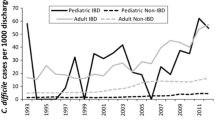
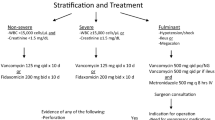
Abstract
Clostridium difficile (C. difficile) infection has emerged as a significant clinical challenge for patients suffering from inflammatory bowel disease (IBD). C. difficile can both precipitate and worsen flares of IBD, contributing to emergent colectomies and mortality. Advances in the management of C. difficile infection in IBD include recommendations for testing for this infection in the setting of clinical flare and hospitalization, improved diagnostic testing, identification of high rates of carriage and infection in pediatric IBD, and new data associating patterns of IBD genetic risk alleles with the development of this infection. Therapeutically, oral vancomycin has emerged as a superior treatment for IBD patients with moderate to severe disease compared with metronidazole. Although highly effective in the general population, fecal microbiome transplantation for recurrent C. difficile infection in IBD patients has been associated with colitis flare in the majority of patients who have received this treatment.
Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as • Of importance
Kelly CP, LaMont JT. Clostridium difficile—more difficult than ever. N Engl J Med. 2008;359(18):1932–40. Erratum in: N Engl J Med. 2010 Oct 14;363(16):1585.
Rodemann JF, Dubberke ER, Reske KA, da Seo H, Stone CD. Incidence of Clostridium difficile infection in inflammatory bowel disease. Clin Gastroenterol Hepatol. 2007;5(3):339–44.
Issa M, Vijayapal A, Graham MB, Beaulieu DB, Otterson MF, Lundeen S, et al. Impact of Clostridium difficile on inflammatory bowel disease. Clin Gastroenterol Hepatol. 2007;5(3):345–51.
Ananthakrishnan AN, McGinley EL, Binion DG. Excess hospitalisation burden associated with Clostridium difficile in patients with inflammatory bowel disease. Gut. 2008;57(2):205–10.
Burke KE, Lamont JT. Clostridium difficile inection: a worldwide disease. Gut Liver. 2014;8(1):1–6.
Jen MH, Saxena S, Bottle A, Aylin P, Pollok RC. Increased health burden associated with Clostridium difficile diarrhoea in patients with inflammatory bowel disease. Aliment Pharmacol Ther. 2011;33(12):1322–31.
Murthy SK, Steinhart AH, Tinmouth J, Austin PC, Daneman N, Nguyen GC. Impact of Clostridium difficile colitis on 5-year health outcomes in patients with ulcerative colitis. Aliment Pharmacol Ther. 2012;36(11–12):1032–9. Approximately 9% of hospitalized ulcerative colitis patients have C. difficile infection in this study from Ontario, Canada.
Masclee GM, Penders J, Jonkers DM, Wolffs PF, Pierik MJ. Is Clostridium difficile associated with relapse of inflammatory bowel disease? Results from a retrospective and prospective cohort study in the Netherlands. Inflamm Bowel Dis. 2013;19(10):2125–31.
Hourigan SK, Chirumamilla SR, Ross T, Golub JE, Rabizadeh S, Saeed SA, et al. Clostridium difficile carriage and serum antitoxin responses in children with inflammatory bowel disease. Inflamm Bowel Dis. 2013;19(13):2744–52.
Mir SA, Kellermayer R. Clostridium difficile infection in newly diagnosed pediatric inflammatory bowel disease in the mid-southern United States. J Pediatr Gastroenterol Nutr. 2013;57(4):487–8. Approximately 8% of newly diagnosed pediatric IBD patients have C . difficile infection, which supports the practice of testing of all newly diagnosed IBD patients.
Dave M, Purohit T, Razonable R, Loftus Jr EV. Opportunistic infections due to inflammatory bowel disease therapy. Inflamm Bowel Dis. 2014;20(1):196–212.
Schneeweiss S, Korzenik J, Solomon DH, Canning C, Lee J, Bressler B. Infliximab and other immunomodulating drugs in patients with inflammatory bowel disease and the risk of serious bacterial infections. Aliment Pharmacol Ther. 2009;30(3):253–64. Corticosteroid exposure tripled risk of IBD patients develo** C. difficile infection in this study from British Columbia, Canada. Anti-TNF agents were not associated with developoing C. difficile in IBD.
Ananthakrishnan AN, McGinley EL. Infection-related hospitalizations are associated with increased mortality in patients with inflammatory bowel diseases. J Crohn’s Colitis. 2013;7(2):107–12.
Li Y, Qian J, Queener E, Shen B. Risk factors and outcome of PCR-detected Clostridium difficile infection in ileal pouch patients. Inflamm Bowel Dis. 2013;19(2):397–403.
Jostins L, Ripke S, Weersma RK, Duerr RH, McGovern DP, Hui KY, et al. Host–microbe interactions have shaped the genetic architecture of inflammatory bowel disease. Nature. 2012;491(7422):119–24.
Ananthakrishnan AN, Oxford EC, Nguyen DD, Sauk J, Yajnik V, Xavier RJ. Genetic risk factors for Clostridium difficile infection in ulcerative colitis. Aliment Pharmacol Ther. 2013;38(5):522–30. Female gender, pancolitis, and anti-TNF therapy were protective against develo** C. difficile infection in this analysis of a small number of ulcerative colitis patients. Increasing number of IBD risk alleles were associated with increased risk of develo** C. difficile infection in ulcerative colitis.
Deshpande A, Pasupuleti V, Patel P, Pant C, Pagadala M, Hall G, et al. Repeat stool testing for Clostridium difficile using enzyme immunoassay in patients with inflammatory bowel disease increases diagnostic yield. Curr Med Res Opin. 2012;28(9):1553–60.
Wang Y, Atreja A, Wu X, Lashner BA, Brzezinski A, Shen B. Similar outcomes of IBD in patients with Clostridium difficile infection detected by ELISA or PCR assay. Dig Dis Sci. 2013;58(8):2308–13.
Sunkesula VC, Kundrapu S, Muganda C, Sethi AK, Donskey CJ. Does empirical Clostridium difficile infection (CDI) therapy result in false-negative CDI diagnostic test results? Clin Infect Dis. 2013;57(4):494–500. Analysis of the performance characteristics of stool PCR testing for C. difficile following initiation of antibiotic therapy against the infection in patients with documented disease. PCR tests turned negative at a rapid rate, with 50% of patients testing negative 3 days into therapy and essentially all patients becoming negative 5 days into treatment. This study demonstrates no role for repeat stool testing as the PCR assay will turn negative prior to completing a therapeutic course of antibiotic therapy for C. difficile infection. It also highlights the importance of completing an empiric course of therapy once initiated, as testing is no longer accurate 3–5 days into treatment.
Surawicz CM, Brandt LJ, Binion DG, Ananthakrishnan AN, Curry SR, Gilligan PH, et al. Guidelines for diagnosis, treatment, and prevention of Clostridium difficile infections. Am J Gastroenterol. 2013;108(4):478–98. quiz 499. Review.
Ananthakrishnan AN, Guzman-Perez R, Gainer V, Cai T, Churchill S, Kohane I, et al. Predictors of severe outcomes associated with Clostridium difficile infection in patients with inflammatory bowel disease. Aliment Pharmacol Ther. 2012;35(7):789–95. Observational clinical trial demonstrating clinical factors which correlate with poor outcome in IBD patients suffering from C. difficile infection.
Libot A, Issa M, Zadvornova Y, Stein DJ, Venu N, Perera LP, et al. Initial vancomycin monotherapy is associated with higher rates of subsequent Clostridium difficile infection in inflammatory bowel disease population. Gastroenterology. 2011;140:S-159. abstract #992.
Louie TJ, Miller MA, Mullane KM, Weiss K, Lentnek A, Golan Y, et al. Fidaxomicin versus vancomycin for Clostridium difficile infection. N Engl J Med. 2011;364(5):422–31. Pivotal trial demonstrating safety and efficacy of fidaxomicin in the treatment of initial C. difficile infection and potential superiority with reduction in short term recurrence. IBD patients were excluded from this trial.
van Nood E, Vrieze A, Nieuwdorp M, et al. Duodenal infusion of donor feces for recurrent Clostridium difficile. N Engl J Med. 2013;368:407–15. Pivotal trial establishing the efficacy and superiority of FMT in the treatment of recurrent C. difficile infection.
Anderson JL, Edney RJ, Whelan K. Systematic review: Faecal microbiota transplantation in the management of inflammatory bowel disease. Aliment Pharmacol Ther. 2012;36(6):503–16.
De Leon LM, Watson JB, Kelly CR. Transient flare of ulcerative colitis after fecal microbiota transplantation for recurrent Clostridium difficile infection. Clin Gastroenterol Hepatol. 2013;11(8):1036–8. First publication demonstrating problems of FMT for treatment of recurrent C difficile infection in IBD.
Vermeire S, Joossens M, Verbeke K, et al. Pilot study on the safety and effi cacy of faecal microbiota transplantation in refractory Crohn’s disease. Gastroenterology. 2012;142:S360.
Angelberger S, Reinisch W, Makristathis A, Lichtenberger C, Dejaco C, Papay P, et al. Temporal bacterial community dynamics vary among ulcerative colitis patients after fecal microbiota transplantation. Am J Gastroenterol. 2013;108(10):1620–30.
Compliance with Ethics Guidelines
Conflict of Interest
Jana G. Hashash and David G. Binion have nothing to disclose.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is part of the Topical Collection on Inflammatory Bowel Disease
Rights and permissions
About this article
Cite this article
Hashash, J.G., Binion, D.G. Managing Clostridium difficile in Inflammatory Bowel Disease (IBD). Curr Gastroenterol Rep 16, 393 (2014). https://doi.org/10.1007/s11894-014-0393-1
Published:
DOI: https://doi.org/10.1007/s11894-014-0393-1