Abstract
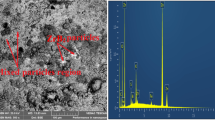
Crude oil storage tanks usually undergo corrosion problems due to the activity of sulfur compounds, water content, and salts in crude oil. In this study, the corrosion resistance of carbon steel A285 grade C was improved by adding SiO2 nanoparticles (NPs) as a corrosion inhibitor. The SiO2 NPs were extracted from sand (from the city of Al-Ramadi in western Iraq) using a combined physical and chemical method. The present work investigated the effect of various additions of SiO2 NPs as a corrosion inhibitor (i.e., 0.2, 0.4, 0.6, 0.8, and 1 g/L) on the corrosion resistance of carbon steel A285 grade C. The corrosion rate was evaluated by applying potentiostatic technique under a 1 M concentration of sulfuric acid as the corrosion medium. The results indicated the formation of a thin film of SiO2 NPs adhering to the surface of the carbon steel and working as a corrosion inhibitor. This film will protect the metal from corrosion by preventing any corrosion activity. Moreover, the results showed that the corrosion rate reduced greatly by increasing the amount of nano-addition. Furthermore, it was found that the corrosion resistance was enhanced dramatically with increasing nano-additions. An optimal corrosion inhibition of more than a 91.6% was achieved with a nano-addition of 1 g/L for the immersion time of 10 h. Additionally, the corrosion inhibition in this process with the eco-friendly material showed a high protection rate and low operating cost.









Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Abd E-R, Khamis EA, Abou Kana MT, Negm NA (2018) Electrochemical and quantum chemical evaluation of new bis (coumarins) derivatives as corrosion inhibitors for carbon steel corrosion in 0.5 M H2SO4. J Mol Liq 255:341–353. https://doi.org/10.1016/j.molliq.2018.01.148
Ajeel SA, Sukkar KA, Zedin NK (2021) New magnesio-thermal reduction technique to produce high-purity crystalline nano-silicon via semi-batch reactor. Mater Today Proc 42:1966–1972. https://doi.org/10.1016/j.matpr.2020.12.243
Alao AO, Popoola AP, Sanni O (2022) The influence of nanoparticle inhibitors on the corrosion protection of some industrial metals: a review. J Bio Tribo-Corros 8:1–16. https://doi.org/10.1007/s40735-022-00665-1
Alvarez PE, Fiori-Bimbi MV, Neske A, Brandan SA, Gervasi CA (2018) Rollinia occidentalis extract as green corrosion inhibitor for carbon steel in HCl solution. J Ind Eng Chem 58:92–99. https://doi.org/10.1016/j.jiec.2017.09.012
American Petroleum Institute (2012) Welded steel tanks for oil storage standard edition section 650 (API 650) 11th. https://law.resource.org/pub/us/cfr/ibr/002/api.650.2007.pdf
Asaad MA, Sarbini NN, Sulaiman A, Ismail M, Huseien GF, Majid ZA, Raja PB (2018) Improved corrosion resistance of mild steel against acid activation: impact of novel Elaeis guineensis and silver nanoparticles. J Ind Eng Chem 63:139–148. https://doi.org/10.1016/j.jiec.2018.02.010
Asaad MA, Raja PB, Huseien GF, Fediuk R, Ismail M, Alyousef R (2021) Self-healing epoxy coating doped with Elaesis guineensis/silver nanoparticles: a robust corrosion inhibitor. Constr Build Mater 312:125396. https://doi.org/10.1016/j.conbuildmat.2021.125396
Aslam R, Mobin M, Aslam J (2022) Corrosion inhibitors for refinery industries. In: Environmentally sustainable corrosion inhibitors, pp 385–404. https://doi.org/10.1016/B978-0-323-85405-4.00004-5
ASTM Standard, C146-21 (2021) Standard test methods for chemical analysis of glass sand. ASTM International, West Conshohocken 1–16.
Awad AM, Sukkar KA, Jaed DM (2022) Development of an extremely efficient Iraqi nano-lubricating oil (base-60) employing SiO2 and Al2O3 nanoparticles. AIP Conf Proc 2443(1):030049. https://doi.org/10.1063/5.0091951
Azzam E, Sami RM, Alenezi KM, El Moll H, Haque A (2021) Nanoparticles modified with cationic thiol surfactant as efficient inhibitors for the corrosion of carbon steel. J Electrochem Sci Technol 12(3):308–316. https://doi.org/10.33961/jecst.2021.00010
Basik M, Mobin M (2022) 20-Environmentally sustainable corrosion inhibitors in the oil and gas industry. In: Environmentally sustainable corrosion inhibitors. Elsevier, pp 405–421. https://doi.org/10.1016/B978-0-323-85405-4.00017-3
Benmahammed I, Douadi T, Issaadi S, Al-Noaimi M, Chafaa S (2019) Heterocyclic Schiff bases as corrosion inhibitors for carbon steel in 1 M HCl solution: hydrodynamic and synergetic effect. J Dispers Sci Technol. https://doi.org/10.1080/01932691.2019.1614038
Bharatiya U, Gal P, Agrawal A, Shah M, Sircar A (2019) Effect of corrosion on crude oil and natural gas pipeline with emphasis on prevention by ecofriendly corrosion inhibitors: a comprehensive review. J Bio Tribo-Corros 5(2):1–12. https://doi.org/10.1007/s40735-019-0225-9
El Refay HM, Hyba AM, Gaber GA (2022) Fabrication, characterization and corrosion feature evaluation of mild steel in 1 M HCl by nanoparticle-modified activated carbon. Chem Pap 76(2):813–825. https://doi.org/10.1007/s11696-021-01895-2
Falcón JM, Batista FF, Aoki IV (2014) Encapsulation of dodecylamine corrosion inhibitor on silica nanoparticles. Electrochim Acta 124:109–118. https://doi.org/10.1016/j.electacta.2013.06.114
Fandi Z, Ameur N, Brahimi FT, Bedrane S, Bachir R (2020) Photocatalytic and corrosion inhibitor performances of CeO2 nanoparticles decorated by noble metals: Au, Ag. Pt J Environ Chem Eng 8(5):104346. https://doi.org/10.1016/j.jece.2020.104346
Feng Y, Cheng YF (2017) An intelligent coating doped with inhibitor-encapsulated nanocontainers for corrosion protection of pipeline steel. Chem Eng J 315:537–551. https://doi.org/10.1016/j.cej.2017.01.064
Finšgar M, Jackson J (2014) Application of corrosion inhibitors for steels in acidic media for the oil and gas industry: a review. Corros Sci 86:17–41. https://doi.org/10.1016/j.corsci.2014.04.044
Hadi BA, Ibraheem HH (2022) Synthesis, Characterization and Corrosion Inhibition of Novel Pyridine on Mild Steel in Hydrochloric Acid Environment. J Appl Sci Nanotech 2(2):55–63. https://doi.org/10.53293/jasn.2021.3862.1050
Hameed WF, Rashid KH, Khadom AA (2022) Investigation of tetraazaadamantane as corrosion inhibitor for mild steel in oilfield produced water under sweet corrosive environment. J Bio Tribo Corros 8:27. https://doi.org/10.1007/s40735-021-00626-0
Hoai NT, Van Hien P, Vu NSH, Son DL, Van Man T, Tri MD, Nam ND (2019) An improved corrosion resistance of steel in hydrochloric acid solution using Hibiscus sabdariffa leaf extract. Chem Pap 73(4):909–925. https://doi.org/10.1007/s11696-018-0649-6
Ji G, Anjum S, Sundaram S, Prakash R (2015) Musa paradisica peel extract as green corrosion inhibitor for mild steel in HCl solution. Corros Sci 90:107–117. https://doi.org/10.1016/j.corsci.2014.10.002
Kamburova K, Boshkova N, Boshkov N, Radeva T (2021) Composite coatings with polymeric modified ZnO nanoparticles and nanocontainers with inhibitor for corrosion protection of low carbon steel. Colloids Surf A 609:125741. https://doi.org/10.1016/j.colsurfa.2020.125741
Khadar YS, Surendhiran S, Gowthambabu V, Banu SHAS, Devabharathi V, Balamurugan A (2021) Enhancement of corrosion inhibition of mild steel in acidic media by green-synthesized nano-manganese oxide. Mater Today Proc 47:889–893. https://doi.org/10.1016/j.matpr.2021.04.335
Kim SW, Park SY, Roh CH, Shim JH, Kim SB (2022) Effect of corrosion inhibitors on SA106 grade B carbon steel in H2SO4-N2H4 solution for the hydrazine-based reductive metal ion decontamination process. Chem Pap. https://doi.org/10.1007/s11696-022-02338-2
Liang T, Yuan H, Li C, Dong S, Zhang C, Cao G, Cao X (2021) Corrosion inhibition effect of nano–SiO2 for galvanized steel superhydrophobic surface. Surf Coat Technol 406:126673. https://doi.org/10.1016/j.surfcoat.2020.126673
Ma L, Wang J, Zhang D, Huang Y, Huang L, Wang P, Mol JM (2021) Dual-action self-healing protective coatings with photothermal responsive corrosion inhibitor nanocontainers. Chem Eng J 404:127118. https://doi.org/10.1016/j.cej.2020.127118
Manssouri M, Znini M, Lakbaibi Z, Ansari A, El Ouadi Y (2021) Experimental and computational studies of perillaldehyde isolated from Ammodaucus leucotrichus essential oil as a green corrosion inhibitor for mild steel in 1.0 M HCl. Chem Pap 75(3):1103–1114. https://doi.org/10.1007/s11696-020-01353-5
Masroor S, Mobin M, Singh AK, Rao RAK, Shoeb M, Alam MJ (2020) Aspartic di-dodecyl ester hydrochloride acid and its ZnO-NPs derivative, as ingenious green corrosion defiance for carbon steel through theoretical and experimental access. SN Appl Sci 2(2):1–16. https://doi.org/10.1007/s42452-019-1515-z
Momoh A, Adams FV, Samuel O, Bolade OP, Olubambi PA (2022) Corrosion prevention: The use of nanomaterials. In: Modified nanomaterials for environmental applications. Part of the Engineering Materials book series, Springer, pp 91–105. https://doi.org/10.1007/978-3-030-85555-0_5
Pasley H, Clark C (2000) Computational fluid dynamics study of flow around floating-roof oil storage tanks. J Wind Eng Ind Aerodyn 86(1):37–54. https://doi.org/10.1016/j.jpcs.2021.110341
Peter A, Obot IB, Sharma SK (2015) Use of natural gums as green corrosion inhibitors: an overview. Int J Ind Chem 6(3):153–164. https://doi.org/10.1007/s40090-015-0040-1
Pramudita M, Sukirno S, Nasikin M (2019) Synergistic corrosion inhibition effect of rice husk extract and KI for mild steel in H2SO4 solution. Bull Chem React Eng Catal 14(3):697–704. https://doi.org/10.9767/bcrec.14.3.4249.697-704
Quraishi MA, Chauhan DS (2022) Recent trends in the development of corrosion inhibitors. In: A treatise on corrosion science, engineering and technology. Part of the Indian Institute of Metals Series book series, Springer, pp 783–799. https://doi.org/10.1007/978-981-16-9302-1_40
Rizlan Z, Mamat O (2014) Process parameters optimization of silica sand nanoparticles production using low speed ball milling method. Chin J Eng. https://doi.org/10.1155/2014/802459
Saxena A, Prasad D, Haldhar R, Singh G, Kumar A (2018) Use of Saraca ashoka extract as green corrosion inhibitor for mild steel in 0.5 M H2SO4. J Mol Liq 258:89–97. https://doi.org/10.1016/j.molliq.2018.02.104
Singh A, Ansari KR, Chauhan DS, Quraishi MA, Lgaz H, Chung IM (2020) Comprehensive investigation of steel corrosion inhibition at macro/micro level by ecofriendly green corrosion inhibitor in 15% HCl medium. J Colloid Interface Sci 560:225–236. https://doi.org/10.1016/j.jcis.2019.10.040
Solomon MM, Umoren SA (2016) In-situ preparation, characterization and anticorrosion property of polypropylene glycol/silver nanoparticles composite for mild steel corrosion in acid solution. J Colloid Interface Sci 462:29–41. https://doi.org/10.1016/j.jcis.2015.09.057
Solomon MM, Gerengi H, Umoren SA, Essien NB, Essien UB, Kaya E (2018) Gum Arabic-silver nanoparticles composite as a green anticorrosive formulation for steel corrosion in strong acid media. Carbohydr Polym 181:43–55. https://doi.org/10.1016/j.carbpol.2017.10.051
Son GC, Hwang DK, Jang J, Chee SS, Cho K, Myoung JM, Ham MH (2019) Solution-processed highly adhesive graphene coatings for corrosion inhibition of metals. Nano Res 12:19–23. https://doi.org/10.1007/s12274-018-2056-2
Suganya M, Preethi PS, Narenkumar J, Prakash AA, Devanesan S, AlSalhi MS, Rajasekar A, Nanthini AUR (2022) Synthesis of silver nanoparticles from Indian red yeast rice and its inhibition of biofilm in copper metal in cooling water environment. Environ Sci Pollut Res. https://doi.org/10.21203/rs.3.rs-1179555/v1
Sukkar KA, Mahmood HY, Makilf WK (2019) Heat transfer performance improving for brass tubes heat exchanger coated by nanoparticles. Int Rev Mech Eng 13:597–607. https://doi.org/10.15866/ireme.v13i10.17579
Taheri M, Naderi R, Saremi M, Mahdavian M (2017) Development of an ecofriendly silane sol-gel coating with zinc acetylacetonate corrosion inhibitor for active protection of mild steel in sodium chloride solution. J Sol-Gel Sci Technol 81(1):154–166. https://doi.org/10.1007/s10971-016-4180-3
Tamalmani K, Husin H (2020) Review on corrosion inhibitors for oil and gas corrosion issues. Appl Sci 10(10):3389. https://doi.org/10.3390/app10103389
Umoren SA, Madhankumar A (2016) Effect of addition of CeO2 nanoparticles to pectin as inhibitor of X60 steel corrosion in HCl medium. J Mol Liq 224:72–82. https://doi.org/10.1016/j.molliq.2016.09.082
Zea C, Barranco-García R, Alcántara J, Simancas J, Morcillo M, De La Fuente D (2018) pH-dependent release of environmentally friendly corrosion inhibitor from mesoporous silica nanoreservoirs. Microporous Mesoporous Mater 255:166–173. https://doi.org/10.1016/j.micromeso.2017.07.035
Zedin NK, Ajeel SA, Sukkar KA (2020) Nanosilicon powder extraction as a sustainable source (from Iraqi rice husks) by hydrothermal process. AIP Conf Proc 2213:020155. https://doi.org/10.1063/5.0000147
Zhang Y, Liu X, Lai J, Wei Y, Luo J (2021) Corrosion fatigue life prediction of crude oil storage tank via improved equivalent initial flaw size. Theor Appl Fract Mech 114:103023. https://doi.org/10.1016/j.tafmec.2021.103023
Zhu M, He Z, Guo L, Zhang R, Anadebe VC, Obot IB, Zheng X (2021) Corrosion inhibition of eco-friendly nitrogen-doped carbon dots for carbon steel in acidic media: performance and mechanism investigation. J Mol Liq 342:117583. https://doi.org/10.1016/j.molliq.2021.117583
Zhuang W, Wang X, Zhu W, Zhang Y, Sun D, Zhang R, Wu C (2021) Imidazoline Gemini surfactants as corrosion inhibitors for carbon steel X70 in NaCl solution. ACS Omega 6(8):5653–5660. https://doi.org/10.1021/acsomega.0c06103
Acknowledgements
The authors are thankful to the staff of the Design and Industrial Production Research Unit/Department of Chemical Engineering/University of Technology- Iraq for their scientific support of this project.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest or non-financial interest in the subject matter or materials discussed in this manuscript. All authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Abdulhussein, B.A., Ali, A.M. & Sukkar, K.A. Reducing corrosion attacks on carbon steel A285 grade C in petroleum storage tanks by forming a thin film of nano-SiO2 from sand. Chem. Pap. 77, 1533–1543 (2023). https://doi.org/10.1007/s11696-022-02571-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-022-02571-9