Abstract
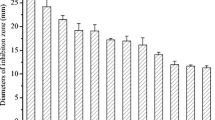
In this study, agroactive and bioactive compound production by actinobacteria isolated from rhizospheres of different plants was screened. Six of the forty-two isolates showed the highest production of different compounds and were identified by using molecular methods. In the primary and secondary screening antimicrobial activities were determined by inhibition zones against six bacterial and three fungal pathogens. Streptomyces labedae CYP25 showed the highest antibacterial activity in screening with extracts obtained from Actinomycetes Isolation Broth (AIB) against Staphylococcus aureus (18 mm); Streptomyces sp. CYP30 showed the highest antifungal activities in screening with ethyl acetate extracts obtained from Glucose Soybean Meal Broth against Candida albicans (60 mm) and Alternaria sp. D21 (52 mm) and showed the highest antifungal activities in screening with extracts obtained from International Streptomyces Project-3 (ISP-3) Broth against Aspergillus niger (16 mm). The highest indole acetic acid (IAA) production was showed by Nocardiopsis sp. CYP39. This isolate produced the highest amount of IAA (76.5 µg/mL) in the presence of 0.9% L-tryptophan. Thirty-two of forty-two isolates were identified as siderophore producers. The best hydrogen cyanide producer was Nocardiopsis sp. CYP15. Streptomyces moderatus CYP35 and Streptomyces chartreusis CYP19 constitute the highest halo-zone diameter on phosphate solubilizing activity tests.






Similar content being viewed by others
Abbreviations
- IAA:
-
Indole acetic acid
- AIA:
-
Actinomycetes Isolation Agar
- AIB:
-
Actinomycetes Isolation Broth
- GSB:
-
Glucose Soybean Meal Broth
- SCB:
-
Starch Casein Broth
- ISP:
-
International Streptomycetes Project
- CAS:
-
Chromium Azurol S
- HCN:
-
Hydrogen cyanide
- PVKA:
-
Pkiovskaya’s Agar
- PGPR:
-
Plant Growth Promoting Rhizobacteria
- BLAST:
-
Basic Local Alignment Search Tool
- NCBI:
-
National Center for Biotechnology Information
References
Abd-Alla MH, El-Sayed EA, Rasmey AM (2013) Indole-3-acetic acid (IAA) production by Streptomyces atrovirens isolated from rhizospheric soil in Egypt. J Biology Earth Sci 3:182–193
Adegboye M, Babalola O (2013) Actinomycetes: A Yet Inexhaustive Source of Bioactive Secondary Metabolites. In: Mendez-Vilas A (ed) Microbial Pathogens and Strategies for Combating Them, Science, Technology and Education. Formatex, Badajoz, pp 786–795
Ahemad M (2015) Phosphate-solubilizing bacteria-assisted phytoremediation of metalliferous soils: a review. Biotech 5:111–121. https://doi.org/10.1007/s13205-014-0206-0
Anwar S, Basharat A, Sajid I (2016) Screening of rhizospheric actinomycetes for various in vitro and in vivo plant growth promoting (PGP) traits and for agroactive compounds. Front Microbiol 7:1–11. https://doi.org/10.3389/fmicb.2016.01334
Aouar L, Lerat S, Boulahrouf A, Beaulieu C (2016) Bacterial antagonism and plant growth promoting traits of actinobacterial strains previously selected for their antifungal properties. Transylv Rev 24:2408–2417
Bhosale HJ, Kadam TA (2015) Generic diversity and a comparative account on plant growth promoting characteristics of actinomycetes in roots and rhizosphere of Saccharum officinarum. Int J Curr Microbiol Appl Sci 4:230–244
Boukhatem ZF, Merabet C, Tsaki H (2022) Plant Growth Promoting Actinobacteria, the Most Promising Candidates as Bioinoculants? Front. Agron 4:849911. https://doi.org/10.3389/fagro.2022.849911
Chandrakar S, Gupta AK (2017) Production and Characterization of Actinomycin D from Streptomyces parvulus Isolated from Aloe vera (l.) Burm. F. and its Antimicrobial Activity. International Journal of Pharmaceutical Sciences Review and Research 46: 169–175
Chaudhary HS, Yadav J, Shrivastava AR, Singh S, Singh AK, Gopalan N (2013) Antibacterial activity of actinomycetes isolated from different soil samples of Sheopur (A city of central India). J Adv Pharm Technol Res 4:118–123. https://doi.org/10.4103/2231-4040.111528
Damam M, Moinuddin MK, Kausar R (2016) Isolation and screening of plant growth promoting actinomycetes from rhizosphere of some forest medicinal plants. Int J ChemTech Res 9:521–528
Dastager SG, Damare S (2013) Marine Actinobacteria Showing Phosphate-Solubilizing Efficiency in Chorao Island, Goa, India. Curr Microbiol 66:421–427. https://doi.org/10.1007/s00284-012-0288-z
De Oliveira MF, Da Silva MG, Der Sand TV (2010) Antiphytopathogen potential of endophytic actinobacteria isolated from tomato plants (Lycopersicon esculentum) in southern Brazil, and characterization of Streptomyces sp. R18(6), a potential biocontrol agent. Res Microbiol 161:565–572. https://doi.org/10.1016/j.resmic.2010.05.008
De Souza R, Ambrosini A, Passaglia LMP (2015) Plant growth promoting bacteria as inoculants in agricultural soils. Genet Mol Biology 38:401–419. https://doi.org/10.1590/S1415-475738420150053
Duddu MK, Guntuku G (2016) Isolation, screening and characterization of antibiotic producing actinomycetes from Kapuluppada plastic waste dum** yard, Visakhapatnam. Int J Pharm Pharm Sci 8:221–229. https://doi.org/10.22159/ijpps.2016v8i11.10110
El-Tarabily KA (2008) Promotion of tomato (Lycopersicon esculentun Mill.) plant growth by rhizosphere competent 1-aminocyclopropane-1-carboxylic acid deaminase–producing Streptomycete actinomycetes. Plant Soil 308:161–174. https://doi.org/10.1007/s11104-008-9616-2
Farhat MF, Boukhris I, Chouayekh H (2014) Mineral phosphate solubilization by Streptomyces sp. CTM396 involves the excretion of gluconic acid and is stimulated by humic acids. FEMS Microbiol Lett 362:1–8. https://doi.org/10.1093/femsle/fnv008
Franco-Correa M, Quintana A, Duque C, Suarez C, Rodriguez MX, Barea JM (2010) Evaluation of actinomycete strains for key traits related with plant growth promotion and mycorrhiza hel** activities. Appl Soil Ecol 45:209–217. https://doi.org/10.1016/j.apsoil.2010.04.007
Frey B, Rieder SR, Brunner I, Plötze M, Koetzsch S, Lapanje A (2010) Weathering-associated bacteria from the Damma glacier forefield: physiological capabilities and impact on granite dissolution. Appl Environ Microbiol 76:4788–4796. https://doi.org/10.1128/AEM.00657-10
Ganesan P, Reegan AD, David RHA, Gandhi MR, Paulraj MG, Al- Dhabi N, Ignacimuthu S (2017) Antimicrobial activity of some actinomycetes from Western Ghats of Tamil Nadu, India. Alexandria J Med 53:101–110. https://doi.org/10.1016/j.ajme.2016.03.004
Gopalakrishnan S, Vadlamudi S, Bandikinda P, Sathya A, Vijayabharathi R, Rupela O, Kudapa H, Katta K, Varshney RK (2014) Evaluation of Streptomyces strains isolated from herbal vermicompost for their plant growth-promotion traits in rice. Microbiol Res 169:40–48. https://doi.org/10.1016/j.micres.2013.09.008
Gopinath BV, Vootla PK, Jyothi R, Redd KS (2013) Antimicrobial Activity of Actinomycetes Isolated From Coal Mine Soils of Godavari Belt Region, A.P, India. Asian J Exp Biol Sci 4:518–523
Guo S, Feng B, **ao C, Wang Q, Chi R (2021) Phosphate-solubilizing microorganisms to enhance phytoremediation of excess phosphorus pollution in phosphate mining wasteland soil. Bioremediat J 25:271–281. https://doi.org/10.1080/10889868.2021.1884528
Jobim ML, Santos RCV, Alves CFS, Oliveira RM, Mostardeiro CP, Sagrillo MR, Filho OCS, Garcia LFM, Manica-Cattani MF, Ribeiroe EE, Da Cruz IBM (2014) Antimicrobial activity of Amazon Astrocaryum aculeatum extracts and its association to oxidative metabolism. Microbiol Res 169:314–323. https://doi.org/10.1016/j.micres.2013.06.006
Kaur G (2014) Studies on microbial phosphate solubilization and development of inoculum formulations. Dissertation, University of Thapar
Kaur T, Sharma D, Kaur A, Manhas KR (2013) Antagonistic and plant growth promoting activities of endophytic and soil actinomycetes. Archives of Phytopathology and Plant Protection 46:1756–1768. https://doi.org/10.1080/03235408.2013.777169
Khamna S, Yokota A, Lumyong S (2009) Actinomycetes isolated from medicinal plant rhizosphere soils: diversity and screening of antifungal compounds, indole-3-acetic acid and siderophore production. World J Microbiol Biotechnol 25:649–655. https://doi.org/10.1007/s11274-008-9933-x
Koçyiğit A (2009) Termofilik Bakterilerden Lipaz Üretimi ve Karakterizasyonu. Dissertation, Ege University
Lakshmanan V, Shantharaj D, Li G, Seyfferth AL, Sherrier DJ, Bais HP (2015) A natural rice rhizospheric bacterium abates arsenic accumulation in rice (Oryza sativa L.). Planta 242:1037–1050. https://doi.org/10.1007/s00425-015-2340-2
Lapanje A, Wimmersberger C, Furrer G, Brunner I, Frey B (2012) Pattern of elemental release during the granite dissolution can be changed by aerobic heterotrophic bacterial strains isolated from Damma glacier (Central Alps) deglaciated granite sand. Microb Ecol 63:865–882. https://doi.org/10.1007/s00248-011-9976-7
Legault G, Lerat S, Nicolas P, Beaulieu C (2011) Tryptophan regulates thaxtomin A and indole-3-acetic acid production in Streptomyces scabiei and modifies its interactions with radish seedlings. Phytopathol 101:1045–1051. https://doi.org/10.1094/PHYTO-03-11-0064
Mitra D, Mondal R, Khoshru B, Senapati A, Radha TK, Mahakur B, Uniyal N, Myo EM, Boutaj H, Sierra BEG, Panneerselvam P, Ganeshamurthy AN, Elković SA, Vasić T, Rani A, Dutta S, Mohapatra PKD (2022) Actinobacteria-enhanced plant growth, nutrient acquisition, and crop protection: Advances in soil, plant, and microbial multifactorial interactions. Pedosphere 32:149–170. https://doi.org/10.1016/S1002-0160(21)60042-5
Mohandas S, Poovarasan S, Panneerselvam P, Saritha B, Upreti KK, Kamala R, Sita T (2013) Guava (Psidium guajava L.) rhizosphere Glomus mosseae spores harbor actinomycetes with growth promoting and antifungal attributes. Sci Hort 150:371–376. https://doi.org/10.1016/j.scienta.2012.11.019
Mohanraj D, Bharathi S, Radhakrishnan M, Balagurunathan R (2011) Bioprospecting of actinobacteria from Yelagiri hills with special reference to antibacterial activity. J Chem Pharm Res 3:439–446
Mohseni M, Norouzi H, Hamedi J, Roohi A (2013) Screening of Antibacterial Producing Actinomycetes from Sediments of the Caspian Sea. Int J Mol Cell Med 2:64–71
Namlı A, Mahmood A, Sevilir B, Özkır E (2017) Effect of phosphorus solubilizing bacteria on some soil properties, wheat yield and nutrient contents. Eurasian J Soil Sci 6:249–258. https://doi.org/10.18393/ejss.293157
Ng YZ, Amsaveni S (2012) Isolation, Screening and Characterization of Antibiotic-Producing Actinomycetes from Rhizosphere Region of Different Plants from a Farm of Sungai Ramal Luar, Malaysia. J Adv Biomedical Pathobiology 2:96–107
Ningthoujam DS, Chanu SB, Tamreihao K, Lynda R, Devi KA, Jeeniita N (2016) Plant Growth Promotion and Biocontrol Potential of a Streptomyces sp. strain N3-3b isolated from the rhizosphere of Chakhao, a Black Rice Variety of Manipur, India. Br Microbiol Res J 16:1–11
Passari AK, Mishra VK, Gupta VK, Yadav MK, Saikia R, Singh BP (2015) In Vitro and In Vivo Plant Growth Promoting Activities and DNA Fingerprinting of Antagonistic Endophytic Actinomycetes Associates with Medicinal Plants. PLoS ONE 10:1–18. https://doi.org/10.1371/journal.pone.0139468
Patel MV, Patel RK (2014) Indole-3-acetic acid (IAA) production by endophytic bacteria isolated from saline dessert, The Little Runn of Kutch. CIBTech J Microbiol 3:17–28
Rajan BM, Kannabiran K (2014) Extraction and Identification of Antibacterial Secondary Metabolites from Marine Streptomyces sp. VITBRK2. Int J Mol Cell Med 3:130–137
Rijavec T, Lapanje A (2016) Hydrogen Cyanide in the Rhizosphere: Not Suppressing Plant Pathogens, but Rather Regulating Availability of Phosphate. Front Microbiol 7:1785. https://doi.org/10.3389/fmicb.2016.01785
Rodrigues AA, Araujo MVF, Soares RS, De Oliveira BFR, Sibov TD, Vieira JDG (2018) Isolation and Screening for Multi-trait Plant Growth Promoting Actinobacteria From Organic Sugarcane Rhizosphere. Int J Microbiol Res 10:1193–1198. https://doi.org/10.4161/cib.27683
Singh LS, Sharma H, Talukdar NC (2014) Production of potent antimicrobial agent by actinomycete, Streptomyces sannanensis strain SU118 isolated from phoomdi in Loktak Lake of Manipur, India. BMC Microbiol 14:1–13. https://doi.org/10.1186/s12866-014-0278-3
Singh N, Rai V (2012) Optimization of cultural parameters for antifungal and antibacterial metabolite from microbial isolate; Streptomyces rimosus MTCC 10792 from soil of Chhattisgarh. Int J Pharm Pharm Sci 4:94–101
Sreevidya M, Gopalakrishnan S, Kudapa H, Varshney RK (2016) Exploring plant growth-promotion actinomycetes from vermicompost and rhizosphere soil for yield enhancement in chickpea. Brazilian J Microbiol 47:85–95. https://doi.org/10.1016/j.bjm.2015.11.030
Sudha SSK, Hemalatha R (2015) Isolation and screening of antibiotic producing actinomycetes from garden soil of Sathyabama University, Chennai. Asian J Pharm Clin Res 8:1210–1140
Tamura K, Dudley J, Nei M, Kumar S (2007) MEGA4: Molecular Evolutionary Genetics Analysis (MEGA) software version 4,0. Mol Biol Evol 24:1596–1599. https://doi.org/10.1093/molbev/msm092
Toker SK, Evlat H, Koçyi̇ği̇t A (2021) Screening of newly isolated marine-derived fungi for their laccase production and decolorization of different dye types. Reg Stud Mar Sci 45:1–12. https://doi.org/10.1016/j.rsma.2021.101837
Varalakshmi T, Sekhar KM, Charyulu PBB (2014) Taxonomic studies and phylogenetic characterization of potential and pigmented antibiotic producing actinomycetes isolated from rhizosphere soils. Int J Pharm Pharm Sci 6:511–519
Vurukonda SSKP, Giovanardi D, Stefani E (2018) Plant Growth Promoting and Biocontrol Activity of Streptomyces spp. as Endophytes. Int J Mol Sci 19:1–26. https://doi.org/10.3390/ijms19040952
Wang W, Qiu Z, Tan H, Cao L (2014) Siderophore production by actinobacteria. Biometals 27:623–631. https://doi.org/10.1007/s10534-014-9739-2
Wani AP, Irene OI (2014) Screening of microbes for their metal, antibiotic resistance and plant growth promoting activity. Currrent Res Bacteriol 7:22–31. https://doi.org/10.3923/pjbs.2014.206.212
Wongfun N, Plötze M, Furrer G, Brand H (2014) Weathering of granite from the Damma glacier area: the contribution of cyanogenic bacteria. Geomicrobiol J 31:93–10. https://doi.org/10.1080/01490451.2013.802396
Author information
Authors and Affiliations
Contributions
This study was performed based on a Master of Science (MSc) thesis addressed to Hüseyin EVLAT. All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Hüseyin EVLAT, Sultan Kübra TOKER and Ali KOÇYİĞİT. Ali KOÇYİĞİT supervised the study.The first draft of the manuscript was written by Hüseyin EVLAT and all authors commented and make corrections on all versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Statements and declarations
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Evlat, H., Toker, S.K. & Koçyiğit, A. Screening for agroactive and bioactive metabolites production by actinobacteria isolated from rhizospheric soils. Biologia 78, 187–200 (2023). https://doi.org/10.1007/s11756-022-01226-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11756-022-01226-0