Abstract
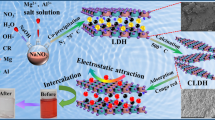
The synthesis of hierarchically porous materials from industrial solid waste as catalysts or adsorbents has attracted much attention in environmental pollution control. The present work highlights the synthesis of a series of MgFeAl layered double hydroxide (LDH) with different ratios of Mg/(Fe + Al) from red mud and ferronickel slag under different pH to decolorize three red dye solutions, namely, Congo red (CR), acid red (AR), and reactive red (RR). The maximum adsorption capacity (386.1 mg.g−1) for CR was obtained by the MgFeAl-LDH sample with Mg/(Fe + Al) mole ratio of 2 synthesized under pH 10. The optimized synthesis parameters led to a high surface area and large pore volume of MgFeAl-LDH, thereby enhancing the mass transfer of adsorbate to the active sites of the adsorbent. Further, the MgFeAl-LDH sample was also efficient in removing AR (120.9 mg.g−1) and RR (114 mg.g−1). Moreover, the adsorption kinetics and isotherms of MgFeAl-LDH fitted well with the pseudo-second-order and Langmuir models, respectively. The MgFeAl-LDH system developed a valuable approach to utilize industrial waste for dye removal.










Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article. The datasets used and/or analyzed in the current study are available from the corresponding author upon reasonable request.
References
Z.Q. Bai, Y.J. Zheng, Z.P. Zhang, J. Mater. Chem. A. 5, 6630 (2017)
F. Zhao, E. Repo, D. Yin, Y. Meng, S. Jafari, M. Sillanpa, Environ. Sci. Technol. 49, 10570 (2015)
H. Ma, A. Kong, Y. Ji, B. He, Y. Song, J. Li, J. Clean. Prod. 214, 89 (2019)
A. Ghaffar, L. Zhang, X. Zhu, B. Chen, Environ. Sci. Technol. 52, 4265 (2018)
G.A. Kloster, M.A. Mosiewicki, N.E. Marcovich, Carbohydr. Polym. 221, 186 (2019)
U.J. Kim, S. Kimura, M. Wada, Carbohydr. Polym. 214, 294 (2019)
S.J. Lim, T.H. Kim, Sep. Purif. Technol. 146, 42 (2015)
R. Li, B. Gao, K. Guo, Q. Yue, H. Zheng, Y. Wang, Bioresour. Technol. 240, 59 (2017)
D.B. Jiang, X. Liu, X. Xu, Y.X. Zhang, J. Phys. Chem. Solids 112, 209 (2018)
N.P. Khumalo, L.N. Nthunya, E. De Canck, S. Derese, A.R. Verliefde, A.T. Kuvarega, B.B. Mamba, S.D. Mhlanga, D.S. Dlamini, Sep. Purif. Technol. 211, 578 (2019)
D. Xu, B. Cheng, S. Cao, J. Yu, Appl. Catal. B Environ. 164, 380 (2015)
R.K. Sonwani, G. Swain, B.S. Giri, R.S. Singh, B.N. Rai, Bioresour. Technol. 302, 122811 (2020)
S. Singh, S. Perween, A. Ranjan, J. Environ. Chem. Eng. 9, 105149 (2021)
H. Chen, X. Wang, J. Li, X. Wang, J. Mater. Chem. A 3, 6073 (2015)
M. Nagpal, R. Kakkar, Sep. Purif. Technol. 211, 522 (2019)
M. Salari, M.H. Dehghani, A. Azari, M.D. Motevalli, A. Shabanloo, I. Ali, J. Mol. Liq. 285, 146 (2019)
M. Zubair, I. Ihsanullah, H.A. Aziz, M.A. Ahmad, M.A. Al-Harthi, Bioresour. Technol. 319, 124128 (2021)
Z.-H. **e, H.-Y. Zhou, C.-S. He, Z.-C. Pan, G. Yao, B. Lai, Chem. Eng. J. 414, 128713 (2021)
P.-F. Guo, Y. Yang, W.-J. Wang, B. Zhu, W.-T. Wang, Z.-Y. Wang, J.-L. Wang, K. Wang, Z.-H. He, Z.-T. Liu, Chem. Eng. J. 426, 130768 (2021)
R. Li, T. Xue, R. Bingre, Y. Gao, B. Louis, Q. Wang, A.C.S. Appl, Mater. Interfaces 10, 34834 (2018)
P. Sahoo, S. Ishihara, K. Yamada, K. Deguchi, S. Ohki, M. Tansho, T. Shimizu, N. Eisaku, R. Sasai, J. Labuta, D. Ishikawa, J.P. Hill, K. Ariga, B.P. Bastakoti, Y. Yamauchi, N. Iyi, A.C.S. Appl, Mater. Interfaces 6, 18352 (2014)
D. Bharali, R.C. Deka, J. Environ. Chem. Eng. 5, 2056 (2017)
H. Hu, S. Wageh, A.A. Al-Ghamdi, S. Yang, Z. Tian, B. Cheng, W. Ho, Appl. Surf. Sci. 511, 145570 (2020)
N. Hossain, M.A. Bhuiyan, B.K. Pramanik, S. Nizamuddin, G. Griffin, J. Clean. Prod. 255, 120261 (2020)
D. Sun, X. Zhang, Y. Wu, X. Liu, J. Hazard. Mater. 181, 335 (2010)
S. Wang, Y. Boyjoo, A. Choueib, Z.H. Zhu, Water Res. 39, 129 (2005)
L. Spessato, K.C. Bedin, A.L. Cazetta, I.P.A.F. Souza, V.A. Duarte, L.H.S. Crespo, M.C. Silva, R.M. Pontes, V.C. Almeida, J. Hazard. Mater. 371, 499 (2019)
J. Ma, Y. Liu, O. Ali, Y. Wei, S. Zhang, Y. Zhang, T. Cai, C. Liu, S. Luo, J. Hazard. Mater. 344, 1034 (2018)
N. Ye, Y. Chen, J. Yang, S. Liang, Y. Hu, B. **ao, Q. Huang, Y. Shi, J. Hu, X. Wu, J. Hazard. Mater. 318, 70 (2016)
N. You, B. Li, R. Cao, J. Shi, C. Chen, Y. Zhang, Constr. Build. Mater. 227, 116614 (2019)
B. Li, B. Huo, R. Cao, S. Wang, Y. Zhang, Cem. Concr. Compos. 96, 204 (2019)
S. Zhang, Q. Wang, P. Puthiaraj, W.-S. Ahn, J. Co2 Util. 34, 395 (2019)
J. Tu, X. Peng, S. Wang, C. Tian, H. Deng, Z. Dang, G. Lu, Z. Shi, Z. Lin, Sci. Total. Environ. 677, 556 (2019)
Y. Su, S. Qiu, D. Yang, S. Liu, H. Zhao, L. Wang, Q. Xu, J. Hazard. Mater. 391, 122215 (2020)
Q. Sun, M. Tang, P.V. Hendriksen, B. Chen, J. Alloy. Compound. 829, 154552 (2020)
X. Zhu, C. Chen, H. Suo, Q. Wang, Y. Shi, D. O’Hare, N. Cai, Energy 167, 960 (2019)
C. Chen, M. Yang, Q. Wang, J.-C. Buffet, D. O’Hare, J. Mater. Chem. A 2, 15102 (2014)
J. Li, H. Yu, X. Zhang, R. Zhu, L. Yan, Front. Environ. Sci. Eng. 14, 52 (2020)
R. Shan, L. Yan, Y. Yang, K. Yang, S. Yu, H. Yu, B. Zhu, B. Du, J. Ind. Eng. Chem. 21, 561 (2015)
I.M. Ahmed, M.S. Gasser, Appl. Surf. Sci. 259, 650 (2012)
S. Yang, L. Wang, X. Zhang, W. Yang, G. Song, Chem. Eng. J. 275, 315 (2015)
R. Lafi, K. Charradi, M.A. Djebbi, A.B.H. Amara, A. Hafiane, Adv. Powder Technol. 27, 232 (2016)
M. Li, G. Wu, Z. Liu, X. **, Y. **a, J. Ning, D. Yang, A. Dong, J. Hazard. Mater. 397, 122766 (2020)
Y. Zheng, B. Cheng, W. You, J. Yu, W. Ho, J. Hazard. Mater. 369, 214 (2019)
Acknowledgements
This work was supported by Inha University Research Grant.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, S., Lee, J., Li, B. et al. Industrial Solid Waste-Based Layered Double Hydroxide Composite with Enhanced Adsorption Affinity to Congo Red, Acid Red, and Reactive Red. Korean J. Chem. Eng. 41, 829–838 (2024). https://doi.org/10.1007/s11814-024-00088-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-024-00088-3