Abstract
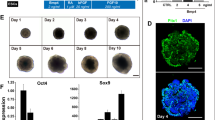
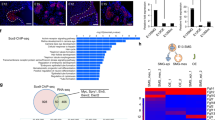
Derivation of salivary gland epithelial progenitors (SGEPs) from human pluripotent stem cells (hPSCs) has great potential in developmental biology and regenerative medicine. At present, no efficient method is available to generate salivary gland cells from hPSCs. Here, we described for the first time a robust protocol for direct differentiation of hPSCs into SGEPs by mimicking retinoic acid and Wnt signaling. These hPSC-derived SGEPs expressed SOX9, KRT5, and KRT19, important progenitor markers of develo** salivary glands. CD24 and α-SMA positive cells, capable of restoring the functions of injured salivary glands, were also present in SGEP cultures. Importantly, RNA-sequencing revealed that the SGEPs resembled the transcript profiles of human fetal submandibular glands. Therefore, we provided an efficient protocol to induce hPSCs differentiation into SGEPs. Our study provides a foundation for generating functional hPSCs derived salivary gland acinar cells and three-dimensional organoids, potentially serving as new models for basic study and future translational research.
Graphical Abstract







Similar content being viewed by others
Data Availability
The data used in this article will be available from the authors on reasonable request.
Code Availability
The RNA-seq data files in this paper have been deposited into Gene Expression Omnibus (GEO). The accession number is GEO: GSE198097.
References
Zhang, Y., Yang, Y., Jiang, M., Huang, S. X., Zhang, W., Al, A. D., Danopoulos, S., Mori, M., Chen, Y. W., Balasubramanian, R., de Sousa, C., Lopes, S. M., Serra, C., Bialecka, M., Kim, E., Lin, S., Toste de Carvalho, A. L. R., Riccio, P. N., Cardoso, W. V., et al. (2018). 3D modeling of esophageal development using human PSC-derived basal progenitors reveals a critical role for notch signaling. Cell Stem Cell, 23(4), 516–29 e5.
Lungova, V., Chen, X., Wang, Z., Kendziorski, C., Thibeault, S., & L. (2019). Human induced pluripotent stem cell-derived vocal fold mucosa mimics development and responses to smoke exposure. Nature Communications, 10(1), 4161.
Huang, S. X. L., Islam, M. N., O’Neill, J., Hu, Z., Yang, Y.-G., Chen, Y.-W., Mumau, M., Green, M. D., Vunjak-Novakovic, G., Bhattacharya, J., & Snoeck, H.-W. (2014). Efficient generation of lung and airway epithelial cells from human pluripotent stem cells. Nature Biotechnology, 32(1), 84–91.
Matsumoto, R., & Takahashi, Y. (2021). Human pituitary development and application of iPSCs for pituitary disease. Cellular and Molecular Life Sciences, 78(5), 2069–2079.
Joy, D. A., Libby, A. R. G., & McDevitt, T. C. (2021). Deep neural net tracking of human pluripotent stem cells reveals intrinsic behaviors directing morphogenesis. Stem Cell Reports, 16(5), 1317–1330.
Lee, J., Rabbani, C. C., Gao, H., Steinhart, M. R., Woodruff, B. M., Pflum, Z. E., Kim, A., Heller, S., Liu, Y., Shipchandler, T. Z., Koehler, K., & R. (2020). Hair-bearing human skin generated entirely from pluripotent stem cells. Nature, 582(7812), 399–404.
Miho, O., & Takashi, T. (2017). Functional Salivary Gland Regeneration by Organ Replacement Therapy. Salivary Gland Development and Regeneration: Advances in Research and Clinical Approaches to Functional Restoration.(p. 193-203). Springer International Publishing.
Mattingly, A., Finley, J. K., Knox, S., & M. (2015). Salivary gland development and disease. Wiley Interdisciplinary Reviews: Developmental Biology, 4(6), 573–590.
Pedersen, A. M., Bardow, A., Jensen, S., & Beier, N. B. (2002). Saliva and gastrointestinal functions of taste, mastication, swallowing and digestion. Oral Diseases, 8(3), 117–129.
Mattingly, A., Finley, J. K., Knox, S., & M. (2015). Salivary gland development and disease. Wiley Interdisciplinary Reviews Developmental Biology, 4(6), 573–590.
Patel, V. N., Hoffman, M., & P. (2014). Salivary gland development: A template for regeneration. Seminars in Cell & Developmental Biology, 25-26, 52–60.
Athwal, H. K., Murphy 3rd, G., Tibbs, E., Cornett, A., Hill, E., Yeoh, K., Berenstein, E., Hoffman, M. P., & Lombaert, I. M. A. (2019). Sox10 regulates plasticity of epithelial progenitors toward secretory units of exocrine glands. Stem Cell Reports, 12(2), 366–380.
Chatzeli, L., Gaete, M., Tucker, A., & S. (2017). Fgf10 and Sox9 are essential for the establishment of distal progenitor cells during mouse salivary gland development. Development, 144(12), 2294–2305.
Tanaka, J., Ogawa, M., Hojo, H., Kawashima, Y., Mabuchi, Y., Hata, K., Nakamura, S., Yasuhara, R., Takamatsu, K., Irie, T., Fukada, T., Sakai, T., Inoue, T., Nishimura, R., Ohara, O., Saito, I., Ohba, S., Tsuji, T., & Mishima, K. (2018). Generation of orthotopically functional salivary gland from embryonic stem cells. Nature Communications, 9(1), 4216.
Wright, D. M., Buenger, D. E., Abashev, T. M., Lindeman, R. P., Ding, J., Sandell, L., & L. (2015). Retinoic acid regulates embryonic development of mammalian submandibular salivary glands. Developmental Biology, 407(1), 57–67.
Metzler, M. A., Raja, S., Elliott, K. H., Friedl, R. M., Tran, N. Q. H., Brugmann, S. A., Larsen, M., Sandell, L., & L. (2018). RDH10-mediated retinol metabolism and RARalpha-mediated retinoic acid signaling are required for submandibular salivary gland initiation. Development, 145(15), dev164822.
Matsumoto, S., Kurimoto, T., Taketo, M. M., Fujii, S., & Kikuchi, A. (2016). The WNT/MYB pathway suppresses KIT expression to control the timing of salivary proacinar differentiation and duct formation. Development, 143(13), 2311–2324.
Miho, O., & Takashi, T. (2017). Functional Salivary Gland Regeneration. Organ Regeneration: 3D Stem Cell Culture & Manipulation. (p. 135-51). Springer New York.
Li, Q., Zhang, S., Sui, Y., Fu, X., Li, Y., & Wei, S. (2019). Sequential stimulation with different concentrations of BMP4 promotes the differentiation of human embryonic stem cells into dental epithelium with potential for tooth formation. Stem Cell Research & Therapy, 10(1), 276.
Hauser, B. R., Aure, M. H., Kelly, M. C., Genomics, Computational Biology Core, Hoffman, M. P., & Chibly, A. M. (2020). Generation of a single-cell RNAseq atlas of murine salivary gland development. iScience, 23(12), 101838.
Knox, S. M., Lombaert, I. M. A., Reed, X., Vitale-Cross, L., Gutkind, J. S., Hoffman, M., & P. (2010). Parasympathetic innervation maintains epithelial progenitor cells during salivary organogenesis. Science, 329(5999), 1645–1647.
National Institute of Dental and Craniofacial Research (2011). [online] Available at: https://sgmap.nidcr.nih.gov/sgmap/sgexp.html. [cited 12 Nov. 2021].
Nedvetsky, P. I., Emmerson, E., Finley, J. K., Ettinger, A., Cruz-Pacheco, N., Prochazka, J., Haddox, C. L., Northrup, E., Hodges, C., Mostov, K. E., Hoffman, M. P., Knox, S., & M. (2014). Parasympathetic innervation regulates tubulogenesis in the develo** salivary gland. Developmental Cell, 30(4), 449–462.
Knox, S. M., Lombaert, I. M. A., Reed, X., Vitale-Cross, L., Gutkind, J. S., Hoffman, M., & P. (2010). Parasympathetic innervation maintains epithelial progenitor cells during salivary organogenesis. Science (New York, N.Y.), 329(5999), 1645–1647.
Nanduri, L. S., Baanstra, M., Faber, H., Rocchi, C., Zwart, E., de Haan, G., van Os, R., & Coppes, R. P. (2014). Purification and ex vivo expansion of fully functional salivary gland stem cells. Stem Cell Reports, 3(6), 957–964.
Ninche, N., Kwak, M., & Ghazizadeh, S. (2020). Diverse epithelial cell populations contribute to the regeneration of secretory units in injured salivary glands. Development, 147(19), dev192807.
Saitou, M., Gaylord, E. A., Xu, E., May, A. J., Neznanova, L., Nathan, S., Grawe, A., Chang, J., Ryan, W., Ruhl, S., Knox, S. M., & Gokcumen, O. (2020). Functional specialization of human salivary glands and origins of proteins intrinsic to human saliva. Cell Reports, 33(7), 108402.
Gluck, C., Min, S., Oyelakin, A., Smalley, K., Sinha, S., Romano, R., & A. (2016). RNA-seq based transcriptomic map reveals new insights into mouse salivary gland development and maturation. BMC Genomics, 17(1), 923.
Acknowledgements
We thank the professor. Duanqin Pei of the Guangzhou Institutes of Biomedicine and Health kindly provides the hiPS cell lines. We also thank the professor. Yuanyuan Zhang of the Wake Forest Institute for Regenerative Medicine for the guidance of this work. Moreover, the authors thank National Center for Protein Sciences at Peking University in Bei**g, China, for assistance with providing Roche qPCR equipment and Nikon A1R confocal microscopy photography.
Funding
This work was supported by Discipline Development Fund of School of Stomatology, Peking University.
Author information
Authors and Affiliations
Contributions
Conceptualization: S.Z., J.X. and S.W.; Methodology: S.Z., S.Y. and C.D.; Investigation: S.Z., Y.Z. and Y.F.; Formal Analysis: Y.Z. and S.Y.; Verification: S.Z., Y.S, and S.W.; Writing – Original Draft: S.Z., Y.S.; Writing – Review & Editing: Y.S., Y.Z., J.X., and S.W.; Resources: S.Z., Y.F.; Supervision: S.Z., S.W., and J.X.; Project Administration: S.Z., S.W.; Funding Acquisition: J.X., S.W.
Corresponding author
Ethics declarations
Ethics Approval
This work involved isolation of embryonic mouse SMGs and adult human SMGs. All procedures performed in this study were reviewed and approved by the Biomedical Ethics Committee of Peking University in China. The permit number for the animal part is LA2021052. The permit number for the human part is 19093.
Consent to Participate
The human salivary gland tissues were collected after obtaining informed consent from the donors.
Consent for Publication
Not applicable.
Conflict of Interest
No competing interests declared.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 5038 kb)
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, S., Sui, Y., Zhang, Y. et al. Derivation of Human Salivary Epithelial Progenitors from Pluripotent Stem Cells via Activation of RA and Wnt Signaling. Stem Cell Rev and Rep 19, 430–442 (2023). https://doi.org/10.1007/s12015-022-10431-y
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12015-022-10431-y