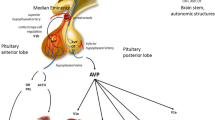
Abstract
The radioimmunoassay (RIA) method is widely used to determine the levels of arginine vasopressin (AVP) in studies, especially in cell culture studies. However, there are many difficulties and disadvantages in performing this method. Therefore, this study aimed to assess the comparison between enzyme-linked immunosorbent assay (ELISA) and RIA methods by using the Bland–Altman statistical method and to investigate whether the commonly used RIA can be replaced by the ELISA method for measurement of AVP levels in cell culture medium. For this purpose, wild-type (WT) and three different mutant AVP-NPII vectors were transiently transfected to mouse neuroblastoma (Neuro2A) cells and AVP secretion into the cell culture medium by transfected Neuro2A cells was determined by both RIA and ELISA methods. Following the use of the normalization method, Bland–Altman method, currently the most commonly used statistical method assessing comparison between two methods, was performed to assess agreement between two methods. The order of normalized AVP values obtained using the RIA method was as follows: WT (0.921 ± 0.08), 207_209delGGC mutant (0.801 ± 0.09), G45C mutant (0.508 ± 0.10), and G88V mutant (0.497 ± 0.12). The normalized AVP values obtained using the ELISA method were as follows: WT (0.865 ± 0.12), 207_209delGGC mutant (0.704 ± 0.13), G88V mutant (0.255 ± 0.16), and G45C mutant (0.250 ± 0.15). The order of AVP levels measured from each transfected cell using both methods was quite similar. According to the Bland–Altman method, there is an agreement between RIA and ELISA for measuring the AVP levels in cell culture. It can be recommended to apply ELISA instead of the RIA method for determinating AVP levels.




Similar content being viewed by others
Abbreviations
- ADH:
-
Antidiuretic hormone
- AQP2:
-
Aquaporin 2
- AVP:
-
Arginine vasopressin
- AVPR2:
-
Arginine vasopressin type 2 receptor
- BCA:
-
Bicinchoninic acid
- °C:
-
Celsius
- cDNA:
-
Complementary DNA
- DI:
-
Diabetes insipidus
- EGFP:
-
Enhanced green fluorescent protein
- ELISA:
-
Enzyme-linked immunosorbent assay
- EMEM:
-
Eagle's Minimum Essential Medium
- ER:
-
Endoplasmic reticulum
- FNDI:
-
Familial neurohypophyseal diabetes insipidus
- g:
-
Centrifugal force
- h:
-
Hour
- 125I:
-
Iodine-125
- L:
-
Liter
- LoA:
-
Limits of agreement
- mg:
-
Milligram
- min:
-
Minute
- mL:
-
Milliliter
- mM:
-
Millimolar
- Neuro2A:
-
Neuroblastoma
- nm:
-
Nanometer
- NPII:
-
Neurophysin 2
- PCP:
-
Porcine C-peptide
- pg:
-
Picogram
- pmol:
-
Picomole
- RIA:
-
Radioimmunoassay
- SD:
-
Standard deviations
- U:
-
Unit
- WT:
-
Wild-type
- µg:
-
Microgram
- μm:
-
Micrometer
References
Abbes AP, Bruggeman B, van Den Akker EL, de Groot MR, Franken AA, Drexhage VR, Engel H (2000) Identification of two distinct mutations at the same nucleotide position, concomitantly with a novel polymorphism in the vasopressin-neurophysin II gene (AVP-NP II) in two Dutch families with familial neurohypophyseal diabetes insipidus. Clin Chem 46(10):1699–1702. https://doi.org/10.1093/clinchem/46.10.1699
Arima H, Oiso Y (2010) Mechanisms underlying progressive polyuria in familial neurohypophysial diabetes insipidus. J Neuroendocrinol 22(7):754–757. https://doi.org/10.1111/j.1365-2826.2010.02028.x
Babaei Jandaghi A, Shakiba M, Nasseh H, Korouji Y, Esmaeili S, Khadem Maboudi AA, Khorshidi A (2015) Application of Bland-Altman method in comparing transrectal and transabdominal ultrasonography for estimating prostate volume. Iran J Med Sci 40(1):34–39
Babey M, Kopp P, Robertson GL (2011) Familial forms of diabetes insipidus: clinical and molecular characteristics. Nat Rev Endocrinol 7:701–714. https://doi.org/10.1038/nrendo.2011.100
Ball SG (2007) Vasopressin and disorders of water balance: the physiology and pathophysiology of vasopressin. Ann Clin Biochem 44:417–431. https://doi.org/10.1258/000456307781646030
Baribeau DA, Anagnostou E (2015) Oxytocin and vasopressin: linking pituitary neuropeptides and their receptors to social neurocircuits. Front Neurosci 9(335):1–21. https://doi.org/10.3389/fnins.2015.00335
Bichet DG (2012) Genetics and diagnosis of central diabetes insipidus. Ann Endocrinol (Paris) 73(2):117–127. https://doi.org/10.1016/j.ando.2012.03.030
Bland JM, Altman DG (1986) Statistical methods for assessing agreement between two methods of clinical measurement. Lancet 1(8476):307–310. https://doi.org/10.1016/S0140-6736(86)90837-8
Bland JM, Altman DG (2010) Statistical methods for assessing agreement between two methods of clinical measurement. Int J Nurs Stud 47:931–936. https://doi.org/10.1016/j.ijnurstu.2009.10.0015
Bolignano D, Cabassi A, Fiaccadori E, Ghigo E, Pasquali R, Peracino A, Peri A, Plebani M, Santoro A, Settanni F, Zoccali C (2014) Copeptin (CTproAVP), a new tool for understanding the role of vasopressin in pathophysiology. Clin Chem Lab Med 52(10):1447–1456. https://doi.org/10.1515/cclm-2014-0379
Brydges NM, Hall J, Best C, Rule L, Watkin H, Drake AJ, Lewis C, Thomas KL, Hall J (2019) Childhood stress impairs social function through AVP-dependent mechanisms. Transl Psychiatry 9(330):1–12. https://doi.org/10.1038/s41398-019-0678-0
Bunce C (2009) Correlation, agreement, and Bland-Altman analysis: statistical analysis of method comparison studies. Am J Ophthalmol 148(1):4–6. https://doi.org/10.1016/j.ajo.2008.09.032
Christensen JH, Siggaard C, Corydon TJ, Robertson GL, Gregersen N, Bolund L, Rittig S (2004a) Differential cellular handling of defective arginine vasopressin (AVP) prohormones in cells expressing mutations of the AVP gene associated with autosomal dominant and recessive familial neurohypophyseal diabetes insipidus. J Clin Endocrinol Metab 89(9):4521–4531. https://doi.org/10.1210/jc.2003-031813
Christensen JH, Siggaard C, Corydon TJ, deSanctis L, Kovacs L, Robertson GL, Gregersen N, Rittig S (2004b) Six novel mutations in the arginine vasopressin gene in 15 kindreds with autosomal dominant familial neurohypophyseal diabetes insipidus give further insight into the pathogenesis. Eur J Hum Genet 12(1):44–51. https://doi.org/10.1038/sj.ejhg.5201086
Christensen JH, Siggaard C, Corydon TJ, Robertson GL, Gregersen N, Bolund L, Rittig S (2004c) Impaired trafficking of mutated AVP prohormone in cells expressing rare disease genes causing autosomal dominant familial neurohypophyseal diabetes insipidus. Clin Endocrinol (oxf) 60:125–136. https://doi.org/10.1111/j.1365-2265.2004.01953.x
Currie IS, Gillies G, Brooks AN (1994) Modulation of arginine vasopressin secretion from cultured ovine hypothalamic cells by glucocorticoids and opioid peptides. Neuroendocrinology 60(4):360–367. https://doi.org/10.1159/000126770
Custer EE, Ortiz-Miranda S, Knott TK, Rawson R, Elvey C, Lee RH, Lemos JR (2007) Identification of the neuropeptide content of individual rat neurohypophysial terminals. J Neurosci Methods 163:226–234. https://doi.org/10.1016/j.jneumeth.2007.03.006
Dayanithi G, Forostyak O, Forostyak S, Kayano T, Ueta Y, Verkhratsky A (2018) Vasopressin and oxytocin in sensory neurones: expression, exocytotic release and regulation by lactation. Sci Rep 8(1):13084. https://doi.org/10.1038/s41598-018-31361-1
Di Iorgi N, Napoli F, Allegri AE, Olivieri I, Bertelli E, Gallizia A, Rossi A, Maghnie M (2012) Diabetes insipidus–diagnosis and management. Horm Res Paediatr 77(2):69–84. https://doi.org/10.1159/000336333
Ehrlich L, Scrushy M, Meng F, Lairmore TC, Alpini G, Glaser S (2018) Biliary epithelium: A neuroendocrine compartment in cholestatic liver disease. Clin Res Hepatol Gastroenterol 42:296–305. https://doi.org/10.1016/j.clinre.2018.03.009
Erfanian S, Yazdanpour L, Javeshghani D, Roustazadeh A (2019) Association of arginine vasopressin (AVP) promoter polymorphisms with preeclampsia. Pregnancy Hypertens 18:122–125. https://doi.org/10.1016/j.preghy.2019.09.017
Frijling JL, van Zuiden M, Nawijn L, Koch SB, Neumann ID, Veltman DJ, Olff M (2015) Salivary oxytocin and vasopressin levels in police officers with and without post-traumatic stress disorder. J Neuroendocrinol 27(10):743–751. https://doi.org/10.1111/jne.12300
Gallardo-Estrella L, Lynch DA, Prokop M, Stinson D, Zach J, Judy PF, van Ginneken B, van Rikxoort EM (2016) Normalizing computed tomography data reconstructed with different filter kernels: effect on emphysema quantification. Eur Radiol 26:478–486. https://doi.org/10.1007/s00330-015-3824-y
Giavarina D (2015) Understanding Bland Altman analysis. Biochem Med 25(2):141–151. https://doi.org/10.11613/BM.2015.015
Graham ML, Gresch SC, Hardy SK, Mutch LA, Janecek JL, Hegstad-Davies RL (2015) Evaluation of commercial ELISA and RIA for measuring porcine C-peptide: implications for research. Xenotransplantation 22(1):62–69. https://doi.org/10.1111/xen.12143
Goldman MB, Luchins DJ, Robertson GL (1988) Mechanisms of altered water metabolism in psychotic patients with polydipsia and hyponatremia. N Engl J Med 318(7):397–403. https://doi.org/10.1056/NEJM198802183180702
Hedrich CM, Zachurzok-Buczynska A, Gawlik A, Russ S, Hahn G, Koehler K, Malecka-Tendera E, Huebner A (2009) Autosomal dominant neurohypophyseal diabetes insipidus in two families. Molecular analysis of the vasopressin-neurophysin II gene and functional studies of three missense mutations. Horm Res Paediatr 71 (2):111–119. https://doi.org/10.1159/000183900
Husain MK, Fernando N, Shapiro M, Kagan A, Glick SM (1973) Radioimmunoassay of Arginine Vasopressin in Human Plasma. J Clin Endocrinol Metab 37(4):616–625. https://doi.org/10.1210/jcem-37-4-616
Ishikawa E (1987) Development and clinical application of sensitive enzyme immunoassay for macromolecular antigens–a review. Clin Biochem 20(6):375–385. https://doi.org/10.1016/0009-9120(87)90002-6
Kaur P, Stoltzfus JC (2017) Bland-Altman plot: A brief overview. Int J Acad Med 3(1):110–111. https://doi.org/10.4103/IJAM.IJAM_54_17
Kishimoto T, Hirai M, Ohsawa H, Terada M, Matsuoka I, Ikawa G (1989) Manners of arginine vasopressin secretion in schizophrenic patients–with reference to the mechanism of water intoxication. Jpn J Psychiat Neur 43(2):161–169. https://doi.org/10.1111/j.1440-1819.1989.tb02565.x
Koufaris C, Alexandrou A, Sismani C, Skordis N (2015) Identification of an AVP-NPII mutation within the AVP moiety in a family with neurohypophyseal diabetes insipidus: review of the literature. Hormones (Athens) 14 (3):442–446. https://doi.org/10.14310/horm.2002.1604
Marzocchi C, Cantara S, Sagnella A, Castagna MG, Capezzone M (2021) Autosomal dominant familial neurohypophyseal diabetes insipidus caused by a novel missense mutation in AVP gene in a large Italian kindred. Endocrine 74:188–192. https://doi.org/10.1007/s12020-021-02830-x
Mavani GP, DeVita MV, Michelis MF (2015) A review of the nonpressor and nonantidiuretic actions of the hormone vasopressin. Front Med (lausanne) 2:19. https://doi.org/10.3389/fmed.2015.00019
Moeller HB, Rittig S, Fenton RA (2013) Nephrogenic diabetes insipidus: essential insights into the molecular background and potential therapies for treatment. Endocr Rev 34(2):278–301. https://doi.org/10.1210/er.2012-1044
Niwa T, Katsuzaki T, Tatemichi N, Kato K, Yazawa T, Maeda K (1989) Enzyme immunoassay for urinary albumin at low concentration in diabetes mellitus. Clin Chim Acta 186(3):391–396. https://doi.org/10.1016/0009-8981(90)90325-M
Olofsen E, Dahan A, Borsboom G, Drummond G (2015) Improvements in the application and reporting of advanced Bland-Altman methods of comparison. J Clin Monit Comput 29:127–139. https://doi.org/10.1007/s10877-014-9577-3
Özcan Türkmen M, Karaduman T, Erdem Tunçdemir B, Ünal MA, Mergen H (2021) Functional analyses of three different mutations in the AVP-NPII gene causing familial neurohypophyseal diabetes insipidus. Endocrine 74(3):658–665. https://doi.org/10.1007/s12020-021-02803-0
Polat K, Nour M (2020) Epileptic seizure detection based on new hybrid models with electroencephalogram signals. IRBM 41:331–353. https://doi.org/10.1016/j.irbm.2020.06.008
Risch L, Hoefle G, Saely C, Berchthold S, Weber M, Gouya G, Rein P, Langer P, Marte T, Aczel S, Drexel H (2006) Evaluation of two fully automated novel enzyme-linked immunosorbent assays for the determination of human adiponectin in serum. Clin Chim Acta 373:121–126. https://doi.org/10.1016/j.cca.2006.05.017
Rutishauser J, Spiess M, Kopp P (2016) Genetic forms of neurohypophyseal diabetes insipidus. Best Pract Res Clin Endocrinol Metab 30(2):249–262. https://doi.org/10.1016/j.beem.2016.02.008
Sachidhanandam M, Singh SN, Salhan AK, Ray US (2010) Evaluation of plasma hormone concentrations using Enzyme-Immunoassay/Enzyme-linked Immunosorbent assay in healthy Indian men: Effect of ethnicity. Indian J Clin Biochem 25(2):153–157. https://doi.org/10.1007/s12291-010-0028-8
Sink TD, Lochmann RT, Fecteau KA (2008) Validation, use, and disadvantages of enzyme-linked immunosorbent assay kits for detection of cortisol in channel catfish, largemouth bass, red pacu, and golden shiners. Fish Physiol Biochem 34:95–101. https://doi.org/10.1007/s10695-007-9150-9
Tamaoki J, Isono K, Kondo M, Yamawaki I, Tagaya E, Nagai A (1998) A human bronchial epithelial cell line releases arginine vasopressin: involvement of Ca2+-activated K+ channels. Regul Pept 74:91–95. https://doi.org/10.1016/S0167-0115(98)00022-6
Tilbrook AJ, Clarke IJ (2006) Neuroendocrine mechanisms of innate states of attenuated responsiveness of the hypothalamo-pituitary adrenal axis to stress. Front Neuroendocrinol 27:285–307. https://doi.org/10.1016/j.yfrne.2006.06.002
van Stralen KJ, Jager KJ, Zoccali C, Dekker FW (2008) Agreement between methods. Kidney Int 74(9):1116–1120. https://doi.org/10.1038/ki.2008.306
Wahlstrom JT, Fowler MJ, Nicholson WE, Kovacs WJ (2004) A novel mutation in the preprovasopressin gene identified in a kindred with autosomal dominant neurohypophyseal diabetes insipidus. J Clin Endocrinol Metab 89(4):1963–1968. https://doi.org/10.1210/jc.2003-031542
Warnken T, Huber K, Feige K (2016) Comparison of three different methods for the quantification of equine insulin. BMC Vet Res 12(1):196. https://doi.org/10.1186/s12917-016-0828-z
Windpessl M, Schwarz C, Wallner M (2017) “Bowel prep hyponatremia” - a state of acute water intoxication facilitated by low dietary solute intake: case report and literature review. BMC Nephrol 18(54). https://doi.org/10.1186/s12882-017-0464-2
Zaki R, Bulgiba A, Ismail R, Ismail NA (2012) Statistical methods used to test for agreement of medical instruments measuring continuous variables in method comparison studies: a systematic review. PLoS ONE 7(5):e37908. https://doi.org/10.1371/journal.pone.0037908
Acknowledgements
We gratefully thank Dr. Muhittin SERDAR and his team for providing the gamma counter in their laboratory for the radioimmunoassay. This study is a part of the Ph. D. thesis of the first author and financially supported by the Scientific and Technological Research Council of Turkey (TUBITAK, Project number: 115S499).
Funding
This research was supported by the Scientific and Technological Research Council of Turkey (TUBİTAK, Project number:115S499).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Formal analysis and investigation were performed by Merve Özcan Türkmen and Tugce Karaduman. The first draft of the manuscript was written by Merve Özcan Türkmen and edited by Tugce Karaduman and Hatice Mergen. The funding acquisition was performed by Hatice Mergen (Supervisor).
Corresponding author
Ethics declarations
Ethics approval
No ethical approval is required, not applicable.
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Özcan Türkmen, M., Karaduman, T. & Mergen, H. Comparison of ELISA and RIA methods to quantify arginine vasopressin hormone levels in cell culture. Biologia 78, 887–894 (2023). https://doi.org/10.1007/s11756-022-01301-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11756-022-01301-6