Abstract
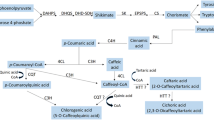
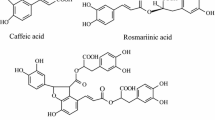
Gibberellic acid (GA3) is reported to have diverse effects on hairy root cultures of many plant species; therefore, the effects of GA3 on the growth, secondary metabolite production (caffeic acid derivatives and lignin), phenylalanine ammonia lyase (PAL) activity, and free radical scavenging activity of light-grown Echinacea purpurea L. hairy roots were investigated. Eight concentrations of GA3, ranging from 0.005 to 1.0 μM, were added to shake flask cultures. The moderate GA3 concentration, 0.025 μM, resulted in the highest concentrations of cichoric acid, caftaric acid, and chlorogenic acid, as well as increased PAL activity, cell viability, and free radical scavenging activity, while higher and lower GA3 concentrations resulted in reduced levels compared to the control (lacking GA3). The moderate GA3 concentration also affected root morphogenesis; supplementation with 0.025 μM GA3 resulted in the development of thick, dense, purple-colored roots, while roots exposed to the higher and lower concentrations of GA3 were thin and off-white. This study demonstrates that supplementation with GA3 may be an excellent strategy to optimize the production of secondary metabolites from E. purpurea hairy root cultures; however, the GA3 concentration is a critical factor.







Similar content being viewed by others
References
Hudson, J. B. (2010). Journal Medicine Plants Research, 4, 2746–2752.
Liu, C. Z., Abbasi, B. H., Min, G., Murch, S. J., & Saxena, P. K. (2006). Journal Agriculturae Food Chemistry, 54, 8456–8460.
Abbasi, B. H., Liu, R., Saxena, P. K., & Liu, C. Z. (2009). Journal Chemistry Technical Biotechnology, 84, 1697–1701.
Lin, Z., Neamati, N., Zhao, H., Kiryu, Y., Turpin, J. A., Aberham, C., et al. (1999). Journal of Medicinal Chemistry, 42, 1401–1414.
Jones, M. P. A., Saxena, P. K., & Murch, S. J. (2009). Engineering Life Science, 9, 205–210.
Abbasi, B. H., Tian, C. L., Murch, S. J., Saxena, P. K., & Liu, C. Z. (2007). Plant Cell Reports, 26, 1367–1372.
Biondi, S., Lenzi, C., Baraldi, R., & Bagni, N. (1997). Journal Plant Growth Regional, 16, 159–167.
Bais, H. P., George, S. J., & Ravishankar, G. A. (2001). In Vitro Development Biology: Plant, 37, 293–299.
Sharaf-Eldin, M. A., Schnitzler, W. H., Nitz, G., Razin, A. M., & El-Oksh, I. I. (2007). Science Horticultural, 111, 326–329.
Smith, T. C., Weathers, P. J., & Cheetham, R. D. (1997). In Vitro Development Biology: Plant, 33, 75–79.
Murashige, T., & Skoog, F. (1962). Physiologia Plantarum, 15, 473–497.
Koukol, J., & Conn, E. E. (1961). Journal of Biological Chemistry, 236, 2692–2698.
Steponkus, P. L., & Lanphear, F. O. (1967). Plant Physiology, 42, 1423–1426.
Harborne, J. B. (1958). Biochemistry Journal, 70, 22–28.
Goering, H. K. and Soest, P. J. (1970). In: Agricultural handbook No. 379, Forage fiber analyses: apparatus, reagents, procedures and some applications (pp. 20–36). Washington: USDA
Rieger, M., & Litvin, P. (1999). Journal of Experimental Botany, 50, 201–209.
Amarowicz, R., Pegg, R. B., Rahimi-Moghaddam, P., Barl, B., & Weil, J. A. (2004). Food Chemistry, 84, 551–562.
Ritchie, S., McCubbin, A., Ambrose, G., Kao, T.-H., & Gilroy, S. (1999). Plant Physiology, 120, 361–370.
Ohkawa, H., Kamada, H., Suodo, H., & Harada, H. (1989). Journal of Plant Physiology, 134, 633–636.
Tuna, A. L., Kaya, C., Dikilitas, M., & Higgs, D. (2007). Environmental and Experimental Botany, 62, 1–9.
Teszlak, P., Gaal, K., & Nikfardjam, M. S. P. (2005). Analytica Chimica Acta, 543, 275–281.
Baluska, R., Parker, J. S., & Barlow, P. A. (1993). Planta, 191, 149–157.
Khan, M. I. (1980). Biologia Plantarum, 22, 401–403.
Ilan, A., & Dougall, D. K. (1994). Journal Plant Growth Regulation, 13, 213–219.
Shirley, B. W. (2001). Plant Physiology, 126, 485–493.
Neves, G. Y. S., Marchiosi, R., Ferrarese, M. L. L., Siqueira, S., & Ferrarese-Filho, O. (2010). Journal of Agronomy and Crop Science, 196, 467–473.
Singh, K., Kumar, S., Rani, A., Gulati, A., & Ahuja, P. S. (2009). Functional Integration Genomics, 9, 125–134.
Ohlsson, A. B., & Berglund, T. (2001). Plant Cell, Tissue and Organ Culture, 64, 77–80.
Gao, J., Zhang, S., Cai, F., Zheng, X., Lin, N., Qin, Y., et al. (2012). Molecular Biology Reports, 39, 3443–3452.
Li, X., Li, S., & Lin, J. X. (2003). Plant Science, 164, 549–556.
Boo, H. O., Chon, S. U., & Lee, S. Y. (2006). Journal Horticulture Science Technical, 81, 478–482.
Pellati, F., Benvenuti, S., Magro, L., Melegari, M., & Soragni, F. (2004). Journal of Pharmaceutical and Biomedical Analysis, 35, 289–301.
Tsai, Y. L., Chiou, S. Y., Chan, K. C., Sung, J. M., & Lin, S. D. (2012). LWT-Food Science Technical, 46, 169–176.
Taveira, M., Pereira, D. M., Sousa, C., Ferreres, F., Andrade, P. B., Martins, A., et al. (2009). Journal Agriculture Food Chemistry, 57, 1247–1252.
Acknowledgments
This work was funded by the National Natural Science Foundation of China (no. 21150110459), the Knowledge Innovation Program of the Chinese Academy of Sciences (nos. YZ-06-03 & Y227051304), the Chinese Academy of Sciences Fellowship for Young International Scientists (no. 2011Y1GA01), the Chinese Academy of Sciences Visiting Professorship for Senior International Scientists (no. 2011T1G05), and the Gosling Research Institute for Plant Preservation, Canada. Abbasi BH acknowledges financial support of Higher Education Commission of Pakistan for providing financial assistance for PhD.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Abbasi, B.H., Stiles, A.R., Saxena, P.K. et al. Gibberellic Acid Increases Secondary Metabolite Production in Echinacea purpurea Hairy Roots. Appl Biochem Biotechnol 168, 2057–2066 (2012). https://doi.org/10.1007/s12010-012-9917-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-012-9917-z