Abstract
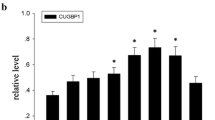
E3 ubiquitin ligase SIAH1 is a protein associated with the onset of nontumorigenicy in revertant tumorigenic cell lines and with several apoptotic processes. However, its role in the injury of the central nervous system remains unknown. In this study, we performed acute spinal cord injury (SCI) in adult rats and investigated the protein expression and cellular localization of SIAH1 in the spinal cord. Western blot analysis revealed that SIAH1 was low expressed in normal spinal cord. It increased at 8 h after SCI, peaked at 1 day, remained for another 3 days, then declined to basal levels at 5 days after injury. Immunohistochemistry further confirmed that SIAH1 immunoactivity was expressed at low levels in gray and white matters in normal condition and increased after SCI. Double immunofluorescence staining showed that SIAH1 was coexpressed with NeuN (neuronal marker), CNPase (oligodendroglial marker), GFAP (astroglial marker), and CD11b (microglial marker) at 1 day post-injury and was also coexpressed with active caspase-3 in neurons and glial cells after injury. In addition, double immunofluorescence staining indicated that p-c-Jun NH2-kinase (JNK) coexpressed with SIAH1 in neurons and glial cells. Coimmunoprecipitation further showed that p-JNK and SIAH1 precipitated with each other in the damaged spinal cord. Taken together, these data suggest SIAH1 involvement in the injury response of the adult spinal cord of the rats.






Similar content being viewed by others
References
Barr RK, Bogoyevitch MA (2001) The c-Jun N-terminal protein kinase family of mitogen-activated protein kinases (JNK MAPKs). Int J Biochem Cell Biol 33:1047–1063
Bodles AM, Barger SW (2005) Secreted beta-amyloid precursor protein activates microglia via JNK and p38-MAPK. Neurobiol Aging 26:9–16
Bozyczko-Coyne D, Saporito MS, Hudkins RL (2002) Targeting the JNK pathway for therapeutic benefit in CNS disease. Curr Drug Targets CNS Neurol Disord 1:31–49
Dai RP, Xu JM, Tao LJ, Li L, Li ZP, Zhang JY (2007) Induction of tumor necrosis-alpha, p38 and JNK in the spinal cord following acute heart injury in the rat model. Acta Anaesthesiol Scand 51:365–371
Davis RJ (2000) Signal transduction by the JNK group of MAP kinases. Cell 103:239–252
Dong Y, Liu HD, Zhao R et al (2009) Ischemia activates JNK/c-Jun/AP-1 pathway to up-regulate 14-3-3gamma in astrocyte. J Neurochem 109(Suppl 1):182–188
Dubreuil CI, Winton MJ, McKerracher L (2003) Rho activation patterns after spinal cord injury and the role of activated Rho in apoptosis in the central nervous system. J Cell Biol 162:233–243
Dumont RJ, Okonkwo DO, Verma S et al (2001) Acute spinal cord injury, part I: pathophysiologic mechanisms. Clin Neuropharmacol 24:254–264
Eilers A, Whitfield J, Babij C, Rubin LL, Ham J (1998) Role of the Jun kinase pathway in the regulation of c-Jun expression and apoptosis in sympathetic neurons. J Neurosci 18:1713–1724
Faden AI, Demediuk P, Panter SS, Vink R (1989) The role of excitatory amino acids and NMDA receptors in traumatic brain injury. Science 244:798–800
Gadea A, Schinelli S, Gallo V (2008) Endothelin-1 regulates astrocyte proliferation and reactive gliosis via a JNK/c-Jun signaling pathway. J Neurosci 28:2394–2408
Gao Y, Signore AP, Yin W et al (2005) Neuroprotection against focal ischemic brain injury by inhibition of c-Jun N-terminal kinase and attenuation of the mitochondrial apoptosis-signaling pathway. J Cereb Blood Flow Metab 25:694–712
Gruner JA (1992) A monitored contusion model of spinal cord injury in the rat. J Neurotrauma 9:123–128
Hall ED, Braughler JM (1993) Free radicals in CNS injury. Res Publ Assoc Res Nerv Ment Dis 71:81–105
Hara MR, Agrawal N, Kim SF et al (2005) S-nitrosylated GAPDH initiates apoptotic cell death by nuclear translocation following Siah1 binding. Nat Cell Biol 7:665–674
Herrmann JE, Imura T, Song B et al (2008) STAT3 is a critical regulator of astrogliosis and scar formation after spinal cord injury. J Neurosci 28:7231–7243
Ho FM, Liu SH, Liau CS, Huang PJ, Lin-Shiau SY (2000) High glucose-induced apoptosis in human endothelial cells is mediated by sequential activations of c-Jun NH(2)-terminal kinase and caspase-3. Circulation 101:2618–2624
Hu G, Fearon ER (1999) Siah-1 N-terminal RING domain is required for proteolysis function, and C-terminal sequences regulate oligomerization and binding to target proteins. Mol Cell Biol 19:724–732
Jacobs TP, Shohami E, Baze W et al (1987) Deteriorating stroke model: histopathology, edema, and eicosanoid changes following spinal cord ischemia in rabbits. Stroke 18:741–750
Katoh K, Ikata T, Katoh S et al (1996) Induction and its spread of apoptosis in rat spinal cord after mechanical trauma. Neurosci Lett 216:9–12
Kim DH, Vaccaro AR, Henderson FC, Benzel EC (2003) Molecular biology of cervical myelopathy and spinal cord injury: role of oligodendrocyte apoptosis. Spine J 3:510–519
Kim SY, Choi DW, Kim EA, Choi CY (2009) Stabilization of HIPK2 by escape from proteasomal degradation mediated by the E3 ubiquitin ligase Siah1. Cancer Lett 279:177–184
Lee JT, Wheeler TC, Li L, Chin LS (2008) Ubiquitination of alpha-synuclein by Siah-1 promotes alpha-synuclein aggregation and apoptotic cell death. Hum Mol Genet 17:906–917
Li QM, Tep C, Yune TY et al (2007) Opposite regulation of oligodendrocyte apoptosis by JNK3 and Pin1 after spinal cord injury. J Neurosci 27:8395–8404
Liu XZ, Xu XM, Hu R et al (1997) Neuronal and glial apoptosis after traumatic spinal cord injury. J Neurosci 17:5395–5406
Lu J, Ashwell KW, Waite P (2000) Advances in secondary spinal cord injury: role of apoptosis. Spine 25:1859–1866
Matsuo K, Satoh S, Okabe H et al (2003) SIAH1 inactivation correlates with tumor progression in hepatocellular carcinomas. Genes Chromosomes Cancer 36:283–291
McEwen ML, Springer JE (2005) A map** study of caspase-3 activation following acute spinal cord contusion in rats. J Histochem Cytochem 53:809–819
Merrill JE, Ignarro LJ, Sherman MP, Melinek J, Lane TE (1993) Microglial cell cytotoxicity of oligodendrocytes is mediated through nitric oxide. J Immunol 151:2132–2141
Nakahara S, Yone K, Sakou T et al (1999) Induction of apoptosis signal regulating kinase 1 (ASK1) after spinal cord injury in rats: possible involvement of ASK1-JNK and -p38 pathways in neuronal apoptosis. J Neuropathol Exp Neurol 58:442–450
Nishisho T, Tonai T, Tamura Y, Ikata T (1996) Experimental and clinical studies of eicosanoids in cerebrospinal fluid after spinal cord injury. Neurosurgery 39:950–956
Okabe H, Satoh S, Furukawa Y et al (2003) Involvement of PEG10 in human hepatocellular carcinogenesis through interaction with SIAH1. Cancer Res 63:3043–3048
Okuno S, Saito A, Hayashi T, Chan PH (2004) The c-Jun N-terminal protein kinase signaling pathway mediates Bax activation and subsequent neuronal apoptosis through interaction with Bim after transient focal cerebral ischemia. J Neurosci 24:7879–7887
Park E, Velumian AA, Fehlings MG (2004) The role of excitotoxicity in secondary mechanisms of spinal cord injury: a review with an emphasis on the implications for white matter degeneration. J Neurotrauma 21:754–774
Roux PP, Blenis J (2004) ERK and p38 MAPK-activated protein kinases: a family of protein kinases with diverse biological functions. Microbiol Mol Biol Rev 68:320–344
Santelli E, Leone M, Li C et al (2005) Structural analysis of Siah1-Siah-interacting protein interactions and insights into the assembly of an E3 ligase multiprotein complex. J Biol Chem 280:34278–34287
Tournier C, Hess P, Yang DD et al (2000) Requirement of JNK for stress-induced activation of the cytochrome c-mediated death pathway. Science 288:870–874
Venables JP, Dalgliesh C, Paronetto MP et al (2004) SIAH1 targets the alternative splicing factor T-STAR for degradation by the proteasome. Hum Mol Genet 13:1525–1534
Vlahopoulos S, Zoumpourlis VC (2004) JNK: a key modulator of intracellular signaling. Biochemistry 69:844–854
Wen YY, Yang ZQ, Song M, Li BL, Zhu JJ, Wang EH (2010a) SIAH1 induced apoptosis by activation of the JNK pathway and inhibited invasion by inactivation of the ERK pathway in breast cancer cells. Cancer Sci 101:73–79
Wen YY, Yang ZQ, Song M et al (2010b) The expression of SIAH1 is downregulated and associated with Bim and apoptosis in human breast cancer tissues and cells. Mol Carcinog 49:440–449
Whitfield J, Neame SJ, Paquet L, Bernard O, Ham J (2001) Dominant-negative c-Jun promotes neuronal survival by reducing BIM expression and inhibiting mitochondrial cytochrome c release. Neuron 29:629–643
Xu Z, Sproul A, Wang W, Kukekov N, Greene LA (2006) Siah1 interacts with the scaffold protein POSH to promote JNK activation and apoptosis. J Biol Chem 281:303–312
Yin KJ, Kim GM, Lee JM, He YY, Xu J, Hsu CY (2005) JNK activation contributes to DP5 induction and apoptosis following traumatic spinal cord injury. Neurobiol Dis 20:881–889
Yong C, Arnold PM, Zoubine MN et al (1998) Apoptosis in cellular compartments of rat spinal cord after severe contusion injury. J Neurotrauma 15:459–472
Yoshibayashi H, Okabe H, Satoh S et al (2007) SIAH1 causes growth arrest and apoptosis in hepatoma cells through beta-catenin degradation-dependent and -independent mechanisms. Oncol Rep 17:549–556
Zhao P, Waxman SG, Hains BC (2007) Extracellular signal-regulated kinase-regulated microglia-neuron signaling by prostaglandin E2 contributes to pain after spinal cord injury. J Neurosci 27:2357–2368
Zhou Y, Li L, Liu Q et al (2008) E3 ubiquitin ligase SIAH1 mediates ubiquitination and degradation of TRB3. Cell Signal 20:942–948
Acknowledgments
This work was supported by the National Natural Science Foundation of China (No.30770488, No.30870320, No.31070723, No. 81071487, and No.81070275); Natural Science Foundation of Jiangsu Province (No.BK2009156, No.BK2009157, and No.BK2009161; BK2009169); The 333 High-level personnel training project of Jiangsu Provience (No. BRA2010137) and Natural Science Foundation of Nantong University (07B21).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Donglin Wang and Qiuhui Lu contributed equally to this work.
Rights and permissions
About this article
Cite this article
Wang, D., Lu, Q., Shao, B. et al. An Upregulation of SIAH1 After Spinal Cord Injury in Adult Rats. J Mol Neurosci 45, 134–144 (2011). https://doi.org/10.1007/s12031-011-9501-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-011-9501-y