Abstract
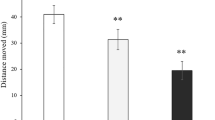
Increased application of the pyrethroid insecticide deltamethrin has adverse effects on the cardiac system and neurobehavior on the non-target organisms, which has raised the public’s attention. Because of spermidine and melatonin considered to have cardioprotective and neuroprotective characteristics, zebrafish were utilized as the model organism to explore the protective effects of spermidine and melatonin against deltamethrin-induced toxicity. We tested the neurobehavior of zebrafish larvae through a rest/wake behavior assay, and evaluated the levels of the expression of Scn5lab, gata4, nkx2.5, hcrt, hcrtr, and aanat2 by qRT-PCR. Besides that cmlc2 was evaluated by whole-mount in situ hybridization. Results have shown that compared with control group, 0.025 mg/L deltamethrin could significantly disturb the cardiac development, downregulating the expression of Scn5lab and transcriptional factors gata4 and nkx2.5, disturbing cardiac loo**, resulting in defects in cardiac morphology and function. Moreover, deltamethrin could alter the expression levels of rest/wake genes and cause hyperactivity in zebrafish larvae. Besides, compared with deltamethrin group, the exogenous 0.01 mg/L spermidine and 0.232 mg/L melatonin could significantly rescue the adverse effects of deltamethrin on the cardiac system and neurobehavior in zebrafish. This indicated that spermidine and melatonin have neuroprotective and cardioprotective effects against deltamethrin-induced adverse effects in zebrafish.





Similar content being viewed by others
References
Elliott, M., Farnham, A. W., Janes, N. F., Needham, P. H., & Pulman, D. A. (1974). Synthetic insecticide with a new order of activity. Nature, 248, 710–711.
Soderlund, D. M. (2012). Molecular mechanisms of pyrethroid insecticide neurotoxicity: Recent advances. Archives of Toxicology, 86, 165–181.
Bouwman, H., Sereda, B., & Meinhardt, H. M. (2006). Simultaneous presence of DDT and pyrethroid residues in human breast milk from a malaria endemic area in South Africa. Environmental Pollution, 144, 902–917.
Whyatt, R. M., Garfinkel, R., Hoepner, L. A., Holmes, D., Borjas, M., Williams, M. K., et al. (2007). Within- and between-home variability in indoor-air insecticide levels during pregnancy among an inner-city cohort from New York City. Environmental Health Perspectives, 115, 383–389.
Naeher, L. P., Tulve, N. S., Egeghy, P. P., Barr, D. B., Adetona, O., Fortmann, R. C., et al. (2010). Organophosphorus and pyrethroid insecticide urinary metabolite concentrations in young children living in a southeastern United States city. Science of the Total Environment, 408, 1145–1153.
Richardson, J. R., Taylor, M. M., Shalat, S. L., Guillot, T. S., Caudle, W. M., Hossain, M. M., et al. (2015). Developmental pesticide exposure reproduces features of attention deficit hyperactivity disorder. The FASEB Journal, 29, 1960–1972.
Dhivya Vadhana, M. S., Siva Arumugam, S., Carloni, M., Nasuti, C., & Gabbianelli, R. (2013). Early life permethrin treatment leads to long-term cardiotoxicity. Chemosphere, 93, 1029–1034.
Luo, H., Masika, J., Guan, X., Nie, L., Ao, D., Qi, Y., et al. (2019). Long term perinatal deltamethrin exposure alters electrophysiological properties of embryonic ventricular cardiomyocyte. Current Medical Science, 39, 21–27.
Widmark, J., Sundstrom, G., Ocampo Daza, D., & Larhammar, D. (2011). Differential evolution of voltage-gated sodium channels in tetrapods and teleost fishes. Molecular Biology and Evolution, 28, 859–871.
Moreno, J. D., & Clancy, C. E. (2012). Pathophysiology of the cardiac late Na current and its potential as a drug target. Journal of Molecular and Cellular Cardiology, 52, 608–619.
Mahboob, S., Niazi, F., AlGhanim, K., Sultana, S., Al-Misned, F., & Ahmed, Z. (2015). Health risks associated with pesticide residues in water, sediments and the muscle tissues of Catla catla at Head Balloki on the River Ravi. Environmental Monitoring and Assessment, 187, 81.
Kylin, H. B. A. H. (2014). Malaria control insecticide residues in breast milk: The need to consider infant health risks. Environmental Health Perspectives, 117(10), 1477–1480.
Li, M., Liu, X., & Feng, X. (2019). Cardiovascular toxicity and anxiety-like behavior induced by deltamethrin in zebrafish (Danio rerio) larvae. Chemosphere, 219, 155–164.
Raina, A., & Jänne, J. (1975). Physiology of the natural polyamines putrescine, spermidine and spermine. Medicine and Biology, 53, 121–147.
Eisenberg, T., Knauer, H., Schauer, A., Büttner, S., Ruckenstuhl, C., Carmona-Gutierrez, D., et al. (2009). Induction of autophagy by spermidine promotes longevity. Nature Cell Biology, 11, 1305–1314.
Eisenberg, T., Abdellatif, M., Schroeder, S., Primessnig, U., Stekovic, S., Pendl, T., et al. (2016). Cardioprotection and lifespan extension by the natural polyamine spermidine. Nature Medicine, 22, 1428–1438.
Sharma, S., Kumar, P., & Deshmukh, R. (2018). Neuroprotective potential of spermidine against rotenone induced Parkinson's disease in rats. Neurochemistry International, 116, 104–111.
Karbownik, M., & Reiter, R. J. (2000). Antioxidative effects of melatonin in protection against cellular damage caused by ionizing radiation. Proceedings of the Society for Experimental Biology and Medicine, 225, 9–22.
Iggena, D., Winter, Y., & Steiner, B. (2017). Melatonin restores hippocampal neural precursor cell proliferation and prevents cognitive deficits induced by jet lag simulation in adult mice. Journal of Pineal Research, 62, e12397.
Calvo, J. R., Gonzalez-Yanes, C., & Maldonado, M. D. (2013). The role of melatonin in the cells of the innate immunity: A review. Journal of Pineal Research, 55, 103–120.
Su, S.-C., Hsieh, M.-J., Yang, W.-E., Chung, W.-H., Reiter, R. J., & Yang, S.-F. (2017). Cancer metastasis: Mechanisms of inhibition by melatonin. Journal of Pineal Research, 62, e12370.
Pandi-Perumal, S. R., BaHammam, A. S., Ojike, N. I., Akinseye, O. A., Kendzerska, T., Buttoo, K., et al. (2017). Melatonin and human cardiovascular disease. Journal of Cardiovascular Pharmacology and Therapeutics, 22, 122–132.
Brown, D. R., Samsa, L. A., Qian, L., & Liu, J. (2016). Advances in the study of heart development and disease using zebrafish. Journal of Cardiovascular Development and Disease, 3(2), 13.
Renier, C., Faraco, J. H., Bourgin, P., Motley, T., Bonaventure, P., Rosa, F., et al. (2007). Genomic and functional conservation of sedative-hypnotic targets in the zebrafish. Pharmacogenetics and Genomics, 17, 237–253.
Zhu, J.-J., Xu, Y.-Q., He, J.-H., Yu, H.-P., Huang, C.-J., Gao, J.-M., et al. (2014). Human cardiotoxic drugs delivered by soaking and microinjection induce cardiovascular toxicity in zebrafish: Zebrafish model for assessing drug-induced cardiovascular toxicity. Journal of Applied Toxicology, 34, 139–148.
Haverinen, J., & Vornanen, M. (2016). Deltamethrin is toxic to the fish (crucian carp, Carassius carassius) heart. Pesticide Biochemistry and Physiology, 129, 36–42.
Zhang, S., Xu, J., Kuang, X., Li, S., Li, X., Chen, D., et al. (2017). Biological impacts of glyphosate on morphology, embryo biomechanics and larval behavior in zebrafish (Danio rerio). Chemosphere, 181, 270–280.
Haverinen, J., Hassinen, M., Korajoki, H., & Vornanen, M. (2018). Cardiac voltage-gated sodium channel expression and electrophysiological characterization of the sodium current in the zebrafish (Danio rerio) ventricle. Progress in Biophysics and Molecular Biology, 138, 59–68.
Hoage, T., Ding, Y., & Xu, X. (2012). Quantifying cardiac functions in embryonic and adult zebrafish. In X. Peng & M. Antonyak (Eds.), Cardiovascular development: Methods and protocols, methods in molecular biology (pp. 11–20). Totowa, NJ: Humana Press.
Poss, K. D., Keating, M. T., & Nechiporuk, A. (2003). Tales of regeneration in zebrafish. Developmental Dynamics, 226, 202–210.
Chopra, S. S., Stroud, D. M., Watanabe, H., Bennett, J. S., Burns, C. G., Wells, K. S., et al. (2010). Voltage-gated sodium channels are required for heart development in zebrafish. Circulation Research, 106, 1342–1350.
Chueh, T. C., Hsu, L. S., Kao, C. M., Hsu, T. W., Liao, H. Y., Wang, K. Y., et al. (2017). Transcriptome analysis of zebrafish embryos exposed to deltamethrin. Environmental Toxicology, 32, 1548–1557.
Gorge, G., & Nagel, R. (1990). Toxicity of lindane, atrazine, and deltamethrin to early life stages of zebrafish (Brachydanio rerio). Ecotoxicology and Environmental Safety, 20, 246–255.
Novak, A. E., Taylor, A. D., Pineda, R. H., Lasda, E. L., Wright, M. A., & Ribera, A. B. (2006). Embryonic and larval expression of zebrafish voltage-gated sodium channel α-subunit genes. Developmental Dynamics, 235, 1962–1973.
Papadatos, G. A., Wallerstein, P. M. R., Head, C. E. G., Ratcliff, R., Brady, P. A., Benndorf, K., et al. (2002). Slowed conduction and ventricular tachycardia after targeted disruption of the cardiac sodium channel gene Scn5a. Proceedings of the National academy of Sciences of the United States of America, 99, 6210–6215.
Balci, M. M., & Akdemir, R. (2011). NKX2.5 mutations and congenital heart disease: Is it a marker of cardiac anomalies? International Journal of Cardiology, 147, e44–45.
Zhou, P., He, A., & Pu, W. T. (2012). Regulation of GATA4 transcriptional activity in cardiovascular development and disease. Current Topics in Developmental Biology, 100, 143–169.
Yeung, H.-M., Hung, M.-W., Lau, C.-F., & Fung, M.-L. (2015). Cardioprotective effects of melatonin against myocardial injuries induced by chronic intermittent hypoxia in rats. Journal of Pineal Research, 58, 12–25.
Zheng, J., Yu, Y., Feng, W., Li, J., Liu, J., Zhang, C., et al. (2019). Influence of nanomolar deltamethrin on the hallmarks of primary cultured cortical neuronal network and the role of ryanodine receptors. Environmental Health Perspectives, 127, 67003.
Zhdanova, I. V., Wang, S. Y., Leclair, O. U., & Danilova, N. P. (2001). Melatonin promotes sleep-like state in zebrafish. Brain Research, 903, 263–268.
Appelbaum, L., Wang, G. X., Maro, G. S., Mori, R., Tovin, A., Marin, W., et al. (2009). Sleep–wake regulation and hypocretin–melatonin interaction in zebrafish. Proceedings of the National Academy of Sciences of the United States of America, 106, 21942–21947.
Appelbaum, L., Vallone, D., Anzulovich, A., Ziv, L., Tom, M., Foulkes, N. S., et al. (2006). Zebrafish arylalkylamine-N-acetyltransferase genes—Targets for regulation of the circadian clock. Journal of Molecular Endocrinology, 36, 337–347.
Acknowledgements
X. Z. F and X. Y. L. conceived and designed experiments. Q. G. performed in situ hybridization and qRT-PCR. Z. Y. F. and X. L. carried out the surface tension test. X. Y. L. performed all other experiments. X. L. and Q. G., performed data analysis. X. L. drafted the manuscript, Y. Q. T. made important contributions to the discussion, and all author commented on the manuscript.
Funding
This project was initiated in the State Key Laboratory of Medicinal Chemical Biology at Nankai University. This work was supported by the Special Fund for Basic Research on Scientific Instruments of the Chinese National Natural Science Foundation [Grant No: 61633012], and the National Basic Research Program (973 program) of China [Grant No: 2015CB856500].
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
No competing interests declared.
Additional information
Handling Editor: Vittorio Fineschi .
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Liu, X., Gao, Q., Feng, Z. et al. Protective Effects of Spermidine and Melatonin on Deltamethrin-Induced Cardiotoxicity and Neurotoxicity in Zebrafish. Cardiovasc Toxicol 21, 29–41 (2021). https://doi.org/10.1007/s12012-020-09591-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12012-020-09591-5