Abstract
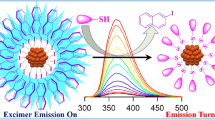
Cyanide anion sensing using gold-aryl nanoparticles bioconjugated with lysozyme is reported. The chemical dissolution of lysozyme capped gold nanoparticles (Lys-AuNPs) in the presence and absence of aryl shell was studied in cyanide (CN−) solutions. It was found that gold nanoparticles functionalized with an aryl shell exhibited different dissolution and luminescence behavior compared to gold nanoparticles without an aryl shell. Here, we show the fluorescence enhancement upon the interaction of Lys-AuNPs-COOH with CN− as a function of CN− ion concentration as linear with an excellent correlation coefficient R2 value of 0.9906 in the range from 1 × 10–3 mM to 1.2 × 10–2 mM. This study furthermore argues that the emission enhancement that occurs on gold dissolution is contributed from the complex from gold cyanidation rather than lysozyme. The emission intensity was enhanced accompanied with a blueshift upon CN− reaction with the gold core which originated from [Au(CN)2]− and characterized by CN− stretching frequency at 2175 cm−1 and 2126 cm−1 using Raman spectroscopy. Fluorescence lifetime decay analysis showed a shortened lysozyme lifetime upon nanoparticles formation and a significant increase after gold dissolution with KCN indicating emission enhancement.








Similar content being viewed by others
References
Ahmad AAL, Parambath JBM, Postnikov PS, Guselnikova O, Chehimi MM, Bruce MRM, Bruce AE, Mohamed AA (2021) Conceptual developments of aryldiazonium salts as modifiers for gold colloids and surfaces. Langmuir 37(30):8897–8907
Cheng C, Chen HY, Wu CS, Meena JS, Simon T, Ko FH (2016) A highly sensitive and selective cyanide detection using a gold nanoparticle-based dual fluorescence–colorimetric sensor with a wide concentration range. Sens Actuators B 227:283–290
Gale PA (2001) Anion receptor chemistry: highlights from 1999. Coord Chem Rev 213(1):79–128
Hameed M, Panicker S, Abdallah SH, Khan AA, Han C, Chehimi MM, Mohamed AA (2020) Protein-coated aryl modified gold nanoparticles for cellular uptake study by osteosarcoma cancer cells. Langmuir 36(40):11765–11775
Hernández Y, Coello Y, Fratila RM, Jesús M, Lionberger TA (2017) Highly sensitive ratiometric quantification of cyanide in water with gold nanoparticles via resonance Rayleigh scattering. Talanta 167:51–58
Jeffrey MI, Ritchie IM (2000) The leaching of gold in cyanide solutions in the presence of impurities II the effect of silver. J Electrochem Soc 147(9):3272–3276
**dra T, Prokop Z, Šnita D (2012) Etching and recovery of gold from aluminum substrate in thiourea solution. Chem Pap 66(6):617–620
Li X, Patterson HH (2013) A review of luminescent anionic nano system: D10 metallocyanide excimers and exciplexes in alkali halide hosts. Materials 6(7):2595–2611
Lu D, Liu L, Fengxia LF, Shuang S, Li Y, Choi MMF, Dong C (2014) Lysozyme-stabilized gold nanoclusters as a novel fluorescence probe for cyanide recognition. Spectrochim Acta A 121:77–80
McCarthy AJ, Coleman RG, Nicol MJ (1998) The mechanism of the oxidative dissolution of colloidal gold in cyanide media. J Electrochem Soc 145(2):408–414
McGivney E, Gao X, Liu Y, Lowry GV, Casman E, Gregory KB, VanBriesen JM, Avellan A (2018) Biogenic cyanide production promotes dissolution of gold nanoparticles in soil. Env Sci Technol 53(3):1287–1295
Muravyov M (2019) Two-step processing of refractory gold-containing sulfidic concentrate via biooxidation at two temperatures. Chem Pap 73(1):173–183
Shadman S, Massinaei M, Moghaddam AZ (2021) Removal of cyanide from the gold cyanidation plant tailings using graphene-based magnetic nanocomposites. Chem Pap 75(10):5543–5560
Sitaula S, Mackiewicz MR, Reed SM (2008) Gold nanoparticles become stable to cyanide etch when coated with hybrid lipid bilayers. Chem Commun 26:3013–3015
Wadsworth ME, Zhu X, Thompson JS, Pereira CJ (2000) Gold dissolution and activation in cyanide solution: kinetics and mechanism. Hydrometallurgy 57(1):1–11
Wang J, Qiu Y, Li D, Liu X, Jiang C, Huang L, Wen H, Hu J (2019) Ratiometric fluorometric and visual determination of cyanide based on the use of carbon dots and gold nanoclusters. Microchim Acta 186(12):1–9
Wei SC, Hsu PH, Lee YF, Lin YW, Huang CC (2012) Selective detection of iodide and cyanide anions using gold-nanoparticle-based fluorescent probes. ACS Appl Mater Interf 4(5):2652–2658
Yang H, Yang Y, Liu S, Zhan X, Zhou H, Li X, Yuan Z (2020) Ratiometric and sensitive cyanide sensing using dual-emissive gold nanoclusters. Anal Bioanal Chem 412(23):5819–5826
Acknowledgements
AAM acknowledges support from the University of Sharjah with competitive grants 160-2142-029-P and 150-2142-017-P.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
There are no conflicts to declare.
Human and animal rights
This research does not involve human participants and/or animals.
Informed consent
There is no need for informed consent.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Parambath, J.B.M., Hameed, M. & Mohamed, A.A. Potential sensing of cyanide anion using fluorescent lysozyme gold-aryl bioconjugates. Chem. Pap. 76, 3619–3626 (2022). https://doi.org/10.1007/s11696-022-02117-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-022-02117-z