Abstract
Diospyros peregrina is a dioecious plant which is native to India. It belongs to the family of Ebenaceae and is extensively used to treat various ailments, such as leucorrhoea and other uterine-related problems. Though few studies have been on D. peregrina for their anti-tumour response, little is known. Therefore, this intrigued us to understand its immunomodulator capabilities on various types of cancer extensively. Our primary focus is on NSCLC (Non-Small Cell Lung Cancer), which is ranked as the second largest form of cancer in the world, and the treatments demand non-invasive agents to target NSCLC effectively. In an objective to generate an efficient Lung Cancer Associated Antigen (LCA) specific anti-tumour immune response, LCA was presented using dendritic cells (DCs) in the presence of D. peregrina fruit preparation (DFP). Moreover, we also investigated DFP’s role in the differentiation of T-helper (TH) cells. Therefore, this study aimed at better LCA presentation mediated by DFP by activating the LCA pulsed DCs and T helper cell differentiation for better immune response. DCs were pulsed with LCA for tumour antigen presentation in vitro, with and without DFP. Differentially pulsed DCs were irradiated to co-culture with autologous and allogeneic lymphocytes. Extracellular supernatants were collected for the estimation of cytokine levels by ELISA. LDH release assay was performed to test Cytotoxic T lymphocytes (CTLs) mediated lung tumour cell cytotoxicity. Thus, DFP may be a potential vaccine to generate anti-LCA immune responses to restrict NSCLC.
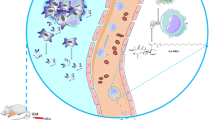
Graphical Abstract
A graphical representation of the cascade of events pertaining to the augmentation of the process of cancer antigen presentation, TH cell differentiation and subsequent tumour cell cytotoxicity in presence or absence of DFP








Similar content being viewed by others
Data availability
Data are available on request from the authors.
Abbreviations
- NSCLC:
-
Non-small cell lung cancer
- LCA:
-
Lung cancer associated antigens
- DFP:
-
Diospyros peregrina Fruit preparation
- DC:
-
Dendritic cell
- TH :
-
T helper
- ELISA:
-
Enzyme-linked immunosorbent assay
- LDH:
-
Lactate dehydrogenase
- CTL:
-
Cytotoxic T lymphocytes
- Treg:
-
Regulatory T cells
- TLR:
-
Toll-like receptors
- IL:
-
Interleukin
- PBMC:
-
Peripheral blood mononuclear cells
- rhGM-CSF:
-
Recombinant human granulocyte–macrophage colony-stimulating factor
- PBS:
-
Phosphate buffer saline
- TMB:
-
3,3′,5,5′-Tetramethylbenzidine
- HRP:
-
Horseradish peroxidase
- RT-qPCR:
-
Real-time quantitative polymerase chain reaction
- SDS-PAGE:
-
Sodium dodecyl sulphate polyacrylamide gel electrophoresis
- BSA:
-
Bovine serum albumin
- PBST:
-
Phosphate buffer saline tween-20
References
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: A Cancer J Clin. 2018;68(6):394–424. https://doi.org/10.3322/caac.21492.
Ferlay J, Colombet M, Soerjomataram I, Parkin DM, Piñeros M, Znaor A, Bray F. Cancer statistics for the year 2020: an overview. Int J Cancer. 2021;149(4):778–89. https://doi.org/10.1002/ijc.33588.
View of Global Cancer Statistics: The trends projection analysis. (n.d.). 2022 https://pubs.thesciencein.org/journal/index.php/cbl/article/view/451/293
Gridelli C, Rossi A, Carbone DP, Guarize J, Karachaliou N, Mok T, Petrella F, Spaggiari L, Rosell R. Non-small-cell lung cancer. Natre Rev Dis Prim. 2015. https://doi.org/10.1038/nrdp.2015.9.
Duma N, Santana-Davila R, Molina JR. Non-small cell lung cancer: epidemiology, screening, diagnosis, and treatment. Mayo Clin Proc. 2019;94(8):1623–40. https://doi.org/10.1016/j.mayocp.2019.01.013.
Schabath MB, Cote ML. Cancer progress and priorities: lung cancer. Cancer Epidemiol, Biomark Prev. 2019;28(10):1563–79. https://doi.org/10.1158/1055-9965.epi-19-0221.
Colby TV, Wistuba II, Gazdar A. Precursors to pulmonary neoplasia. Adv Anatomic Pathol. 1998;5(4):205–15. https://doi.org/10.1097/00125480-199807000-00001.
Yano T, Haro A, Shikada Y, Maruyama R, Maehara Y. Non-small cell lung cancer in never smokers as a representative ‘non-smoking-associated lung cancer’: epidemiology and clinical features. Int J Clin Oncol. 2011;16(4):287–93. https://doi.org/10.1007/s10147-010-0160-8.
Balan S, Saxena M, Bhardwaj N. Dendritic cell subsets and locations. Int Rev Cell Mol Biol. 2019. https://doi.org/10.1016/bs.ircmb.2019.07.004.
Cassetta L, Kitamura T. Macrophage targeting: opening new possibilities for cancer immunotherapy. Immunology. 2018;155(3):285–93. https://doi.org/10.1111/imm.12976.
Szabo SJ, Sullivan BM, Peng SL, Glimcher LH. Molecular mechanisms regulating Th1 immune responses. Annu Rev Immunol. 2003;21(1):713–58. https://doi.org/10.1146/annurev.immunol.21.120601.140942.
Abbas AK, Murphy KM, Sher A. Functional diversity of helper T lymphocytes. Nature. 1996;383(6603):787–93. https://doi.org/10.1038/383787a0.
Biswal S, Mehta R. Cutaneous adverse reactions of chemotherapy in cancer patients: a clinicoepidemiological study. Ind J Dermatol. 2018;63(1):41. https://doi.org/10.4103/ijd.ijd_65_17.
Sun LX, Li WD, Lin ZB, Duan XS, Li XF, Yang N, Lan TF, Li M, Sun Y, Yu M, Lu J. Protection Against Lung Cancer Patient Plasma-Induced Lymphocyte Suppression by Ganoderma LucidumPolysaccharides. Cell Physiol Biochem. 2014;33(2):289–99. https://doi.org/10.1159/000356669.
Peschel W, Kump A, Prieto JM. Effects of 20-hydroxyecdysone, Leuzea carthamoides extracts, dexamethasone and their combinations on the NF-κB activation in HeLa cells. J Pharm Pharmacol. 2011;63(11):1483–95. https://doi.org/10.1111/j.2042-7158.2011.01349.x.
Kaushik RR, Koranne M, Rao MS, Rakshit S, Shanmugam G, George M, Sarkar K. Role of Diospyros peregrina fruit preparation in suppressing regulatory T (Treg) cells in the tumor microenvironment of breast and lung cancer. Phytomedicine Plus. 2022;2(4):100353. https://doi.org/10.1016/j.phyplu.2022.100353.
Y, A, G, B, SA. (2012). Antitumor and antioxidant activity of Diospyros peregrina against Dalton’s ascites lymphoma in rodents. Annal Biol Res. 2012; 3 11:4985–4992. https://www.scholarsresearchlibrary.com/articles/antitumor-and-antioxidant-activity-of-diospyros-peregrina-against-daltonsascites-lymphoma-in-rodents.pdf. Accessed 22 from 2023
Raju AB, Venu Gopal Y, Ravindranath A, Kalpana G, Prabhakar RV. Antitumor activity of Diospyros peregrina on ehrlich ascites carcinoma in mice. J Sci Res. 2011;3(2):413–9. https://doi.org/10.3329/jsr.v3i2.6787.
Roy A, Ghosh A, Sinha K, Mitra B, Rakshit S, George M, Sarkar K. Diospyros peregrina fruit preparation mediated immunomodulation of lymphocytes isolated from the blood of breast cancer patients. Iran J Immunol. 2021;18(2):111–8. https://doi.org/10.22034/iji.2021.85430.1710.
Mahalakshmi Surendran A, Rai A, Rakshit S, George M, Sarkar K. Immunomodulatory role of Diospyros peregrina fruit preparation in breast cancer by utilizing macrophage mediated antigen presentation and T helper cell (Th) differentiation. Clin Breast Cancer. 2023;23(3):e95–102. https://doi.org/10.1016/j.clbc.2022.12.020.
Bøyum A. Separation of blood leucocytes, granulocytes and lymphocytes. Tissue Antigens. 1974;4(3):269–74. https://doi.org/10.1111/j.1399-0039.1974.tb00252.x.
Petersen TR, Dickgreber N, Hermans IF. Tumor antigen presentation by dendritic cells. Crit Rev™ in Immunol. 2010;30(4):345–86. https://doi.org/10.1615/critrevimmunol.v30.i4.30.
Sarkar K, Bose A, Chakraborty K, Haque E, Ghosh D, Goswami S, Chakraborty T, Laskar S, Baral R. Neem leaf glycoprotein helps to generate carcinoembryonic antigen specific anti-tumor immune responses utilizing macrophage-mediated antigen presentation. Vaccine. 2008;26(34):4352–62. https://doi.org/10.1016/j.vaccine.2008.06.048.
Mukherjee O, Paul S, Das S, Rakshit S, Shanmugam G, George M, Sarkar K. Doxorubicin induced epigenetic regulation of dendritic cell maturation in association with T cell activation facilitates tumor protective immune response in non-small cell lung cancer (NSCLC). Pathol—Res Pract. 2024;253:155004. https://doi.org/10.1016/j.prp.2023.155004.
Li Y, Zhao C, Liu J, Lu Z, Lu M, Gu J, Liu R. CD1d highly expressed on DCs reduces lung tumor burden by enhancing antitumor immunity. Oncol Rep. 2019. https://doi.org/10.3892/or.2019.7037.
Fried S, Tosun S, Troost G, Keil S, Zaenker KS, Dittmar T. Lipopolysaccharide (LPS) promotes apoptosis in human breast epithelial × breast cancer hybrids, but not in parental cells. PLoS ONE. 2016;11(2):e0148438. https://doi.org/10.1371/journal.pone.0148438.
Janeway AJC, Travers P, Walport M, Shlomchik, MJ. (2001, January 1). T cell-mediated cytotoxicity—Immunobiology—NCBI Bookshelf. T Cell-mediated Cytotoxicity—Immunobiology - NCBI Bookshelf. https://www.ncbi.nlm.nih.gov/books/NBK27101/
Sabado RL, Balan S, Bhardwaj N. Dendritic cell-based immunotherapy. Cell Res. 2016;27(1):74–95. https://doi.org/10.1038/cr.2016.157.
Faget J, Biota C, Bachelot T, Gobert M, Treilleux I, Goutagny N, Durand I, Léon-Goddard S, Blay JY, Caux C, Ménétrier-Caux C. Early detection of tumor cells by innate immune cells leads to treg recruitment through CCL22 production by tumor cells. Cancer Res. 2011;71(19):6143–52. https://doi.org/10.1158/0008-5472.can-11-0573.
Tokumaru Y, Le L, Oshi M, Katsuta E, Matsuhashi N, Futamura M, Yoshida K, Takabe K. Association of Th2 high tumors with aggressive features of breast cancer. J Clin Oncol. 2020;38:e12584–e12584.
Frafjord A, Buer L, Hammarström C, Aamodt H, Woldbæk PR, Brustugun OT, Helland S, Øynebråten I, Corthay A. The immune landscape of human primary lung tumors is Th2 skewed. Front Immunol. 2021. https://doi.org/10.3389/fimmu.2021.764596.
Chang SH, Mirabolfathinejad SG, Katta H, Cumpian AM, Gong L, Caetano MS, Moghaddam SJ, Dong C. T helper 17 cells play a critical pathogenic role in lung cancer. Proc Natl Acad Sci. 2014;111(15):5664–9. https://doi.org/10.1073/pnas.1319051111.
Indigenous Drugs of India. The indian medical gazette. 1892. 27 9:271–273. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5136941/
Mallavadhani U, Panda AK, Rao Y. Review article number 134 pharmacology and chemotaxonomy of diospyros. Phytochemistry. 1998;49(4):901–51. https://doi.org/10.1016/s0031-9422(97)01020-0.
Dewanjee S, Kundu M, Maiti A, Majumdar R, Majumdar A, Mandel SC. In vitro evaluation of antimicrobial activity of crude extract from plants Diospyros peregrine, Coccinia grandis and Swietenia macrophylla. Trop J Pharm Res. 2007. https://doi.org/10.4314/tjpr.v6i3.14658.
Dewanjee S, Das AK, Sahu R, Gangopadhyay M. Antidiabetic activity of Diospyros peregrina fruit: effect on hyperglycemia, hyperlipidemia and augmented oxidative stress in experimental type 2 diabetes. Food Chem Toxicol. 2009;47(10):2679–85. https://doi.org/10.1016/j.fct.2009.07.038.
Alex AT, Nawagamuwa NH, Joseph A, Rao JV, Mathew JA, Udupa N. In vitro anti-cancer and anti-oxidant activity of different fractions of Diospyros peregrina unripe fruit extract. Free Radic Antioxid. 2012;2(4):45–9. https://doi.org/10.5530/ax.2012.4.8.
Bhootra S, Jill N, Rajak R, Shanmugam G, Rakshit S, Kannanthodi S, Thakkar V, George M, Sarkar K. Diospyros malabarica fruit preparation mediates immunotherapeutic modulation and epigenetic regulation to evoke protection against non–small cell lung cancer (NSCLC). J Ethnopharmacol. 2023;314:116525. https://doi.org/10.1016/j.jep.2023.116525.
Gerlini G, Hefti HP, Kleinhans M, Nickoloff BJ, Burg G, Nestle FO. CD1d is expressed on dermal dendritic cells and monocyte-derived dendritic cells. J Investig Dermatol. 2001;117(3):576–82. https://doi.org/10.1046/j.0022-202x.2001.01458.x.
Wan CK, Andraski AB, Spolski R, Li P, Kazemian M, Oh J, Samsel L, Swanson PA, McGavern DB, Sampaio EP, Freeman AF, Milner JD, Holland SM, Leonard WJ. Opposing roles of STAT1 and STAT3 in IL-21 function in CD4 + T cells. Proc Natl Acad Sci. 2015;112(30):9394–9. https://doi.org/10.1073/pnas.1511711112.
Jiang X, Du W, Yang C, Wang S, Li Y, Shen X, Yang X, Yao J, Du R, Zhang X, Huang Y, Shen W. TBX21 attenuates colorectal cancer progression via an ARHGAP29/RSK/GSK3β dependent manner. Cell Oncol. 2023;46(5):1269–83. https://doi.org/10.1007/s13402-023-00809-6.
Katerina Andreev I, Trufa DI, Siegemund R, Rieker R, Hartmann A, Schmidt J, Sirbu H, Finotto S. Impaired T-bet-pSTAT1α and perforin-mediated immune responses in the tumoral region of lung adenocarcinoma. Br J Cancer. 2015;113(6):902–13. https://doi.org/10.1038/bjc.2015.255.
Thieu VT, Yu Q, Chang HC, Yeh N, Nguyen ET, Sehra S, Kaplan MH. Signal transducer and activator of transcription 4 is required for the transcription factor T-bet to promote T helper 1 cell-fate determination. Immunity. 2008;29(5):679–90. https://doi.org/10.1016/j.immuni.2008.08.017.
Zhu J, Yamane H, Cote-Sierra J, et al. GATA-3 promotes Th2 responses through three different mechanisms: induction of Th2 cytokine production, selective growth of Th2 cells and inhibition of Th1 cell-specific factors. Cell Res. 2006;16:3–10. https://doi.org/10.1038/sj.cr.7310002.
Unutmaz D. RORC2: The master of human Th17 cell programming. Eur J Immunol. 2009;39(6):1452–5. https://doi.org/10.1002/eji.200939540.
Tao JH, Cheng M, Tang JP, et al. Foxp3, regulatory T cell, and autoimmune diseases. Inflammation. 2017;40:328–39. https://doi.org/10.1007/s10753-016-0470-8.
Yang H, Yao F, Marti TM, Schmid RA, Peng RW. Beyond DNA repair: DNA-PKcs in tumor metastasis. Metab Immun Cancers. 2020;12(11):3389. https://doi.org/10.3390/cancers12113389.
Zinatizadeh MR, Schock B, Chalbatani GM, Zarandi PK, Jalali SA, Miri SR. The nuclear factor kappa B (NF-kB) signaling in cancer development and immune diseases. Genes Dis. 2021;8(3):287–97. https://doi.org/10.1016/j.gendis.2020.06.005.
Acknowledgements
We thank DST-FIST Facility of Department of Biotechnology at SRM Institute of Science and Technology, Kattankulathur, India for allowing us to perform flow cytometry on BD FACS Melody™ cell sorter.
Funding
This research was supported by Science and Engineering Research Board (SERB), Department of Science and Technology (DST), Govt. of India (sanction order No. ECR/2016/000965).
Author information
Authors and Affiliations
Contributions
NS and AS: investigation, data curation, writing–original draft. SN and NM: investigation, data curation, writing–original draft. OM and PR: standardization of techniques. MG: resource person. KS: conceptualization, writing–review & editing, project administration, supervision, funding acquisition.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethical approval
For molecular studies involved in the current research project, blood samples of normal subjects and NSCLC patients were collected and handled under the protocol approved by the institutional ethics committee at SRM Medical College Hospital, Kattankulathur, Chennai, Tamil Nadu, India (Ethics Clearance Number 2909/IEC/2021).
Informed consent
Signed informed consent was collected from each NSCLC patient and normal individuals who took part in the current study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sriraman, N., Sarkar, A., Naskar, S. et al. Immunomodulatory effects of Diospyros peregrina fruit preparation (DFP) in non-small cell lung cancer (NSCLC) by utilizing dendritic cell-mediated antigen presentation and T helper (TH) cell differentiation. Med Oncol 41, 107 (2024). https://doi.org/10.1007/s12032-024-02331-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-024-02331-7