Abstract
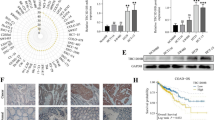
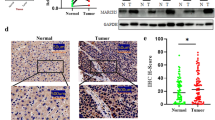
SOX genes play an important role in a number of developmental processes. SOXs have been demonstrated to have potential roles as either tumor suppressors or promoters in various neoplastic tissues depending on the tumor status and type. The aim of this study was to investigate the functional role of SOXs in human cancers. Gene expression changes of SOXs in human hepatocellular carcinoma (HCC) tissues were detected using real-time quantitative reverse transcriptase-polymerase chain reaction (qRT-PCR) analysis and immunohistochemistry and compared with those in non-cancerous hepatic tissues. We found by qRT-PCR analysis and immunohistochemistry that the gene SOX8 was significantly upregulated in HCC. Furthermore, we discovered that SOX8 promoted cancer cell proliferation in vitro and that its expression was correlated with elevated β-catenin levels in HCC, whose function was required for the oncogenic effects of SOX8.



Similar content being viewed by others
References
Tanaka S, Arii S. Molecular targeted therapies in hepatocellular carcinoma. Semin Oncol. 2012;39(4):486–92.
Vinas A, et al. Map** of DNA sex-specific markers and genes related to sex differentiation in turbot (Scophthalmus maximus). Mar Biotechnol (NY). 2012;14(5):655–63.
Ng CK, et al. Deciphering the Sox–Oct partner code by quantitative cooperativity measurements. Nucleic Acids Res. 2012;40(11):4933–41.
Coco C, et al. Increased expression of CD133 and reduced dystroglycan expression are strong predictors of poor outcome in colon cancer patients. J Exp Clin Cancer Res. 2012;31:71.
Zhang W, et al. Epigenetic inactivation of the canonical Wnt antagonist SRY-box containing gene 17 in colorectal cancer. Cancer Res. 2008;68(8):2764–72.
Scott EL, Brann DW. Estrogen regulation of Dkk1 and Wnt/beta-catenin signaling in neurodegenerative disease. Brain Res. 2013;1514:63–74.
Laudet V, Stehelin D, Clevers H. Ancestry and diversity of the HMG box superfamily. Nucleic Acids Res. 1993;21(10):2493–501.
Cao JM, et al. High mobility group B proteins regulate mesoderm formation and dorsoventral patterning during zebrafish and Xenopus early development. Mech Dev. 2012;129(9–12):263–74.
Marchetti B, et al. Uncovering novel actors in astrocyte-neuron crosstalk in Parkinson’s disease: the Wnt/beta-catenin signaling cascade as the common final pathway for neuroprotection and self-repair. Eur J Neurosci. 2013;37(10):1550–63.
Hu S, et al. Sox31 is involved in central nervous system anteroposterior regionalization through regulating the organizer activity in zebrafish. Acta Biochim Biophys Sin (Shanghai). 2011;43(5):387–99.
Aksoy I, et al. Sox transcription factors require selective interactions with Oct4 and specific transactivation functions to mediate reprogramming. Stem Cells. 2013;31(12):2632–46.
Jiang T, et al. The SOX gene family: function and regulation in testis determination and male fertility maintenance. Mol Biol Rep. 2013;40(3):2187–94.
Goding CR. Melanocyte development and malignant melanoma. Forum (Genova). 2000;10(3):176–87.
Soullier S, et al. Diversification pattern of the HMG and SOX family members during evolution. J Mol Evol. 1999;48(5):517–27.
Argenton F, et al. Ectopic expression and knockdown of a zebrafish sox21 reveal its role as a transcriptional repressor in early development. Mech Dev. 2004;121(2):131–42.
Juarez M, et al. Characterization of the trunk neural crest in the bamboo shark, Chiloscyllium punctatum. J Comp Neurol. 2013;521(14):3303–20.
Lakiza O, et al. SoxE gene duplication and development of the lamprey branchial skeleton: insights into development and evolution of the neural crest. Dev Biol. 2011;359(1):149–61.
Lavery R, et al. Testicular differentiation occurs in absence of R-spondin1 and Sox9 in mouse sex reversals. PLoS Genet. 2012;8(12):e1003170.
Klaus A, Birchmeier W. Wnt signalling and its impact on development and cancer. Nat Rev Cancer. 2008;8(5):387–98.
Liu Q, et al. Homologues of sox8 and sox10 in the orange-spotted grouper Epinephelus coioides: sequences, expression patterns, and their effects on cyp19a1a promoter activities in vitro. Comp Biochem Physiol B: Biochem Mol Biol. 2012;163(1):86–95.
Baarsma HA, et al. Activation of WNT/beta-catenin signaling in pulmonary fibroblasts by TGF-beta(1) is increased in chronic obstructive pulmonary disease. PLoS ONE. 2011;6(9):e25450.
Vijayakumar S, et al. High-frequency canonical Wnt activation in multiple sarcoma subtypes drives proliferation through a TCF/beta-catenin target gene, CDC25A. Cancer Cell. 2011;19(5):601–12.
Kormish JD, Sinner D, Zorn AM. Interactions between SOX factors and Wnt/beta-catenin signaling in development and disease. Dev Dyn. 2010;239(1):56–68.
Korbut E, Ptak-Belowska A, Brzozowski T. Mechanisms promoting physiological cells progression into tumorigenesis. J Physiol Pharmacol. 2012;63(6):565–70.
Baylin SB, Ohm JE. Epigenetic gene silencing in cancer—a mechanism for early oncogenic pathway addiction? Nat Rev Cancer. 2006;6(2):107–16.
L’Episcopo F, et al. Reactive astrocytes and Wnt/beta-catenin signaling link nigrostriatal injury to repair in 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine model of Parkinson’s disease. Neurobiol Dis. 2011;41(2):508–27.
Bafico A, et al. An autocrine mechanism for constitutive Wnt pathway activation in human cancer cells. Cancer Cell. 2004;6(5):497–506.
Liu F, Millar SE. Wnt/beta-catenin signaling in oral tissue development and disease. J Dent Res. 2010;89(4):318–30.
Acknowledgments
We thank Ms. Min Ding, Dr. Yumin Chen and Dr. Melissa Cruz of Institute of Shanghai Springermedia for a critical review of the manuscript and the scholar services of After Tumor technology (AfterTumor.com). This work was supported by Foundation of Science Technology Department of Zhejiang Province, China (No. 2012C23072), Natural Science Foundation of Zhejiang Province, China (No. Y2091061), Medical and health science and technology Foundation of Zhejiang Province, China (No. 2013KYA170) and Foundation of Science Technology Department of Hangzhou, Zhejiang Province, China (No. 20110733Q04).
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
Siquan Zhang and Cong Zhu have contributed equally to this work.
Rights and permissions
About this article
Cite this article
Zhang, S., Zhu, C., Zhu, L. et al. Oncogenicity of the transcription factor SOX8 in hepatocellular carcinoma. Med Oncol 31, 918 (2014). https://doi.org/10.1007/s12032-014-0918-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-014-0918-3