission may contribute to the analgesic effects of propacetamol. Biomolecules 11(4):493. https://doi.org/10.3390/BIOM11040493
Kinney GG, Sur C, Burno M et al (2003) The glycine transporter type 1 inhibitor N-[3-(4′-fluorophenyl)-3-(4′-phenylphenoxy)propyl]sarcosine potentiates NMDA receptor-mediated responses in vivo and produces an antipsychotic profile in rodent behavior. J Neurosci 23:7586. https://doi.org/10.1523/JNEUROSCI.23-20-07586.2003
Hopkins CR (2011) ACS chemical neuroscience molecule spotlight on RG1678. ACS Chem Neurosci 2:685–686. https://doi.org/10.1021/CN200108Z/ASSET/IMAGES/LARGE/CN-2011-00108Z_0001.JPEG
Moons T, De Roo M, Claes S, Dom G (2011) Relationship between P-glycoprotein and second-generation antipsychotics. Pharmacogenomics 12:1193–1211. https://doi.org/10.2217/PGS.11.55
Mao Q, Unadkat JD (2015) Role of the breast cancer resistance protein (BCRP/ABCG2) in drug transport—an update. AAPS J 17:65. https://doi.org/10.1208/S12248-014-9668-6
Sodani K, Patel A, Kathawala RJ, Chen ZS (2012) Multidrug resistance associated proteins in multidrug resistance. Chin J Cancer 31:58. https://doi.org/10.5732/CJC.011.10329
Lee WK, Jung SM, Kwak JO, Cha SH (2006) Introduction of organic anion transporters (SLC22A) and a regulatory mechanism by caveolins. Electrolytes Blood Press E BP 4:8. https://doi.org/10.5049/EBP.2006.4.1.8
Roth M, Obaidat A, Hagenbuch B (2012) OATPs, OATs and OCTs: the organic anion and cation transporters of the SLCO and SLC22A gene superfamilies. Br J Pharmacol 165:1260. https://doi.org/10.1111/J.1476-5381.2011.01724.X
Wagner DJ, Hu T, Wang J (2016) Polyspecific organic cation transporters and their impact on drug intracellular levels and pharmacodynamics. Pharmacol Res 111:237. https://doi.org/10.1016/J.PHRS.2016.06.002
Vijay N, Morris ME (2014) Role of monocarboxylate transporters in drug delivery to the brain. Curr Pharm Des 20:1487. https://doi.org/10.2174/13816128113199990462
Lee JH, Lee JE, Kim Y et al (2014) Multidrug and toxic compound extrusion protein-1 (MATE1/SLC47A1) is a novel flavonoid transporter. J Agric Food Chem 62:9690–9698. https://doi.org/10.1021/JF500916D
Blanco-Vázquez C, Alonso-Hearn M, Iglesias N et al (2022) Use of ATP-binding cassette subfamily A member 13 (ABCA13) for sensitive detection of focal pathological forms of subclinical bovine paratuberculosis. Front Vet Sci 9:126. https://doi.org/10.3389/FVETS.2022.816135/BIBTEX
Pragallapati S, Manyam R (2019) Glucose transporter 1 in health and disease. J Oral Maxillofac Pathol 23:443. https://doi.org/10.4103/JOMFP.JOMFP_22_18
Meier C, Ristic Z, Klauser S, Verrey F (2002) Activation of system L heterodimeric amino acid exchangers by intracellular substrates. EMBO J 21:580. https://doi.org/10.1093/EMBOJ/21.4.580
Adams RH, Sato K, Shimada S et al (1995) Gene structure and glial expression of the glycine transporter GlyT1 in embryonic and adult rodents. J Neurosci 15:2524–2532. https://doi.org/10.1523/JNEUROSCI.15-03-02524.1995
Khan A, Ostaku J, Aras E, Safak Seker UO (2022) Combating infectious diseases with synthetic biology. ACS Synth Biol 11:528–537. https://doi.org/10.1021/ACSSYNBIO.1C00576/ASSET/IMAGES/LARGE/SB1C00576_0004.JPEG
Greenbaum U, Mahadeo KM, Kebriaei P et al (2020) Chimeric antigen receptor T-cells in B-acute lymphoblastic leukemia: state of the art and future directions. Front Oncol 10:1594. https://doi.org/10.3389/FONC.2020.01594
Bălașa AF, Chircov C, Grumezescu AM (2020) Marine biocompounds for neuroprotection—a review. Mar Drugs 18(6):290. https://doi.org/10.3390/MD18060290
Choudhuri S, Cui Y, Klaassen CD (2010) Molecular targets of epigenetic regulation and effectors of environmental influences. Toxicol Appl Pharmacol 245:378. https://doi.org/10.1016/J.TAAP.2010.03.022
Eshraghi M, Adlimoghaddam A, Mahmoodzadeh A et al (2021) Alzheimer’s disease pathogenesis: role of autophagy and mitophagy focusing in microglia. Int J Mol Sci 22(7):3330. https://doi.org/10.3390/IJMS22073330
De Oliveira J, Kucharska E, Garcez ML et al (2021) Inflammatory cascade in Alzheimer’s disease pathogenesis: a review of experimental findings. Cells 10(10):2581. https://doi.org/10.3390/CELLS10102581
Azizian K, Pustokhina I, Ghanavati R et al (2021) The potential use of theranostic bacteria in cancer. J Cell Physiol 236:4184–4194. https://doi.org/10.1002/JCP.30152
Wang L, Hu C, Shao L (2017) The antimicrobial activity of nanoparticles: present situation and prospects for the future. Int J Nanomedicine 12:1227. https://doi.org/10.2147/IJN.S121956
Suh SB, Jo A, Traore MA et al (2019) Nanoscale bacteria-enabled autonomous drug delivery system (nanoBEADS) enhances intratumoral transport of nanomedicine. Adv Sci 6:1801309. https://doi.org/10.1002/ADVS.201801309
Ye Z, Liang L, Lu H et al (2021) Nanotechnology-employed bacteria-based delivery strategy for enhanced anticancer therapy. Int J Nanomedicine 16:8069. https://doi.org/10.2147/IJN.S329855
Wang S, Hossack JA, Klibanov AL (2018) Targeting of microbubbles - contrast agents for ultrasound molecular imaging. J Drug Target 26:420. https://doi.org/10.1080/1061186X.2017.1419362
Aryal M, Vykhodtseva N, Zhang YZ, McDannold N (2015) Multiple sessions of liposomal doxorubicin delivery via focused ultrasound mediated blood-brain barrier disruption: a safety study. J Control Release 204:60. https://doi.org/10.1016/J.JCONREL.2015.02.033
Kamaly N, Yameen B, Wu J, Farokhzad OC (2016) Degradable controlled-release polymers and polymeric nanoparticles: mechanisms of controlling drug release. Chem Rev 116:2602–2663. https://doi.org/10.1021/ACS.CHEMREV.5B00346/ASSET/IMAGES/ACS.CHEMREV.5B00346.SOCIAL.JPEG_V03
Karimzadeh M, Rashidi L, Ganji F (2017) Mesoporous silica nanoparticles for efficient rivastigmine hydrogen tartrate delivery into SY5Y cells. Drug Dev Ind Pharm 43:628–636. https://doi.org/10.1080/03639045.2016.1275668
Meng F, Han N, Yeo Y (2017) Organic nanoparticle systems for spatiotemporal control of multimodalchemotherapy. Expert Opin Drug Deliv 14:427. https://doi.org/10.1080/17425247.2016.1218464
Petusseau AF, Bruza P, Pogue BW (2022) Protoporphyrin IX delayed fluorescence imaging: a modality for hypoxia-based surgical guidance. J Biomed Opt 27(10):106005. https://doi.org/10.1117/1.JBO.27.10.106005
Hammerer F, Poyer F, Fourmois L et al (2018) Mitochondria-targeted cationic porphyrin-triphenylamine hybrids for enhanced two-photon photodynamic therapy. Bioorganic Med Chem 26:107–118. https://doi.org/10.1016/J.BMC.2017.11.024
Xu S, Luo S, Yao H et al (2016) Probing the anticancer action of oridonin with fluorescent analogues: visualizing subcellular localization to mitochondria. J Med Chem 59:5022–5034. https://doi.org/10.1021/ACS.JMEDCHEM.6B00408
Gao Y, Tong H, Li J et al (2021) Mitochondria-targeted nanomedicine for enhanced efficacy of cancer therapy. Front Bioeng Biotechnol 9:682. https://doi.org/10.3389/FBIOE.2021.720508/BIBTEX
Zorkina Y, Abramova O, Ushakova V et al (2020) Nano carrier drug delivery systems for the treatment of neuropsychiatric disorders: advantages and limitations. Molecules 25(22):5294. https://doi.org/10.3390/MOLECULES25225294
Khayal A, Dawane V, Amin MA et al (2021) Advances in the methods for the synthesis of Carbon dots and their emerging applications. Polymers (Basel) 13(18):3190. https://doi.org/10.3390/POLYM13183190
Saboktakin M (2019) Polymeric Nanoencapsulation of indocyanine green for photodynamic therapy technique. Am J Biomed Sci Res 3:35–48. https://doi.org/10.34297/AJBSR.2019.03.000630
Collins L, Binder P, Chen H, Wang X (2020) Regulation of long non-coding RNAs and MicroRNAs in heart disease: insight into mechanisms and therapeutic approaches. Front Physiol 11:798. https://doi.org/10.3389/FPHYS.2020.00798
O’Brien J, Hayder H, Zayed Y, Peng C (2018) Overview of microRNA biogenesis, mechanisms of actions, and circulation. Front Endocrinol (Lausanne) 9:402. https://doi.org/10.3389/FENDO.2018.00402/BIBTEX
Saito T, Sadoshima J (2015) The molecular mechanisms of mitochondrial autophagy/mitophagy in theheart. Circ Res 116:1477. https://doi.org/10.1161/CIRCRESAHA.116.303790
Gozuacik D, Akkoc Y, Gulfem Ozturk D, Kocak M (2017) Autophagy-regulating microRNAs and cancer. Front. Oncol 7:1. https://doi.org/10.3389/FONC.2017.00065
Zhou L, Liu S, Han M et al (2017) MicroRNA-185 induces potent autophagy via AKT signaling in hepatocellular carcinoma. Tumor Biol 39(2):1–13. https://doi.org/10.1177/1010428317694313
Song J, Oh Y, Lee JE (2015) miR-Let7A modulates autophagy induction in LPS-activated microglia. Exp Neurobiol 24:117. https://doi.org/10.5607/EN.2015.24.2.117
Dutta P, Haller E, Sharp A, Nanjundan M (2016) MIR494 reduces renal cancer cell survival coinciding with increased lipid droplets and mitochondrial changes. BMC Cancer 16(1):33. https://doi.org/10.1186/S12885-016-2053-3
Yang J, Rao S, Cao R et al (2021) miR-30a-5p suppresses lung squamous cell carcinoma via ATG5 - mediated autophagy. Aging 9Albany NY) 13:17462. https://doi.org/10.18632/AGING.203235
Xu L, Hui AY, Albanis E et al (2005) Human hepatic stellate cell lines, LX-1 and LX-2: new tools for analysis of hepatic fibrosis. Gut 54:142. https://doi.org/10.1136/GUT.2004.042127
Lan SH, Lin SC, Wang WC et al (2021) Autophagy upregulates miR-449a expression to suppress progression of colorectal cancer. Front Oncol 11:738144. https://doi.org/10.3389/FONC.2021.738144/FULL
Huang N, Wu J, Qiu W et al (2015) MiR-15a and miR-16 induce autophagy and enhance chemosensitivity of camptothecin. Cancer Biol Ther 16:941. https://doi.org/10.1080/15384047.2015.1040963
Tian L, Ma L, Guo E et al (2013) 20-Hydroxyecdysone upregulates Atg genes to induce autophagy in the Bombyx fat body. Autophagy 9:1172–1187. https://doi.org/10.4161/AUTO.24731
Yuan Y, Zhang Y, Han L et al (2018) MiR-183 inhibits autophagy and apoptosis in gastric cancer cells by targeting ultraviolet radiation resistance-associated gene. Int J Mol Med 42:3562–3570. https://doi.org/10.3892/IJMM.2018.3871/HTML
Acknowledgements
The study was successful due to the moral courage of the HOD, IPR, GLA University.
Funding
This study is financially supported by GLA University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics Approval
NA.
Consent to Participate
NA.
Consent for Publication
Both authors approved the manuscript.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Both authors contributed equally to the manuscript.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Panda, S.P., Singh, V. The Dysregulated MAD in Mad: A Neuro-theranostic Approach Through the Induction of Autophagic Biomarkers LC3B-II and ATG. Mol Neurobiol 60, 5214–5236 (2023). https://doi.org/10.1007/s12035-023-03402-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-023-03402-y