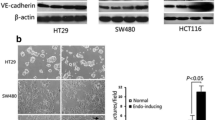
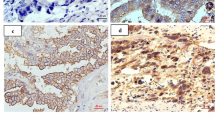
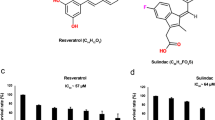
Abstract
Our previous studies have highlighted the pivotal role of gastric cancer mesenchymal stem cells (GCMSCs) in tumor initiation, progression, and metastasis. In parallel, it is well-documented that endothelial cells (ECs) undergo functional alterations in response to challenging tumor microenvironment. This study aims to elucidate whether functional changes in ECs might be induced by GCMSCs and thus influence cancer progression. Cell proliferation was assessed through CCK-8 and colony formation assays, while cell migration and invasion capabilities were evaluated by wound-healing and Transwell assays. Immunohistochemistry was employed to examine protein distribution and expression levels. Additionally, quantitative analysis of protein and mRNA expression was carried out through Western blotting and qRT-PCR respectively, with gene knockdown achieved using siRNA. Our findings revealed that GCMSCs effectively stimulate cell proliferation, migration, and angiogenesis of human umbilical vein endothelial cells (HUVECs), both in vitro and in vivo. GCMSCs promote the migration and invasion of gastric cancer cells by inducing the expression of Slit2 in HUVECs. Notably, the inhibition of phosphorylated AKT partially mitigates the aforementioned effects. In conclusion, GCMSCs may exert regulatory control over Slit2 expression in ECs via the AKT signaling pathway, thereby inducing functional changes in ECs that promote tumor progression.
Graphical Abstract







Similar content being viewed by others
Data Availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Code Availability
Not applicable.
References
Zheng, R., Zhang, S., Zeng, H., Wang, S., Sun, K., Chen, R., Li, L., Wei, W., & He, J. (2022). Cancer incidence and mortality in China, 2016. Journal of the National Cancer Center.
Quail, D., & Joyce, J. (2013). Microenvironmental regulation of tumor progression and metastasis. Nature Medicine, 19(11), 1423–1437. https://doi.org/10.1038/nm.3394.
Rankin, E., & Giaccia, A. (2016). Hypoxic control of metastasis. Science (New York N Y), 352(6282), 175–180. https://doi.org/10.1126/science.aaf4405.
Pitt, J., Marabelle, A., Eggermont, A., Soria, J., Kroemer, G., & Zitvogel, L. (2016). Targeting the tumor microenvironment: Removing obstruction to anticancer immune responses and immunotherapy. Annals of Oncology: Official Journal of the European Society for Medical Oncology, 27(8), 1482–1492. https://doi.org/10.1093/annonc/mdw168.
Yang, L., **, Y., Yu, X., Qian, F., Guo, Z., Qian, C., Cui, Y., & Bian, X. (2011). Gastric cancer stem-like cells possess higher capability of invasion and metastasis in association with a mesenchymal transition phenotype. Cancer Letters, 310(1), 46–52. https://doi.org/10.1016/j.canlet.2011.06.003.
Quante, M., Tu, S., Tomita, H., Gonda, T., Wang, S., Takashi, S., Baik, G., Shibata, W., Diprete, B., Betz, K., Friedman, R., Varro, A., Tycko, B., & Wang, T. (2011). Bone marrow-derived myofibroblasts contribute to the mesenchymal stem cell niche and promote tumor growth. Cancer Cell, 19(2), 257–272. https://doi.org/10.1016/j.ccr.2011.01.020.
Cao, H., Xu, W., Qian, H., Zhu, W., Yan, Y., Zhou, H., Zhang, X., Xu, X., Li, J., Chen, Z., & Xu, X. (2009). Mesenchymal stem cell-like cells derived from human gastric cancer tissues. Cancer Letters, 274(1), 61–71. https://doi.org/10.1016/j.canlet.2008.08.036.
Sun, L., Wang, Q., Chen, B., Zhao, Y., Shen, B., Wang, H., Xu, J., Zhu, M., Zhao, X., Xu, C., Chen, Z., Wang, M., Xu, W., & Zhu, W. (2018). Gastric cancer mesenchymal stem cells derived IL-8 induces PD-L1 expression in gastric cancer cells via STAT3/mTOR-c-Myc signal axis. Cell Death & Disease, 9(9), 928. https://doi.org/10.1038/s41419-018-0988-9.
Li, W., Zhang, X., Wu, F., Zhou, Y., Bao, Z., Li, H., Zheng, P., & Zhao, S. (2019). Dec 4). Gastric cancer-derived mesenchymal stromal cells trigger M2 macrophage polarization that promotes metastasis and EMT in gastric cancer. Cell Death and Disease, 10(12), 918. https://doi.org/10.1038/s41419-019-2131-y.
Shen, J., & Zhu, W. (2021). Research advances in the role of gastric cancer–derived mesenchymal stem cells in tumor progression (review). International Journal of Molecular Medicine, 47(2), 455–462. https://doi.org/10.3892/ijmm.2020.4810.
Wan, L., Pantel, K., & Kang, Y. (2013). Tumor metastasis: Moving new biological insights into the clinic. Nature Medicine, 19(11), 1450–1464. https://doi.org/10.1038/nm.3391.
Dammeijer, F., van Gulijk, M., Mulder, E., Lukkes, M., Klaase, L., van den Bosch, T., van Nimwegen, M., Lau, S., Latupeirissa, K., Schetters, S., van Kooyk, Y., Boon, L., Moyaart, A., Mueller, Y., Katsikis, P., Eggermont, A., Vroman, H., Stadhouders, R., Hendriks, R., Thüsen, J., Grünhagen, D., Verhoef, C., van Hall, T., & Aerts, J. (2020). The PD-1/PD-L1-Checkpoint restrains T cell immunity in Tumor-Draining Lymph Nodes. Cancer Cell, 38(5), 685–700e688. https://doi.org/10.1016/j.ccell.2020.09.001.
Pradeep, S., Kim, S., Wu, S., Nishimura, M., Chaluvally-Raghavan, P., Miyake, T., Pecot, C., Kim, S., Choi, H., Bischoff, F., Mayer, J., Huang, L., Nick, A., Hall, C., Rodriguez-Aguayo, C., Zand, B., Dalton, H., Arumugam, T., Lee, H., Han, H., Cho, M., Rupaimoole, R., Mangala, L., Sehgal, V., Oh, S., Liu, J., Lee, J., Coleman, R., Ram, P., Lopez-Berestein, G., Fidler, I., & Sood, A. (2014). Hematogenous metastasis of ovarian cancer: Rethinking mode of spread. Cancer Cell, 26(1), 77–91. https://doi.org/10.1016/j.ccr.2014.05.002.
Sturtzel, C. (2017). Endothelial cells. Advances in Experimental Medicine and Biology, 1003, 71–91. https://doi.org/10.1007/978-3-319-57613-8_4.
Gaikwad, A. V., Eapen, M. S., McAlinden, K. D., Chia, C., Larby, J., Myers, S., Dey, S., Haug, G., Markos, J., Glanville, A. R., & Sohal, S. S. (2020, Oct). Endothelial to mesenchymal transition (EndMT) and vascular remodeling in pulmonary hypertension and idiopathic pulmonary fibrosis. Expert Review of Respiratory Medicine, 14(10), 1027–1043. https://doi.org/10.1080/17476348.2020.1795832.
Huang, H., Chen, L., Sun, W., Du, H., Dong, S., Ahmed, A., Cao, D., Cui, J., Zhang, Y., & Cao, Q. (2021). Collagenase IV and clusterin-modified polycaprolactone-polyethylene glycol nanoparticles for penetrating dense tumor tissues. Theranostics, 11(2), 906–924. https://doi.org/10.7150/thno.47446.
Seoane, J., & Gomis, R. (2017). TGF-β Family Signaling in Tumor suppression and Cancer progression. Cold Spring Harbor Perspectives in Biology, 9(12). https://doi.org/10.1101/cshperspect.a022277.
Kaushik, N., Kim, S., Suh, Y., & Lee, S. (2019). Proinvasive extracellular matrix remodeling for tumor progression. Archives of Pharmacal Research, 42(1), 40–47. https://doi.org/10.1007/s12272-018-1097-0.
Shu, D. Y., Butcher, E., & Saint-Geniez, M. (2020). Jun 16). EMT and EndMT: Emerging roles in age-related Macular Degeneration. International Journal of Molecular Sciences, 21(12). https://doi.org/10.3390/ijms21124271.
Xu-Dubois, Y., Peltier, J., Brocheriou, I., Suberbielle-Boissel, C., Djamali, A., Reese, S., Mooney, N., Keuylian, Z., Lion, J., Ouali, N., Levy, P., Jouanneau, C., Rondeau, E., & Hertig, A. (2016). Markers of endothelial-to-mesenchymal transition: Evidence for antibody-endothelium Interaction during antibody-mediated rejection in kidney recipients. Journal of the American Society of Nephrology: JASN, 27(1), 324–332. https://doi.org/10.1681/asn.2014070679.
Zhang, L., He, J., Wang, J., Liu, J., Chen, Z., Deng, B., Wei, L., Wu, H., Liang, B., Li, H., Huang, Y., Lu, L., Yang, Z., **an, S., & Wang, L. (2021). Knockout RAGE alleviates cardiac fibrosis through repressing endothelial-to-mesenchymal transition (EndMT) mediated by autophagy. Cell Death & Disease, 12(5), 470. https://doi.org/10.1038/s41419-021-03750-4.
Xu, Z., Guo, C., Ye, Q., Shi, Y., Sun, Y., Zhang, J., Huang, J., Huang, Y., Zeng, C., Zhang, X., Ke, Y., & Cheng, H. (2021). Endothelial deletion of SHP2 suppresses tumor angiogenesis and promotes vascular normalization. Nature Communications, 12(1). https://doi.org/10.1038/s41467-021-26697-8.
Shoemaker, L. D., McCormick, A. K., Allen, B. M., & Chang, S. D. (2020). Jun). Evidence for endothelial-to-mesenchymal transition in human brain arteriovenous malformations. Clin Transl Med, 10(2), e99. https://doi.org/10.1002/ctm2.99.
Liang, G., Wang, S., Shao, J., **, Y., Xu, L., Yan, Y., Günther, S., Wang, L., & Offermanns, S. (2022). Tenascin-X mediates Flow-Induced suppression of EndMT and atherosclerosis. Circulation Research, 130(11), 1647–1659. https://doi.org/10.1161/circresaha.121.320694.
Wang, X., Chen, Z., Xu, J., Tang, S., An, N., Jiang, L., Zhang, Y., Zhang, S., Zhang, Q., Shen, Y., Chen, S., Lan, X., Wang, T., Zhai, L., Cao, S., Guo, S., Liu, Y., Bi, A., Chen, Y., Gai, X., Duan, Y., Zheng, Y., Fu, Y., Li, Y., Yuan, L., Tong, L., Mo, K., Wang, M., Lin, S., Tan, M., Luo, C., Chen, Y., Liu, J., Zhang, Q., Li, L., & Huang, M. (2022). SLC1A1-mediated cellular and mitochondrial influx of R-2-hydroxyglutarate in vascular endothelial cells promotes tumor angiogenesis in IDH1-mutant solid tumors. Cell Research, 32(7), 638–658. https://doi.org/10.1038/s41422-022-00650-w.
Cheng, H., Chen, Y., Wong, J., Weng, C., Chen, H., Yu, S., Chen, H., Yuan, A., & Chen, J. (2017). Cancer cells increase endothelial cell tube formation and survival by activating the PI3K/Akt signalling pathway. Journal of Experimental & Clinical cancer Research: CR, 36(1), 27. https://doi.org/10.1186/s13046-017-0495-3.
Romano, E., Manetti, M., Rosa, I., Fioretto, B., Ibba-Manneschi, L., Matucci-Cerinic, M., & Guiducci, S. (2018). Slit2/Robo4 axis may contribute to endothelial cell dysfunction and angiogenesis disturbance in systemic sclerosis. Annals of the Rheumatic Diseases, 77(11), 1665–1674. https://doi.org/10.1136/annrheumdis-2018-213239.
Lu, L., Li, X., Zhong, Z., Zhou, W., Zhou, D., Zhu, M., & Miao, C. (2021). KMT5A downregulation participated in high glucose-mediated EndMT via Upregulation of ENO1 expression in Diabetic Nephropathy. International Journal of Biological Sciences, 17(15), 4093–4107. https://doi.org/10.7150/ijbs.62867.
Huang, C., Chen, B., Wang, X., Xu, J., Sun, L., Wang, D., Zhao, Y., Zhou, C., Gao, Q., Wang, Q., Chen, Z., Wang, M., Zhang, X., Xu, W., Shen, B., & Zhu, W. (2023). Gastric cancer mesenchymal stem cells via the CXCR2/HK2/PD-L1 pathway mediate immunosuppression. Gastric cancer: Official Journal of the International Gastric Cancer Association and the Japanese Gastric Cancer Association, 26(5), 691–707. https://doi.org/10.1007/s10120-023-01405-1.
Han, F., Guo, S., Huang, C., Cui, L., Zhao, Y., Ma, J., Zhu, M., Chen, Z., Wang, M., Shen, B., & Zhu, W. (2021). Gastric cancer mesenchymal stem cells inhibit natural killer cell function by up-regulating FBP1. Central-European Journal of Immunology, 46(4), 427–437. https://doi.org/10.5114/ceji.2021.111753.
Shen, W., Song, Z., Zhong, X., Huang, M., Shen, D., Gao, P., Qian, X., Wang, M., He, X., Wang, T., Li, S., & Song, X. (2022). 2022/07/08). Sangerbox: A comprehensive, interaction-friendly clinical bioinformatics analysis platform [Commentary]. iMeta, 1(3), e36. https://doi.org/10.1002/imt2.36.
Bonnans, C., Chou, J., & Werb, Z. (2014). Remodelling the extracellular matrix in development and disease. Nature Reviews Molecular cell Biology, 15(12), 786–801. https://doi.org/10.1038/nrm3904.
Turajlic, S., & Swanton, C. (2016). Metastasis as an evolutionary process. Science (New York N Y), 352(6282), 169–175. https://doi.org/10.1126/science.aaf2784.
Anderson, N., & Simon, M. (2020). The tumor microenvironment. Current Biology: CB, 30(16), R921–R925. https://doi.org/10.1016/j.cub.2020.06.081.
Maishi, N., & Hida, K. (2017, Oct). Tumor endothelial cells accelerate tumor metastasis. Cancer Science, 108(10), 1921–1926. https://doi.org/10.1111/cas.13336.
Wieland, E., Rodriguez-Vita, J., Liebler, S. S., Mogler, C., Moll, I., Herberich, S. E., Espinet, E., Herpel, E., Menuchin, A., Chang-Claude, J., Hoffmeister, M., Gebhardt, C., Brenner, H., Trumpp, A., Siebel, C. W., Hecker, M., Utikal, J., Sprinzak, D., & Fischer, A. (2017). Endothelial Notch1 activity facilitates metastasis. Cancer Cell, Mar 13(3), 355–367. https://doi.org/10.1016/j.ccell.2017.01.007. 31.
Chen, Y., McAndrews, K., & Kalluri, R. (2021). Clinical and therapeutic relevance of cancer-associated fibroblasts. Nature Reviews Clinical Oncology, 18(12), 792–804. https://doi.org/10.1038/s41571-021-00546-5.
Hong, L., Du, X., Li, W., Mao, Y., Sun, L., & Li, X. (2018). Sep). EndMT: A promising and controversial field. European Journal of Cell Biology, 97(7), 493–500. https://doi.org/10.1016/j.ejcb.2018.07.005.
Ridge, S., Sullivan, F., & Glynn, S. (2017). Mesenchymal stem cells: Key players in cancer progression. Molecular cancer, 16(1), 31. https://doi.org/10.1186/s12943-017-0597-8.
Tavora, B., Mederer, T., Wessel, K. J., Ruffing, S., Sadjadi, M., Missmahl, M., Ostendorf, B. N., Liu, X., Kim, J. Y., Olsen, O., Welm, A. L., Goodarzi, H., & Tavazoie, S. F. (2020, Oct). Tumoural activation of TLR3-SLIT2 axis in endothelium drives metastasis. Nature, 586(7828), 299–304. https://doi.org/10.1038/s41586-020-2774-y.
Liu, J., Hou, W., Guan, T., Tang, L., Zhu, X., Li, Y., Hou, S., Zhang, J., Chen, H., & Huang, Y. (2018). May). Slit2/Robo1 signaling is involved in angiogenesis of glomerular endothelial cells exposed to a diabetic-like environment. Angiogenesis, 21(2), 237–249. https://doi.org/10.1007/s10456-017-9592-3.
Chen, Q., Zhou, X., Hou, R., Zhou, Z., Wang, Z., Chen, Y., Weng, J., & Xu, J. (2021). Sep). Aminophylline modulates the permeability of endothelial cells via the Slit2-Robo4 pathway in lipopolysaccharide-induced inflammation. Exp Ther Med, 22(3), 1042. https://doi.org/10.3892/etm.2021.10474.
Gohrig, A., Detjen, K. M., Hilfenhaus, G., Korner, J. L., Welzel, M., Arsenic, R., Schmuck, R., Bahra, M., Wu, J. Y., Wiedenmann, B., & Fischer, C. (2014, Mar 1). Axon guidance factor SLIT2 inhibits neural invasion and metastasis in pancreatic cancer. Cancer Res, 74(5), 1529–1540. https://doi.org/10.1158/0008-5472.CAN-13-1012.
Zegeye, M., Lindkvist, M., Fälker, K., Kumawat, A., Paramel, G., Grenegård, M., Sirsjö, A., & Ljungberg, L. (2018). Activation of the JAK/STAT3 and PI3K/AKT pathways are crucial for IL-6 trans-signaling-mediated pro-inflammatory response in human vascular endothelial cells. Cell Communication and Signaling: CCS, 16(1), 55. https://doi.org/10.1186/s12964-018-0268-4.
Acknowledgements
The authors would like to further acknowledge the financial support from the National Natural Science Foundation of China, Medical Research project of Jiangsu Provincial Health Commission and Jiangsu National Natural Science Foundation. The authors would also like to express our gratitude for the drawing materials provided by BioRender.
Funding
This work was supported by the National Natural Science Foundation of China (Grant numbers 81972313 and 82203547); Medical Research project of Jiangsu Provincial Health Commission (Grant number X373); and Jiangsu National Natural Science Foundation (Grant number BK20210136).
Author information
Authors and Affiliations
Contributions
LC: Generated experimental data, Manuscript writing, experimental design; TL: Generated experimental data, experimental design; CH: Generated experimental data, experimental design; FY: Experimental data generation, experimental design; LL: Experimental data generation, experimental design; LS: Experimental data generation, samples collection; YZ: Experimental data generation, samples collection; DW: Experimental design, manuscript editing; MW: Experimental design, manuscript revision; YJ: Experimental design, manuscript revision; WZ: Experimental design, manuscript revision.
Corresponding author
Ethics declarations
Ethical Approval
All studies were conducted in accordance with the Ethics Committee of Jiangsu University and informed consent was obtained from all subjects.
Consent to Participate
Informed consent was obtained from all individual participants included in the study.
Consent for Publication
The authors affirm that all participants provided informed consent for publication of the paper.
Conflict of Interest
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Cui, L., Liu, T., Huang, C. et al. Gastric Cancer Mesenchymal Stem Cells Trigger Endothelial Cell Functional Changes to Promote Cancer Progression. Stem Cell Rev and Rep 20, 1285–1298 (2024). https://doi.org/10.1007/s12015-024-10720-8
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12015-024-10720-8