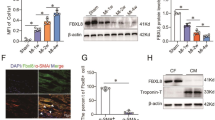
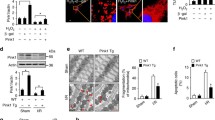
Abstract
The ubiquitin proteasome system is a highly specific and selective protein regulatory system that plays an essential role in the regulation of the cell cycle. Despite its significance, the role of ubiquitination in cardiomyocyte proliferation remains largely unclear. This study aimed to investigate the potential impact of E3 ubiquitin ligase ASB14 (Ankyrin Repeat And SOCS Box Containing 14) on cardiac regeneration. We conducted a microarray analysis of apical resection ventricle tissues, and our findings revealed that ASB14 was down-regulated during the cardiac regenerative response. Subsequently, we examined the effect of ASB14 silencing on cardiomyocyte nuclear proliferation both in vitro and in vivo. Our results indicated that ASB14 silencing promoted cardiomyocyte nuclear proliferation, suggesting that ASB14 may play a role in regulating cardiac regeneration. To further investigate the potential therapeutic implications of ASB14 deficiency, we examined the cardiac function of mice with ASB14 deficiency in response to ischemic injury. Our findings showed that mice with ASB14 deficiency exhibited preserved cardiac function and a therapeutic effect in response to ischemic injury, which was attributed to the enhancement of cardiomyocyte nuclear proliferation. To elucidate the underlying mechanisms, we investigated the effect of ASB14 on microtubule-associated protein RP/EB family member 2 (MAPRE2) protein degradation. Our results indicated that the loss of ASB14 decreased the degradation of MAPRE2 protein, subsequently promoting cardiomyocyte nuclear proliferation and enhancing cardiac repair after myocardial infarction (MI). In conclusion, our study provides evidence that inhibition of ASB14-mediated MAPRE2 ubiquitination promotes cardiomyocyte nuclear proliferation, which may serve as a potential target for treating heart failure induced by MI injury.





Similar content being viewed by others
Data availability
The microarray data was downloaded from Gene Expression Omnibus (accession number, GSE154071). No datasets were generated or analysed during the current study.
References
Virani, S. S., Alonso, A., Benjamin, E. J., Bittencourt, M. S., Callaway, C. W., Carson, A. P., Chamberlain, A. M., Chang, A. R., Cheng, S., Delling, F. N., Djousse, L., Elkind, M. S. V., Ferguson, J. F., Fornage, M., Khan, S. S., Kissela, B. M., Knutson, K. L., Kwan, T. W., Lackland, D. T., Lewis, T. T., Lichtman, J. H., Longenecker, C. T., Loop, M. S., Lutsey, P. L., Martin, S. S., Matsushita, K., Moran, A. E., Mussolino, M. E., Perak, A. M., Rosamond, W. D., Roth, G. A., Sampson, U. K. A., Satou, G. M., Schroeder, E. B., Shah, S. H., Shay, C. M., Spartano, N. L., Stokes, A., Tirschwell, D. L., VanWagner, L. B. & & Tsao, C. W. (2020). Heart disease and stroke statistics update: a report from the American heart association.Circulation, 141, e139–e596. https://doi.org/10.1161/cir.0000000000000757.
Ongstad, E. L., & Gourdie, R. G. (2016). Can heart function lost to disease be regenerated by therapeutic targeting of cardiac scar tissue? Seminar in Cellular and Developmental Biology, 58, 41–54. https://doi.org/10.1016/j.semcdb.2016.05.020.
Senyo, S. E., Steinhauser, M. L., Pizzimenti, C. L., Yang, V. K., Cai, L., Wang, M., Wu, T. D., Guerquin-Kern, J. L., Lechene, C. P., & Lee, R. T. (2013). Mammalian heart renewal by pre-existing cardiomyocytes. Nature, 493, 433–436. https://doi.org/10.1038/nature11682.
Porrello, E. R., Mahmoud, A. I., Simpson, E., Hill, J. A., Richardson, J. A., Olson, E. N., & Sadek, H. A. (2011). Transient regenerative potential of the neonatal mouse heart. Science, 331, 1078–1080. https://doi.org/10.1126/science.1200708.
Yu, W., Huang, X., Tian, X., Zhang, H., He, L., Wang, Y., Nie, Y., Hu, S., Lin, Z., Zhou, B., Pu, W., Lui, K. O., & Zhou, B. (2016). GATA4 regulates Fgf16 to promote heart repair after injury. Development, 143, 936–949. https://doi.org/10.1242/dev.130971.
Fu, W., Ren, H., Shou, J., Liao, Q., Li, L., Shi, Y., Jose, P. A., Zeng, C., & Wang, W. E. (2022). Loss of NPPA-AS1 promotes heart regeneration by stabilizing SFPQ-NONO heteromer-induced DNA repair. Basic Research in Cardiology, 117, 10 https://doi.org/10.1007/s00395-022-00921-y.
Fan, Y., Cheng, Y., Li, Y., Chen, B., Wang, Z., Wei, T., Zhang, H., Guo, Y., Wang, Q., Wei, Y., Chen, F., Sha, J., Guo, X., & Wang, L. (2020). Phosphoproteomic analysis of neonatal regenerative myocardium revealed important roles of checkpoint Kinase 1 via activating mammalian target of rapamycin C1/Ribosomal protein S6 Kinase b-1 pathway. Circulation, 141, 1554–1569. https://doi.org/10.1161/circulationaha.119.040747.
Willis, M. S., Townley-Tilson, W. H., Kang, E. Y., Homeister, J. W., & Patterson, C. (2010). Sent to destroy: the ubiquitin proteasome system regulates cell signaling and protein quality control in cardiovascular development and disease. Circulation Research, 106, 463–478. https://doi.org/10.1161/circresaha.109.208801.
Wang, C., Yang, H., & Gao, C. (2019). Potential biomarkers for heart failure. Journal in Cellular Physiology, 234, 9467–9474. https://doi.org/10.1002/jcp.27632.
Li, L. T., Jiang, G., Chen, Q., & Zheng, J. N. (2015). Ki67 is a promising molecular target in the diagnosis of cancer (review). Molecular Medicine Reports, 11, 1566–1572. https://doi.org/10.3892/mmr.2014.2914.
Auchampach, J., Han, L., Huang, G. N., Kühn, B., Lough, J. W., O’Meara, C. C., Payumo, A. Y., Rosenthal, N. A., Sucov, H. M., Yutzey, K. E., & Patterson, M. (2022). Measuring cardiomyocyte cell-cycle activity and proliferation in the age of heart regeneration. American Journal in Physiological Heart Circulation Physiology, 322, H579–h596. https://doi.org/10.1152/ajpheart.00666.2021.
Wang, X., Li, Y., He, M., Kong, X., Jiang, P., Liu, X., Diao, L., Zhang, X., Li, H., Ling, X., **a, S., Liu, Z., Liu, Y., Cui, C. P., Wang, Y., Tang, L., Zhang, L., He, F., & Li, D. (2022). UbiBrowser 2.0: a comprehensive resource for proteome-wide known and predicted ubiquitin ligase/deubiquitinase-substrate interactions in eukaryotic species. Nucleic Acids Research, 50, D719–d728. https://doi.org/10.1093/nar/gkab962.
Iimori, M., Watanabe, S., Kiyonari, S., Matsuoka, K., Sakasai, R., Saeki, H., Oki, E., Kitao, H., & Maehara, Y. (2016). Phosphorylation of EB2 by Aurora B and CDK1 ensures mitotic progression and genome stability. Nature Communications, 7, 11117 https://doi.org/10.1038/ncomms11117.
Zhu, W., Zhang, E., Zhao, M., Chong, Z., Fan, C., Tang, Y., Hunter, J. D., Borovjagin, A. V., Walcott, G. P., Chen, J. Y., Qin, G., & Zhang, J. (2018). Regenerative potential of neonatal porcine hearts. Circulation, 138, 2809–2816. https://doi.org/10.1161/circulationaha.118.034886.
Sun, X., Han, Q., Luo, H., Pan, X., Ji, Y., Yang, Y., Chen, H., Wang, F., Lai, W., Guan, X., Zhang, Q., Tang, Y., Chu, J., Yu, J., Shou, W., Deng, Y., & Li, X. (2017). Profiling analysis of long non-coding RNAs in early postnatal mouse hearts. Scientific Reports, 7, 43485 https://doi.org/10.1038/srep43485.
Alkass, K., Panula, J., Westman, M., Wu, T. D., Guerquin-Kern, J. L., & Bergmann, O. (2015). No evidence for cardiomyocyte number expansion in preadolescent mice. Cell, 163, 1026–1036. https://doi.org/10.1016/j.cell.2015.10.035.
Hosseini, S. M., Okoye, I., Chaleshtari, M. G., Hazhirkarzar, B., Mohamadnejad, J., Azizi, G., Hojjat-Farsangi, M., Mohammadi, H., Shotorbani, S. S., & Jadidi-Niaragh, F. (2019). E2 ubiquitin-conjugating enzymes in cancer: implications for immunotherapeutic interventions. Clinica Chimica Acta, 498, 126–134. https://doi.org/10.1016/j.cca.2019.08.020.
Tamamori-Adachi, M., Hayashida, K., Nobori, K., Omizu, C., Yamada, K., Sakamoto, N., Kamura, T., Fukuda, K., Ogawa, S., Nakayama, K. I., & Kitajima, S. (2004). Down-regulation of p27Kip1 promotes cell proliferation of rat neonatal cardiomyocytes induced by nuclear expression of cyclin D1 and CDK4. evidence for impaired Skp2-dependent degradation of p27 in terminal differentiation. The Journal of Biological Chemistry, 279, 50429–50436. https://doi.org/10.1074/jbc.M403084200.
Li, B., Li, M., Li, X., Li, H., Lai, Y., Huang, S., He, X., Si, X., Zheng, H., Liao, W., Liao, Y., & Bin, J. (2019). Sirt1-inducible deacetylation of p21 promotes cardiomyocyte proliferation. Aging (Albany NY), 11, 12546–12567. https://doi.org/10.18632/aging.102587.
Huang, S., Li, X., Zheng, H., Si, X., Li, B., Wei, G., Li, C., Chen, Y., Chen, Y., Liao, W., Liao, Y., & Bin, J. (2019). Loss of super-enhancer-regulated circRNA Nfix induces cardiac regeneration after myocardial infarction in adult mice. Circulation, 139, 2857–2876. https://doi.org/10.1161/circulationaha.118.038361.
Zhong, F. J., Li, Y. M., Xu, C., Sun, B., Wang, J. L., & Yang, L. Y. (2021). EB2 promotes hepatocellular carcinoma proliferation and metastasis via MAPK/ERK pathway by modulating microtubule dynamics. Clinical Science (Lond), 135, 847–864. https://doi.org/10.1042/cs20201500.
Funding
The Medical Project of Scientific and Technological Breakthroughs in Henan Province (No. SBGJ202303049).
Author information
Authors and Affiliations
Contributions
Y.Y. performed most of the experiments. D.M. performed most of the analyses, with support from B.L. and X.S. Y.Y, F.L and W.F. performed the experiments. Q.C. supervised the study. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethical approval
Animal care and experimental procedures in the present study were approved by the Institutional Animal Care and Use Committee of Zhengzhou University (ethical approval number: 202353) and followed the National Institutes of Health Guidelines for the Care and Use of Laboratory Animals.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yang, Y., Ma, D., Liu, B. et al. E3 Ubiquitin Ligase ASB14 Inhibits Cardiomyocyte Proliferation by Regulating MAPRE2 Ubiquitination. Cell Biochem Biophys (2024). https://doi.org/10.1007/s12013-024-01223-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12013-024-01223-x