Abstract
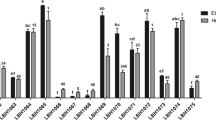
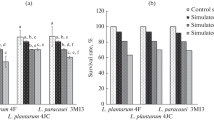
The aim of the present work was to assess the probiotic potential of lactic acid bacteria (LAB) isolated from huauzontle (Chenopodium berlandieri spp. Nuttalliae) inflorescences; additionally, multifunctional bioactivities of the intracellular content and cell wall fractions of the selected strains were also determined. Eight strains identified as Lactiplantibacillus plantarum, one as Lactobacillus pentosus and one as Pediococcus pentosaceus, were pre-selected according to their survival rate to acid (range 78.67 to 97.11%) and bile salt (range 49.62 to 55.23%) exposure, and their potential probiotic properties were evaluated. Overall, all strains were able to survive to several technological conditions and production of exopolysaccharides and β-galactosidase activity, auto-aggregation (> 40%), hydrophobicity (31.27–53.56%), coaggregation (41–63%), and mucosa adhesion capacity (> 70%). Besides, no biogenic amine production, mucin degradation, and γ-hemolysis activity was observed. Strains with the best probiotic properties were selected and showed to be resistant to four different antibiotics, but conjugation frequency was not found for filter mating. A survival higher than 75% was registered after bacterial exposure to simulated gastrointestinal conditions. Furthermore, whole cells, intracellular contents, and cell wall fractions of these selected strains exhibited antioxidant and anti-inflammatory activities and promoted splenocyte cell proliferation. Our results indicated that epiphytic bacteria from huauzontle not only met basic criteria for probiotics, but also represent a source of bioactive metabolites with potential therapeutic or preventive activity against stress- and inflammatory-mediated disorders; however additional experiments are still required.




Similar content being viewed by others
Data Availability
The dataset supporting the conclusions of this study and the information about the materials used are included within the article.
References
Aguilar-Toalá, J. E., Estrada-Montoya, M. C., Liceaga, A. M., Garcia, H. S., González-Aguilar, G. A., Vallejo-Cordoba, B., González-Córdova, A. F., & Hernández-Mendoza, A. (2019). An insight on antioxidant properties of the intracellular content of Lactobacillus casei CRL-431. LWT- Food Science and Technology, 102, 58–63. https://doi.org/10.1016/j.lwt.2018.12.015
Aguilar-Toalá, J. E., Santiago-López, L., Peres, C. M., Peres, C., Garcia, H. S., Vallejo-Cordoba, B., González-Córdova, A. F., & Hernández-Mendoza, A. (2017). Assessment of multifunctional activity of bioactive peptides derived from fermented milk by specific Lactobacillus plantarum strains. Journal of Dairy Science, 100(1), 65–75. https://doi.org/10.3168/jds.2016-11846
Alkalbani, N. S., Turner, M. S., & Ayyash, M. M. (2019). Isolation, identification, and potential probiotic characterization of isolated lactic acid bacteria and in vitro investigation of the cytotoxicity, antioxidant, and antidiabetic activities in fermented sausage. Microbial Cell Factories, 18(188). https://doi.org/10.1186/s12934-019-1239-1
Ayyanna, R., Ankaiah, D., & Arul, V. (2018). Anti-inflammatory and antioxidant properties of probiotic bacterium Lactobacillus mucosae AN1 and Lactobacillus fermentum SNR1 in Wistar albino rats. Frontiers in Microbiology, 9, 3063. https://doi.org/10.3389/fmicb.2018.03063
Bampi, G. B., Backes, G. T., Cansian, R. L., de Matos, F. E., Ansolin, I. M. A., Poleto, B. C., & Core & Favaro-Trindade, C. S. (2016). Spray chilling microencapsulation of Lactobacillus acidophilus and Bifidobacterium animalis subsp. lactis and its use in the preparation of savory probiotic cereal bars. Food and Bioprocess Technology, 9, 1422–1428. https://doi.org/10.1007/s11947-016-1724-z
Barros, C. P., Guimarães, J. T., Esmerino, E. A., Duarte, M. C. K., Silva, M. C., Silva, R., & Cruz, A. G. (2020). Paraprobiotics and postbiotics: Concepts and potential applications in dairy products. Current Opinion in Food Science, 32, 1–8. https://doi.org/10.1016/j.cofs.2019.12.003
Binda, S., Hill, C., Johansen, E., Obis, D., Pot, B., Sanders, M. E., Tremblay, A., & Ouwehand, A. C. (2020). Criteria to qualify microorganisms as “probiotic” in foods and dietary supplements. Frontiers in Microbiology, 11, 1662. https://doi.org/10.3389/fmicb.2020.01662
Bosnea, L. A., Moschakis, T., & Biliaderis, C. G. (2014). Complex coacervation as a novel microencapsulation technique to improve viability of probiotics under different stresses. Food and Bioprocess Technology, 7, 2767–2781. https://doi.org/10.1007/s11947-014-1317-7
Bover-Cid, S., & Holzapfel, W. H. (1999). Improved screening procedure for biogenic amine production by lactic acid bacteria. International Journal of Food Microbiology, 53, 33–41. https://doi.org/10.1016/S0168-1605(99)00152-X
Chang, M., & Chang, H. C. (2012). Development of a screening method for biogenic amine producing Bacillus spp. International Journal of Food Microbiology, 153, 269–274. https://doi.org/10.1016/j.ijfoodmicro.2011.11.008
Collado, M. C., Meriluoto, J., & Salminen, S. (2008). Adhesion and aggregation properties of probiotic and pathogen strains. European Food Research and Technology, 226(5), 1065–1073. https://doi.org/10.1007/s00217-007-0632-x
Cristofori, F., Dargenio, V. N., Dargenio, C., Miniello, V. L., Barone, M., & Francavilla, R. (2021). Anti-inflammatory and immunomodulatory effects of probiotics in gut inflammation: A door to the body. Frontiers in Immunology, 12, 178. https://doi.org/10.3389/fimmu.2021.578386
Cuevas-González, P. F., Aguilar-Toalá, J., & E., García, H. S., González-Córdova, A.F., Vallejo-Cordoba, B., & Hernández-Mendoza, A. (2020). Protective effect of the intracellular content from potential probiotic bacteria against oxidative damage induced by acrylamide in human erythrocytes. Probiotics and Antimicrobial Proteins, 12, 1459–1470. https://doi.org/10.1007/s12602-020-09636-9
Doleyres, Y., Schaub, L., & Lacroix, C. (2005). Comparison of the functionality of exopolysaccharides produced in situ or added as bioingredients on yogurt properties. Journal of Dairy Science, 88, 4146–4156. https://doi.org/10.3168/jds.S0022-0302(05)73100-3
Dridi, C., Millette, M., Aguilar, B., Salmieri, S., & Lacroix, M. (2022). Storage stability of a fermented probiotic beverage enriched with cricket protein hydrolysates. Food and Bioprocess Technology, 15, 2587–2600. https://doi.org/10.1007/s11947-022-02900-6
EFSA Panel on Additives and Products or Substances used in Animal Feed (FEEDAP). (2012). Guidance on the assessment of bacterial susceptibility to antimicrobials of human and veterinary importance. EFSA Journal, 10(6), 2740. https://doi.org/10.2903/j.efsa.2012.2740
Fontana, L., Bermudez-Brito, M., Plaza-Diaz, J., Muñoz-Quezada, S., & Gil, A. (2013). Sources, isolation, characterisation and evaluation of probiotics. British Journal of Nutrition, 109(Supl. 2), S35–S50. https://doi.org/10.1017/S0007114512004011
Fonteles, T. V., Costa, M. G. M., de Jesus, A. L. T., Fontes, C. P. M. L., Fernandes, F. A. N., & Rodrigues, S. (2013). Stability and quality parameters of probiotic cantaloupe melon juice produced with sonicated juice. Food and Bioprocess Technology, 6, 2860–2869. https://doi.org/10.1007/s11947-012-0962-y
Garcia, H. S., Santiago-López, L., González-Córdova, A. F., Vallejo-Cordoba, B., & Hernández-Mendoza, A. (2022). Evaluation of a pseudocereal suitability to prepare a functional fermented beverage with epiphytic lactic acid bacteria of Huauzontle (Chenopodium berlandieri spp. nuttalliae). LWT-Food Science and Technology, 155, 112913. https://doi.org/10.1016/j.lwt.2021.112913
Goa, T., Beyene, G., Mekonnen, M., & Gorems, K. (2022). Isolation and characterization of lactic acid bacteria from fermented milk produced in Jimma Town, Southwest Ethiopia, and evaluation of their antimicrobial activity against selected pathogenic bacteria. International Journal of Food Science, 2022, 2076021. https://doi.org/10.1155/2022/2076021
Guan, C., Chen, X., Jiang, X., Zhao, R., Yuan, Y., Chen, D., Zhang, C. C., Lu, M., Lu, Z., & Gu, R. (2020). In vitro studies of adhesion properties of six lactic acid bacteria isolated from the longevous population of China. RSC Advances, 10(41), 24234–24240. https://doi.org/10.1039/D0RA03517C
Guerrero-Encinas, I., González-González, J. N., Santiago-López, L., Muhlia-Almazán, A., Garcia, H. S., Mazorra-Manzano, M. A., Vallejo-Cordoba, B., González-Córdova, A. F., & Hernández-Mendoza, A. (2021). Protective effect of Lacticaseibacillus casei CRL 431 postbiotics on mitochondrial function and oxidative status in rats with aflatoxin b1–induced oxidative stress. Probiotics and Antimicrobial Proteins, 13(4), 1033–1043. https://doi.org/10.1007/s12602-021-09747-x
Gunathilake, K. D. P. P., Ranaweera, K. K. D. S., & Rupasinghe, H. V. (2018). In vitro anti-inflammatory properties of selected green leafy vegetables. Biomedicines, 6(4), 107. https://doi.org/10.3390/biomedicines6040107
Guo, H., Pan, L., Li, L., Lu, J., Kwok, L., Menghe, B., Zhang, H., & Zhang, W. (2017). Characterization of antibiotic resistance genes from Lactobacillus isolated from traditional dairy products. Journal of Food Science, 82, 724–730. https://doi.org/10.1111/1750-3841.13645
Han, S., Lu, Y., **e, J., Fei, Y., Zheng, G., Wang, Z., Liu, J., Lv, L., Ling, Z., Berglund, B., Yao, M., & Li, L. (2021). Probiotic gastrointestinal transit and colonization after oral administration: A long journey. Frontiers in Cellular Infection Microbiology, 11, 102. https://doi.org/10.3389/fcimb.2021.609722
He, F., Ouwehand, A. C., Isolauri, E., Hashimoto, H., Benno, Y., & Salminen, S. (2001). Comparison of mucosal adhesion and species identification of bifidobacteria isolated from healthy and allergic infants. FEMS Immunology and Medical Microbiology, 30, 43–47. https://doi.org/10.1111/j.1574-695X.2001.tb01548.x
Hernández-Delgado, N. C., Torres-Maravilla, E., Mayorga-Reyes, L., Martín, R., Langella, P., Pérez-Pastén-Borja, S.-P., & M.E., & Bermúdez-Humarán, L.G. (2021). Antioxidant and anti-inflammatory properties of probiotic candidate strains isolated during fermentation of agave (Agave angustifolia Haw). Microorganisms, 9(5), 1063. https://doi.org/10.3390/microorganisms9051063
Hernández-Granados, M. J., & Franco-Robles, E. (2020). Postbiotics in human health: Possible new functional ingredients? Food Research International, 137, 109660. https://doi.org/10.1016/j.foodres.2020.109660
Hevia, A., Delgado, S., Sanchez, B., & Margolles, A. (2015). Molecular players involved in the interaction between beneficial bacteria and the immune system. Frontiers in Microbiology, 6, 1285. https://doi.org/10.3389/fmicb.2015.01285
Hill, C., Guarner, F., Reid, G., Gibson, G. R., Merenstein, D. J., Pot, B., Morelli, L., Canani, R. B., Flint, H. J., Salminen, S., Calder, P. C., & Sanders, M. E. (2014). The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nature Reviews Gastroenterology & Hepatology, 11, 506–514. https://doi.org/10.1038/nrgastro.2014.66
Jonsson, H., Ström, E., & Roos, S. (2001). Addition of mucin to the growth medium triggers mucus-binding activity in different strains of Lactobacillus reuteri in vitro. FEMS Microbiology Letters, 204(1), 19–22. https://doi.org/10.1111/j.1574-6968.2001.tb10855.x
Kiyohara, M., Tanigawa, K., Chaiwangsri, T., Katayama, T., Ashida, H., & Yamamoto, K. (2011). An exo-alpha-sialidase from bifidobacteria involved in the degradation of sialyloligosaccharides in human milk and intestinal glycoconjugates. Glycobiology, 21, 437–447. https://doi.org/10.1093/glycob/cwq175
Kowalska-Krochmal, B., & Dudek-Wicher, R. (2021). The minimum inhibitory concentration of antibiotics: Methods, interpretation, clinical relevance. Pathogens, 10(2), 165. https://doi.org/10.3390/pathogens10020165
Laparra, J. M., & Sanz, Y. (2009). Comparison of in vitro models to study bacterial adhesion to the intestinal epithelium. Letters in Applied Microbiology, 49(6), 695–701. https://doi.org/10.1111/j.1472-765X.2009.02729.x
Lemme-Dumit, J. M., Polti, M. A., Perdigon, G., & Galdeano, C. M. (2018). Probiotic bacteria cell walls stimulate the activity of the intestinal epithelial cells and macrophage functionality. Beneficial Microbes, 9(1), 153–164. https://doi.org/10.3920/BM2016.0220
Li, X. J., Yue, L. Y., Guan, X. F., & Qiao, S. Y. (2008). The adhesion of putative probiotic lactobacilli to cultured epithelial cells and porcine intestinal mucus. Journal of Applied Microbiology, 104, 1082–1091. https://doi.org/10.1111/j.1365-2672.2007.03636.x
Lončarić, M., Strelec, I., Moslavac, T., Šubarić, D., Pavić, V., & Molnar, M. (2021). Lipoxygenase inhibition by plant extracts. Biomolecules, 11(2), 152. https://doi.org/10.3390/biom11020152
López-Monterrubio, D. I., Lobato-Calleros, C., Alvarez-Ramirez, J., & Vernon-Carter, E. J. (2020). Huauzontle (Chenopodium nuttalliae Saff.) protein: Composition, structure, physicochemical and functional properties. Food Hydrocolloids, 108, 106043. https://doi.org/10.1016/j.foodhyd.2020.106043
Ma, J. J., Mao, X. Y., Wang, Q., Yang, S., Zhang, D., Chen, S. W., & Li, Y. H. (2014). Effect of spray drying and freeze drying on the immunomodulatory activity, bitter taste and hygroscopicity of hydrolysate derived from whey protein concentrate. LWT- Food Science and Technology, 56(2), 296–302. https://doi.org/10.1016/j.lwt.2013.12.019
Martins, I. B. A., Deliza, R., dos Santos, K. M. O., Walter, E. H. M., Martins, J. M., & Rosenthal, A. (2018). Viability of probiotics in goat cheese during storage and under simulated gastrointestinal conditions. Food and Bioprocess Technology, 11, 853–863. https://doi.org/10.1007/s11947-018-2060-2
Mateos-Maces, L., Chávez-Servia, J. L., Vera-Guzmán, A. M., Aquino-Bolaños, E. N., Alba-Jiménez, J. E., & Villagómez-González, B. B. (2020). Edible leafy plants from Mexico as sources of antioxidant compounds, and their nutritional, nutraceutical and antimicrobial potential: A review. Antioxidants, 9(6), 541. https://doi.org/10.3390/antiox9060541
Miike, S., McWilliam, A. S., & Kita, H. (2001). Trypsin induces activation and inflammatory mediator release from human eosinophils through protease-activated receptor-2. The Journal of Immunology, 167(11), 6615–6622. https://doi.org/10.4049/jimmunol.167.11.6615
Mishra, V., Shah, C., Mokashe, N., Chavan, R., Yadav, H., & Prajapati, J. (2015). Probiotics as potential antioxidants: A systematic review. Journal of Agricultural and Food Chemistry, 63, 3615–3626. https://doi.org/10.1021/jf506326t
Misra, S., Pandey, P., Dalbhagat, C. G., & Mishra, H. N. (2022). Emerging technologies and coating materials for improved probiotication in food products: A review. Food and Bioprocess Technology, 15, 998–1039. https://doi.org/10.1007/s11947-021-02753-5
Monteagudo-Mera, A., Rastall, R. A., Gibson, G. R., Charalampopoulos, D., & Chatzifragkou, A. (2019). Adhesion mechanisms mediated by probiotics and prebiotics and their potential impact on human health. Applied Microbiology and Biotechnology, 103(16), 6463–6472. https://doi.org/10.1007/s00253-019-09978-7
Mozzi, F., Gerbino, E., Font de Valdez, G., & Torino, M. I. (2008). Functionality of exopolysaccharides produced by lactic acid bacteria in an in vitro gastric system. Journal of Applied Microbiology, 107, 56–64. https://doi.org/10.1111/j.1365-2672.2009.04182.x
Nataraj, B. H., Ali, S. A., Behare, P. V., & Yadav, H. (2020). Postbiotics-parabiotics: The new horizons in microbial biotherapy and functional foods. Microbial Cell Factories, 19(1), 1–22. https://doi.org/10.1186/s12934-020-01426-w
Ndiaye, F., Vuong, T., Duarte, J., Aluko, R. E., & Matar, C. (2012). Anti-oxidant, anti- inflammatory and immunomodulating properties of an enzymatic protein hydrolysate from yellow field pea seeds. European Journal of Nutrition, 51, 29e3. https://doi.org/10.1007/s00394-011-0186-3
Pereira, A. L. F., Almeida, F. D. L., de Jesus, A. L. T., da Costa, J. M. C., & Rodrigues, S. (2013). Storage stability and acceptance of probiotic beverage from cashew apple juice. Food and Bioprocess Technology, 6, 3155–3165.
Peres, C. M., Alves, M., Hernández-Mendoza, A., Moreira, L., Silva, S., Bronze, M. R., Vilas-Boas, L., Peres, C., & Malcata, F. X. (2014). Novel isolates of Lactobacilli from fermented Portuguese olive as potential probiotics. LWT- Food Science and Technology, 59, 234–246. https://doi.org/10.1016/j.lwt.2014.03.003
Prete, R., Long, S. L., Gallardo, A. L., Gahan, C. G., Corsetti, A., & Joyce, S. A. (2020). Beneficial bile acid metabolism from Lactobacillus plantarum of food origin. Scientific Reports, 10, 1165. https://doi.org/10.1038/s41598-020-58069-5
Ranadheera, C. S., Vidanarachchi, J. K., Rocha, R. S., Cruz, A. G., & Ajlouni, S. (2017). Probiotic delivery through fermentation: Dairy vs. non-dairy beverages. Fermentation, 3, 67. https://doi.org/10.3390/fermentation3040067
Ranadheera, C. S., Naumovski, N., & Ajlouni, S. (2018). Non-bovine milk products as emerging probiotic carriers: Recent developments and innovations. Current Opinion in Food Science, 22, 109–114. https://doi.org/10.1016/j.cofs.2018.02.010
Reunanen, J., von Ossowski, I., Hendrickx, A. P. A., Palva, A., & de Vosa, W. M. (2008). Characterization of the SpaCBA pilus fibers in the probiotic Lactobacillus rhamnosus GG. Applied and Environmental Microbiology, 78, 2337–2334. https://doi.org/10.1128/AEM.07047-11
Rivas-Jimenez, L., Ramírez-Ortiz, K., González-Córdova, A. F., Vallejo-Cordoba, B., Garcia, H. S., & Hernandez-Mendoza, A. (2016). Evaluation of acrylamide-removing properties of two Lactobacillus strains under simulated gastrointestinal conditions using a dynamic system. Microbiological Research, 190, 19–26. https://doi.org/10.1016/j.micres.2016.04.016
Rokana, N., Singh, B. P., Thakur, N., Sharma, C., Gulhane, R. D., & Panwar, H. (2018). Screening of cell surface properties of potential probiotic lactobacilli isolated from human milk. Journal of Dairy Research, 85, 347–354. https://doi.org/10.1017/S0022029918000432
Roos, S., Karner, F., Axelsson, L., & Jonsson, H. (2000). Lactobacillus mucosae sp. nov., a new species with in vitro mucus-binding activity isolated from pig intestine. International Journal of Systematic and Evolutionary Microbiology, 50, 251–258. https://doi.org/10.1099/00207713-50-1-251
Ruas-Madiedo, P., Hugenholtz, J., & Zoon, P. (2002). An overview of the functionality of exopolysaccharides produced by lactic acid bacteria. International Dairy Journal, 12(2–3), 163–171. https://doi.org/10.1016/S0958-6946(01)00160-1
Ruiz, L., Margolles, A., & Sánchez, B. (2013). Bile resistance mechanisms in Lactobacillus and Bifidobacterium. Frontiers in Microbiology, 4, 396. https://doi.org/10.3389/fmicb.2013.00396
Sagdic, O., Ozturk, I., Cankurt, H., & Tornuk, F. (2012). Interaction between some phenolic compounds and probiotic bacterium in functional ice cream production. Food and Bioprocess Technology, 5, 2964–2971. https://doi.org/10.1007/s11947-011-0611-x
Saini, K., & Minj, J. (2020). Multifunctional aspects of probiotics and prebiotics in health management: An overview. In J. Minj, V. A. Sudhakaran, & A. Kumari (Eds.), Dairy processing: Advanced research to applications (pp. 119–131). Singapore: Springer. https://doi.org/10.1007/978-981-15-2608-4_17
Santiago-López, L., Hernández-Mendoza, A., Mata-Haro, V., Vallejo-Cordoba, B., & González-Córdova, A. F. (2018). Immune response induced by fermented milk with potential probiotic strains isolated from artisanal Cocido cheese. Food Agricultural and Immunology, 29, 911–929. https://doi.org/10.1080/09540105.2018.1485632
Sornplang, P., & Piyadeatsoontorn, S. (2016). Probiotic isolates from unconventional sources: A review. Journal of Animal Science and Technology, 58(1), 1–11. https://doi.org/10.1186/s40781-016-0108-2
Srivastava, P., Vyas, V. K., Variya, B., Patel, P., Qureshi, G., & Ghate, M. (2016). Synthesis, anti-inflammatory, analgesic, 5-lipoxygenase (5-LOX) inhibition activities, and molecular docking study of 7-substituted coumarin derivatives. Bioorganic Chemistry, 67, 130–138. https://doi.org/10.1016/j.bioorg.2016.06.004
Tallon, R., Bressollier, P., & Urdaci, M. C. (2003). Isolation and characterization of two exopolysaccharides produced by Lactobacillus plantarum EP56. Research in Microbiology, 154(10), 705–712. https://doi.org/10.1016/j.resmic.2003.09.006
Taverniti, V., & Guglielmetti, S. (2011). The immunomodulatory properties of probiotic microorganisms beyond their viability (ghost probiotics: Proposal of paraprobiotic concept). Genes & Nutrition, 6(3), 261–274. https://doi.org/10.1007/s12263-011-0218-x
Teame, & T., Wang, A., **e, M., Zhang, Z., Yang, Y., Ding, Q., Gao, C., Olsen, R.E., Ran, C., Zhou, Z. (2020). Paraprobiotics and postbiotics of probiotic lactobacilli, their positive effects on the host and action mechanisms: A review. Frontiers in Nutrition, 7, 570344. https://doi.org/10.3389/fnut.2020.570344
Thirabunyanon, M., Boonprasom, P., & Niamsup, P. (2009). Probiotic potential of lactic acid bacteria isolated from fermented dairy milks on antiproliferation of colon cancer cells. Biotechnology Letters, 31(4), 571–576. https://doi.org/10.1007/s10529-008-9902-3
Thorakkattu, P., Khanashyam, A. C., Shah, K., Babu, K. S., Mundanat, A. S., Deliephan, A., Deokar, G. S., Santivarangkna, C., & Nirmal, N. P. (2022). Postbiotics: Current trends in food and pharmaceutical industry. Foods, 11(19), 3094. https://doi.org/10.3390/foods11193094
Turroni, F., Bottacini, F., Foroni, E., Mulder, I., Kim, J. H., Zomer, A., Sánchez, B., Bidossi, A., Ferrarini, A., Giubellini, V., Delledonne, M., Henrissat, B., Coutinho, P., Oggioni, M., Fitzgerald, G. F., Mills, D., Margolles, A., Kelly, D., van Sinderen, D., & Ventura, M. (2010). Genome analysis of Bifidobacterium bifidum PRL2010 reveals metabolic pathways for host-derived glycan foraging. Proceedings of the National Academy Science, 107(45), 19514–19519. https://doi.org/10.1073/pnas.1011100107
Vallejo-Cordoba, B., Castro-López, C., García, H. S., González-Córdova, A. F., & Hernández-Mendoza, A. (2020). Postbiotics and paraprobiotics: A review of current evidence and emerging trends. Advances in Food Nutrition Research, 94, 1–34. https://doi.org/10.1016/bs.afnr.2020.06.001
Vinderola, C. G., Medici, M., & Perdigón, G. (2004). Relationship between interaction sites in the gut, hydrophobicity, mucosal immunomodulating capacities and cell wall protein profiles in indigenous and exogenous bacteria. Journal of Applied Microbiology, 96, 230–243. https://doi.org/10.1046/j.1365-2672.2004.02158.x
Vinderola, C. G., & Reinheimer, J. A. (2003). Lactic acid starter and probiotic bacteria: A comparative “in vitro” study of probiotic characteristics and biological barrier resistance. Food Research International, 36, 895–904. https://doi.org/10.1016/S0963-9969(03)00098-X
Vinderola, G., Sanders, M. E., Salminen, S., & Szajewska, H. (2022). Postbiotics: The concept and their use in healthy populations. Frontiers in Nutrition, 9, 1002213. https://doi.org/10.3389/fnut.2022.1002213
Wang, Y., Wu, Y., Wang, Y., Xu, H., Mei, X., Yu, D., & Wang, Y. (2017). Antioxidant properties of probiotic bacteria. Nutrients, 19, 521. https://doi.org/10.3390/nu9050521
Wegh, C., Geerlings, S. Y., Knol, J., Roeselers, G., & Belzer, C. (2019). Postbiotics and their potential applications in early life nutrition and beyond. International Journal of Molecular Sciences, 20(19), 4673. https://doi.org/10.3390/ijms20194673
Wu, D., Fu, R., Scoffield, J., & Kannappan, R. (2018). Probiotic metabolites promote stem cell proliferation and differentiation. FASEB Journal, 32, 615–614. https://doi.org/10.1096/fasebj.2018.32.1_supplement.615.4
Xu, R., Shang, N., & Li, P. (2011). In vitro and in vivo antioxidant activity of exopolysaccharide fractions from Bifidobacterium animalis RH. Anaerobe, 17(5), 226–231. https://doi.org/10.1016/j.anaerobe.2011.07.010
Yahfoufi, N., Alsadi, N., Jambi, M., & Matar, C. (2018). The immunomodulatory and anti-inflammatory role of polyphenols. Nutrients, 10(11), 1618. https://doi.org/10.3390/nu10111618
Yang, R. Y., Zhang, Z. F., Pei, X. R., Han, X. L., Wang, J. B., Wang, L., Long, Z., Shen, X., & Li, Y. (2009). Immunomodulatory effects of marine oligopeptide preparation from Chum Salmon (Oncorhynchus keta) in mice. Food Chemistry, 113, 464e470. https://doi.org/10.1016/j.foodchem.2008.07.086
Yoon, S., Cho, H., Nam, Y., Park, M., Lim, A., Kim, J. H., ... & Kim, W. (2022). Multifunctional probiotic and functional properties of Lactiplantibacillus plantarum LRCC5314, isolated from kimchi. Journal of Microbiology and Biotechnology, 32(1), 72–80. https://doi.org/10.4014/jmb.2109.09025
Zacarias, M. F., Binetti, A., Laco, M., Reinheimer, J., & Vinderola, G. (2011). Preliminary technological and potential probiotic characterization of Bifidobacterial isolated from breast milk for use in dairy products. International Dairy Journal, 21, 548–555. https://doi.org/10.1016/j.idairyj.2011.03.007
Ziarno, M., & Cichońska, P. (2021). Lactic acid bacteria-fermentable cereal- and pseudocereal-based beverages. Microorganisms, 9(12), 2532. https://doi.org/10.3390/microorganisms9122532
Zielinska, D., & Kolozyn-Krajewska, D. (2018). Food-origin lactic acid bacteria may exhibit probiotic properties: Review. BioMed Research International, 2018, 5063185. https://doi.org/10.1155/2018/5063185
Zommiti, M., Feuilloley, M. G., & Connil, N. (2020). Update of probiotics in human world: A nonstop source of benefactions till the end of time. Microorganisms, 8(12), 1907. https://doi.org/10.3390/microorganisms8121907
Zulueta, A., Esteve, M. J., & Frígola, A. (2009). ORAC and TEAC assays comparison to measure the antioxidant capacity of food products. Food Chemistry, 114, 310–316. https://doi.org/10.1016/j.foodchem.2008.09.033
Acknowledgements
The authors thank Maria del Carmen Estrada Montoya for their technical assistance for the standardization of analytical techniques; we also thank the Probiotic Functional Foods for Malnourished Childhood Populations, Proinfant Project (Ref. P916PTE0233), funded by CYTED (Programa Iberoamericano de Ciencia y Tecnología para el Desarrollo) from Spain, and the National Network on Research, Innovation and Technological Development on Functional Foods and Nutraceuticals-ALFANUTRA, CONACyT‐244208.
Funding
This research was supported by the Mexican Council of Science and Technology (CONACyT; Mexico City) research grant 319922.
Author information
Authors and Affiliations
Contributions
Adrián Hernández-Mendoza and Hugo S. García: Conceptualization, Project administration. Lourdes Santiago-López: Date curation, Methodology. Lourdes Santiago-López and Lilia M. Beltrán-Barrientos: Writing-original draft, formal analysis. José I. Méndez-Romero: Molecular identification. Adrián Hernández-Mendoza, Hugo S. García, Aarón F. González-Córdova and Belinda Vallejo-Cordoba: Resources, Supervision, Writing-review and editing.
Corresponding authors
Ethics declarations
Competing of Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Santiago-López, L., Garcia, H.S., Beltrán-Barrientos, L.M. et al. Probiotic Potential of Bacteria Isolated from Huauzontle (Chenopodium berlandieri spp. Nuttalliae) and Multifunctional Properties of Their Intracellular Contents. Food Bioprocess Technol (2024). https://doi.org/10.1007/s11947-024-03334-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11947-024-03334-y