Abstract
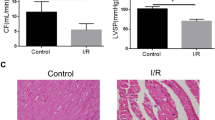
Ischemia-reperfusion (IR) injury commonly arises during cardiac surgery involving Cardiopulmonary Bypass (CPB), and it has relationship with ferroptosis in mice. However, the exact role of ferroptosis in the human cardiac damage caused by cardiac surgery remains unclear. Basic patient data and perioperative period information were collected, and clinic indicators related to cardiac function were detected to assess the extent of cardiac injury. Cardiac tissue samples were collected to determine histopathological changes, ultrastructure of mitochondrial and hallmarks of ferroptosis. 25 patients were involved in this study. In the present study, we observed a significant increase in the clinical indicator hs-cTnT, with levels rising more than 1393 ± 242 folds (P < 0.0001) following the cardiac surgery. Masson staining revealed a notable increase in fibrosis levels by 2.282 ± 0.259% (P = 0.0009). Furthermore, there was a significant elevation in lipid peroxidation, as evidenced by a 61.42 ± 17.33% increase in MDA (P = 0.0006). Additionally, we observed notable swelling, decreased mitochondrial crista, and even fragmented mitochondria. Notably, changes in the marker gene of ferroptosis were observed, with PTGS2 showing a 6.437 ± 0.81 folds increase (P < 0.0001). Furthermore, key regulators such as SLC7A11 and GPX4 proteins exhibited a reduction of 97.33 ± 25.78% (P = 0.0068) and 60.59 ± 14.93% (P = 0.0071), respectively, indicating the occurrence of ferroptosis following the surgery. Ferroptosis exists in myocardial IR injury caused by cardiac surgery with CPB, indicating that targeting ferroptosis could serve as a potential strategy for myocardial protection against CPB-induced IR injury. The trial has been registered in Chinese Clinical Trial Registry (ChiCTR, No. ChiCTR2200061995) on July 16th, 2022.





Similar content being viewed by others
References
Chen, M., Li, X., & Mu, G. (2022). Myocardial protective and anti-inflammatory effects of dexmedetomidine in patients undergoing cardiovascular surgery with cardiopulmonary bypass: a systematic review and meta-analysis. Journal of Anesthesia, 36, 5–16.
Amer, G. F., Elawady, M. S., ElDerie, A., & Sanad, M. (2020). Normothermia versus Hypothermia during Cardiopulmonary Bypass in Cases of Repair of Atrioventricular Septal Defect. Anesthesia Essays and Researches, 14, 112–118.
Cheng, C., Xu, J. M., & Yu, T. (2017). Neutralizing IL-6 reduces heart injury by decreasing nerve growth factor precursor in the heart and hypothalamus during rat cardiopulmonary bypass. Life Science, 178, 61–69.
Virmani, R., Forman, M. B., & Kolodgie, F. D. (1990). Myocardial reperfusion injury. Histopathological effects of perfluorochemical. Circulation, 81, Iv57–68.
Dabkowski, E. R., Williamson, C. L., & Hollander, J. M. (2008). Mitochondria-specific transgenic overexpression of phospholipid hydroperoxide glutathione peroxidase (GPx4) attenuates ischemia/reperfusion-associated cardiac dysfunction. Free Radical Biology and Medicine, 45, 855–865.
Fang, X., et al. (2019). Ferroptosis as a target for protection against cardiomyopathy. Proceedings of the National Academy of Sciences of the United States of America, 116, 2672–2680.
Dixon, S. J., et al. (2012). Ferroptosis: an iron-dependent form of nonapoptotic cell death. Cell, 149, 1060–1072.
Yang, W. S., & Stockwell, B. R. (2016). Ferroptosis: Death by Lipid Peroxidation. Trends in Cell Biology, 26, 165–176.
Ta, N., et al. (2022). Mitochondrial outer membrane protein FUNDC2 promotes ferroptosis and contributes to doxorubicin-induced cardiomyopathy. Proceedings of the National Academy of Sciences of the United States of America, 119, e2117396119.
Chen, X., Li, J., Kang, R., Klionsky, D. J., & Tang, D. (2021). Ferroptosis: machinery and regulation. Autophagy, 17, 2054–2081.
Park, M. W., et al. (2021). NOX4 promotes ferroptosis of astrocytes by oxidative stress-induced lipid peroxidation via the impairment of mitochondrial metabolism in Alzheimer’s diseases. Redox Biology, 41, 101947.
Cung, T. T., et al. (2015). Cyclosporine before PCI in Patients with Acute Myocardial Infarction. The New England Journal of Medicine, 373, 1021–1031.
Nowicki, R., et al. (2010). The estimation of oxidative stress markers and apoptosis in right atrium auricles cardiomyocytes of patients undergoing surgical heart revascularisation with the use of warm blood cardioplegia. Folia Histochemica et Cytobiologica, 48, 202–207.
Wang, K., et al. (2022). Emerging roles of ferroptosis in cardiovascular diseases. Cell Death Discovery, 8, 394.
Huang, Z., et al. (2011). Synergy of isoflurane preconditioning and propofol postconditioning reduces myocardial reperfusion injury in patients. Clinical Science, 121, 57–69.
Yu, Y., et al. (2021). Ferroptosis: a cell death connecting oxidative stress, inflammation and cardiovascular diseases. Cell Death Discovery, 7, 193.
Liu, Y., et al. (2023). The diversified role of mitochondria in ferroptosis in cancer. Cell Death and Disease, 14, 519.
Chen, X., Kang, R., Kroemer, G., & Tang, D. (2021). Organelle-specific regulation of ferroptosis. Cell death and Differentiation, 28, 2843–2856.
Yang, W. S., et al. (2014). Regulation of ferroptotic cancer cell death by GPX4. Cell, 156, 317–331.
Tian, H., et al. (2021). Activation of NRF2/FPN1 pathway attenuates myocardial ischemia-reperfusion injury in diabetic rats by regulating iron homeostasis and ferroptosis. Cell Stress and Chaperones, 27, 149–164.
Doll, S., et al. (2019). FSP1 is a glutathione-independent ferroptosis suppressor. Nature, 575, 693–698.
Ye, Y., et al. (2022). Repression of the antiporter SLC7A11/glutathione/glutathione peroxidase 4 axis drives ferroptosis of vascular smooth muscle cells to facilitate vascular calcification. Kidney International, 102, 1259–1275.
Funding
This work was supported by the National Natural Science Foundation of China (Grant No. 81903314); the Henan Provincial Science and Technology Research Project (Grant No. 232102310515); the Key Research Projects of Colleges and universities in Henan province (Grant NO. 24A330006).
Author information
Authors and Affiliations
Contributions
S.Z. contributed to the study conception and design. Material preparation and clinic data collection were performed by X.Z. The detection of the tissues and data analysis were performed by J.L. The first draft of the manuscript was written by J.L. and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Consent to Participate
Informed consent was obtained from all individual participants included in the study.
Ethics Approval
This trial is approved by the First Affiliated Hospital of Zhengzhou University’s Ethics Committee (No. 2020-KY-0086) and has been successfully registered in the Chinese Clinical Trial Registry (ChiCTR, URL https://www.chictr.org.cn) with the registration number ChiCTR2200061995 on July 16th, 2022. This study adheres to the Helsinki Declaration.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, S., Li, J., Wang, J. et al. Ferroptosis Exists in Ischemia Reperfusion Injury after Cardiac Surgery with Cardiopulmonary Bypass. Cell Biochem Biophys (2024). https://doi.org/10.1007/s12013-024-01228-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12013-024-01228-6