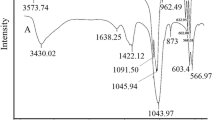
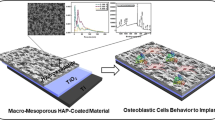
Abstract
Hydroxyapatite (HA) has been widely used for coating metal bone implants. However, HA coating may separate from the metal substrates, resulting in poor performance such as inflammatory reactions which may finally lead to implant failure. Dispersing HA into metal substrate may be a viable solution but such a composite may suffer from inferior mechanical strength. Therefore, a functionally graded material (FGM) with HA dispersing in metal substrate in gradients is introduced to increase surface osteogenic capability and simultaneously maintain a good mechanical strength of the bone implant. In this study, Ti6Al4V(Ti64)-HA composite with different component of HA and their corresponding FGMs were fabricated by selective laser melting (SLM). It was demonstrated that compared with the pure Ti64 and Ti64-2.5%HA composite, Ti64-1%HA composite seemed to have a better osteogenic promoting property in vitro. Both the FGMs significantly promoted the osteogenic activities of the pure Ti64 in vivo. Moreover, functionally graded structure improved the anti-compression properties of composites.





Similar content being viewed by others
References
G.M. Peñarrieta-Juanito, M. Costa, M. Cruz, G. Miranda, B. Henriques, J. Marques, R. Magini, A. Mata, J. Caramês, F. Silva, and J.C.M. Souza, Bioactivity of Novel Functionally Structured Titanium-Ceramic Composites in Contact with Human Osteoblasts, J. Biomed. Mater. Res. A, 2018, 106(7), p 1923–1931. https://doi.org/10.1002/jbm.a.36394
A. Civantos, E. Martínez-Campos, V. Ramos, C. Elvira, A. Gallardo, and A. Abarrategi, Titanium Coatings and Surface Modifications: Toward Clinically Useful Bioactive Implants, ACS Biomater. Sci. Eng., 2017, 3(7), p 1245–1261. https://doi.org/10.1021/acsbiomaterials.6b00604
S. Kyrylenko, M. Sowa, A. Kazek-Kęsik, A. Stolarczyk, M. Pisarek, Y. Husak, V. Korniienko, V. Deineka, R. Moskalenko, I. Matuła, J. Michalska, A. Jakóbik-Kolon, O. Mishchenko, M. Pogorielov, and W. Simka, Nitrilotriacetic acid Improves Plasma Electrolytic Oxidation of Titanium for Biomedical Applications, ACS Appl. Mater. Interfaces, 2023, 15(16), p 19863–19876. https://doi.org/10.1021/acsami.3c00170
S.E. El-Habashy, H.M. Eltaher, A. Gaballah, E.I. Zaki, R.A. Mehanna, and A.H. El-Kamel, Hybrid Bioactive Hydroxyapatite/Polycaprolactone Nanoparticles for Enhanced Osteogenesis, Mater. Sci. Eng. C Mater. Biol. Appl., 2021, 119, p 111599. https://doi.org/10.1016/j.msec.2020.111599
K. Yamada, K. Imamura, H. Itoh, H. Iwata, and S. Maruno, Bone Bonding Behavior of the Hydroxyapatite Containing Glass-Titanium Composite Prepared by the Cullet Method, Biomaterials, 2001, 22(16), p 2207–2214. https://doi.org/10.1016/s0142-9612(00)00402-6
D. Arcos and M. Vallet-Regí, Substituted Hydroxyapatite Coatings of Bone Implants, J. Mater. Chem. B, 2020, 8(9), p 1781–1800. https://doi.org/10.1039/c9tb02710f
E. Yılmaz, B. Çakıroğlu, A. Gökçe, F. Findik, H.O. Gulsoy, N. Gulsoy, Ö. Mutlu, and M. Özacar, Novel Hydroxyapatite/Graphene Oxide/Collagen Bioactive Composite Coating on Ti16Nb Alloys by Electrodeposition, Mater. Sci. Eng. C Mater. Biol. Appl., 2019, 101, p 292–305. https://doi.org/10.1016/j.msec.2019.03.078
Y. Wu, H. Tang, L. Liu, Q. He, L. Zhao, Z. Huang, J. Yang, C. Cao, J. Chen, and A. Wang, Biomimetic Titanium Implant Coated with Extracellular Matrix Enhances and Accelerates Osteogenesis, Nanomedicine, 2020, 15(18), p 1779–1793. https://doi.org/10.2217/nnm-2020-0047
A. Naderi, B. Zhang, J.A. Belgodere, K. Sunder, and G. Palardy, Improved Biocompatible, Flexible Mesh Composites for Implant Applications Via Hydroxyapatite Coating with Potential for 3-Dimensional Extracellular Matrix Network and Bone Regeneration, ACS Appl. Mater. Interfaces, 2021, 13(23), p 26824–26840. https://doi.org/10.1021/acsami.1c09034
W. Harun, R. Asri, J. Alias, F. Zulkifli, K. Kadirgama, S. Ghani, and J. Shariffuddin, A Comprehensive Review of Hydroxyapatite-Based Coatings Adhesion on Metallic Biomaterials, Ceram. Int., 2018, 44(2), p 1250–1268. https://doi.org/10.1016/j.ceramint.2017.10.162
J. Guillem-Marti, N. Cinca, M. Punset, I.G. Cano, F.J. Gil, J.M. Guilemany, and S. Dosta, Porous Titanium-Hydroxyapatite Composite Coating Obtained on Titanium by Cold Gas Spray with High Bond Strength for Biomedical Applications, Colloids Surf. B Biointerfaces, 2019, 180, p 245–253. https://doi.org/10.1016/j.colsurfb.2019.04.048
D. Mondal, L. Nguyen, I.H. Oh, and B.T. Lee, Microstructure and Biocompatibility of Composite Biomaterials Fabricated from Titanium and Tricalcium Phosphate by Spark Plasma Sintering, J. Biomed. Mater. Res. A, 2013, 101(5), p 1489–1501. https://doi.org/10.1002/jbm.a.34455
J. Yang and H.J. **ang, A Three-Dimensional Finite Element Study on the Biomechanical Behavior of an FGBM Dental Implant in Surrounding Bone, J. Biomech., 2007, 40(11), p 2377–2385. https://doi.org/10.1016/j.jbiomech.2006.11.019
C. Han, Q. Wang, B. Song, W. Li, Q. Wei, S. Wen, J. Liu, and Y. Shi, Microstructure and Property Evolutions of Titanium/Nano-Hydroxyapatite Composites In-Situ Prepared by Selective Laser Melting, J. Mech. Behav. Biomed. Mater., 2017, 71, p 85–94. https://doi.org/10.1016/j.jmbbm.2017.02.021
M.J. Ansari, D.S. Nguyen, and H.S. Park, Investigation of SLM Process in Terms of Temperature Distribution and Melting Pool Size: Modeling and Experimental Approaches, Materials, 2019 https://doi.org/10.3390/ma12081272
X. Yan, J. Pang, and Y. **g, Ultrasonic Measurement of Stress in SLM 316L Stainless Steel Forming Parts Manufactured Using Different Scanning Strategies, Materials, 2019 https://doi.org/10.3390/ma12172719
S. Huang, S.L. Sing, G. de Looze, R. Wilson, and W.Y. Yeong, Laser powder Bed Fusion of Titanium-Tantalum Alloys: Compositions and Designs for Biomedical Applications, J. Mech. Behav. Biomed. Mater., 2020, 108, p 103775. https://doi.org/10.1016/j.jmbbm.2020.103775
C.L. Chu, X.Y. Xue, J.C. Zhu, and Z.D. Yin, In Vivo Study on Biocompatibility and Bonding Strength of Hydroxyapatite-20vol%Ti Composite with Bone Tissues in the Rabbit, Biomed. Mater. Eng., 2006, 16(3), p 203–213.
T. Yi, C. Zhou, L. Ma, L. Wu, X. Xu, L. Gu, Y. Fan, G. **an, H. Fan, and X. Zhang, Direct 3-D Printing of Ti-6Al-4V/HA Composite Porous Scaffolds for Customized Mechanical Properties and Biological Functions, J. Tissue Eng. Regen. Med., 2020, 14(3), p 486–496. https://doi.org/10.1002/term.3013
D. Bovand, M.R. Allazadeh, S. Rasouli, E. Khodadad, and E. Borhani, Studying the Effect of Hydroxyapatite Particles in Osteoconductivity of Ti-HA Bioceramic, J. Aust. Ceram. Soc., 2019, 55(2), p 395–403. https://doi.org/10.1007/s41779-018-0247-7
J.D. Avila, K. Stenberg, S. Bose, and A. Bandyopadhyay, Hydroxyapatite Reinforced Ti6Al4V Composites for Load-Bearing Implants, Acta Biomater., 2021, 123, p 379–392. https://doi.org/10.1016/j.actbio.2020.12.060
A. Kumar, S. Dhara, K. Biswas, and B. Basu, In Vitro Bioactivity and Cytocompatibility Properties of Spark Plasma Sintered HA-Ti Composites, J. Biomed. Mater. Res. B Appl. Biomater., 2013, 101(2), p 223–236. https://doi.org/10.1002/jbm.b.32829
M. Shbeh, Z.J. Wally, M. Elbadawi, M. Mosalagae, H. Al-Alak, G.C. Reilly, and R. Goodall, Incorporation of HA into Porous Titanium to Form Ti-HA Biocomposite Foams, J. Mech. Behav. Biomed. Mater., 2019, 96, p 193–203. https://doi.org/10.1016/j.jmbbm.2019.04.043
N. Aslan, B. Aksakal, and F. Findik, Fabrication of Porous-Ti6Al4V alloy by Using Hot Pressing Technique and Mg Space Holder for Hard-Tissue Biomedical Applications, J. Mater. Sci. Mater. Med., 2021, 32(7), p 80. https://doi.org/10.1007/s10856-021-06546-2
J. Kim, E. Nava, and S. Rakici, Nonlinear Finite Element Model for Bending Analysis of Functionally-Graded Porous Circular/Annular Micro-Plates under Thermomechanical Loads Using Quasi-3D Reddy Third-Order Plate Theory, Materials, 2023 https://doi.org/10.3390/ma16093505
M. Roy, V.K. Balla, A. Bandyopadhyay, and S. Bose, Compositionally Graded Hydroxyapatite/Tricalcium Phosphate Coating on Ti by Laser and Induction Plasma, Acta Biomater., 2011, 7(2), p 866–873. https://doi.org/10.1016/j.actbio.2010.09.016
C. Han, Y. Li, Q. Wang, D. Cai, Q. Wei, L. Yang, S. Wen, J. Liu, and Y. Shi, Titanium/hydroxyapatite (Ti/HA) Gradient Materials with Quasi-Continuous Ratios Fabricated by SLM: Material Interface and Fracture Toughness, Mater. Des., 2018, 141, p 256–266. https://doi.org/10.1016/j.matdes.2017.12.037
E. Yilmaz, F. Kabatas, A. Gokce, and F. Findik, Production and Characterization of a Bone-Like Porous Ti/Ti-Hydroxyapatite Functionally Graded Material, J. Mater. Eng. Perform., 2020, 29(10), p 6455–6467. https://doi.org/10.1007/s11665-020-05165-2
Y. Liu, L. Ming, H. Luo, W. Liu, Y. Zhang, H. Liu, and Y. **, Integration of a Calcined Bovine Bone and BMSC-Sheet 3D Scaffold and the Promotion of Bone Regeneration In Large Defects, Biomaterials, 2013, 34(38), p 9998–10006. https://doi.org/10.1016/j.biomaterials.2013.09.040
X. Yu, R. Xu, Z. Zhang, Q. Jiang, Y. Liu, X. Yu, and F. Deng, Different Cell and Tissue Behavior of Micro-/Nano-Tubes and Micro-/Nano-Nets Topographies on Selective Laser Melting Titanium to Enhance Osseointegration, Int. J. Nanomed., 2021, 16, p 3329–3342. https://doi.org/10.2147/ijn.S303770
L. Zhang, J. Tan, Z.Y. He, and Y.H. Jiang, Effect of Calcium Pyrophosphate on Microstructural Evolution and In Vitro Biocompatibility of Ti-35Nb-7Zr Composite by Spark Plasma Sintering, Mater. Sci. Eng. C Mater. Biol. Appl., 2018, 90, p 8–15. https://doi.org/10.1016/j.msec.2018.04.042
P. Balbinotti, E. Gemelli, G. Buerger, S.A.D. Lima, J.D. Jesus, N.H.A. Camargo, V.A.R. Henriques, and G.D.D.A. Soares, Microstructure Development on Sintered Ti/HA Biocomposites Produced by Powder Metallurgy, Mater. Res., 2011, 14(3), p 384–393. https://doi.org/10.1590/S1516-14392011005000044
L. Hao, S. Dadbakhsh, O. Seaman, and M. Felstead, Selective laser Melting of a Stainless Steel and Hydroxyapatite Composite for Load-Bearing Implant Development, J. Mater. Process. Technol., 2009, 209(17), p 5793–5801. https://doi.org/10.1016/j.jmatprotec.2009.06.012
C.A. Terrazas, L.E. Murr, D. Bermudez, E. Arrieta, D.A. Roberson, and R.B. Wicker, Microstructure and Mechanical Properties of Ti-6Al-4V-5% Hydroxyapatite Composite Fabricated Using Electron Beam Powder Bed Fusion, J. Mater. Sci. Technol., 2019, 35(2), p 309–321. https://doi.org/10.1016/j.jmst.2018.10.025
Y. Liu, G. Wang, Y. Cai, H. Ji, G. Zhou, X. Zhao, R. Tang, and M. Zhang, In Vitro Effects of Nanophase Hydroxyapatite Particles on Proliferation and Osteogenic Differentiation of Bone Marrow-Derived Mesenchymal Stem Cells, J. Biomed. Mater. Res. A, 2009, 90(4), p 1083–1091. https://doi.org/10.1002/jbm.a.32192
T.J. Webster, C. Ergun, R.H. Doremus, and W.A. Lanford, Increased Osteoblast Adhesion on Titanium-Coated Hydroxylapatite that Forms CaTiO3, J. Biomed. Mater. Res. A, 2003, 67(3), p 975–980. https://doi.org/10.1002/jbm.a.10160
X. Yang, Y. Li, X. Liu, R. Zhang, and Q. Feng, In Vitro Uptake of Hydroxyapatite Nanoparticles and Their Effect on Osteogenic Differentiation of Human Mesenchymal Stem Cells, Stem Cells Int., 2018, 2018, p 2036176. https://doi.org/10.1155/2018/2036176
T. Numata and Y. Okada, Molecular Determinants of Sensitivity and Conductivity of Human TRPM7 to Mg2+ and Ca2+, Channels, 2008, 2(4), p 283–286. https://doi.org/10.4161/chan.2.4.6695
Y. Gao, M.J. Robertson, S.N. Rahman, A.B. Seven, C. Zhang, J.G. Meyerowitz, O. Panova, F.M. Hannan, R.V. Thakker, H. Bräuner-Osborne, J.M. Mathiesen and G. Skiniotis, Asymmetric Activation of the Calcium-Sensing Receptor Homodimer, Nature, 2021, 595(7867), p 455–459. https://doi.org/10.1038/s41586-021-03691-0
A. González-Vázquez, J.A. Planell, and E. Engel, Extracellular Calcium and CaSR Drive Osteoinduction in Mesenchymal Stromal Cells, Acta Biomater., 2014, 10(6), p 2824–2833. https://doi.org/10.1016/j.actbio.2014.02.004
N. Lu, X. Wang, W. Shi, L. Bian, X. Zhang, G. Yang, X. Tang, J. Wang, Y. Zou, and Y. Weng, Black Phosphorus Nanoparticles Promote Osteogenic Differentiation of EMSCs Through Upregulated TG2 Expression, Nanoscale Res. Lett., 2021, 16(1), p 154. https://doi.org/10.1186/s11671-021-03610-2
C. Chu, X. Xue, J. Zhu, and Z. Yin, In Vivo Study on Biocompatibility and Bonding Strength of Ti/Ti–20 vol% HA/Ti–40 vol% HA Functionally Graded Biomaterial with Bone Tissues in the Rabbit, Mater. Sci. Eng. A, 2006, 429(1–2), p 18–24. https://doi.org/10.1016/j.msea.2006.03.099
X. Wang, C. Wan, X. Feng, F. Zhao, and H. Wang, In Vivo and In Vitro Analyses of Titanium-Hydroxyapatite Functionally Graded Material for Dental Implants, Biomed. Res. Int., 2021, 2021, p 8859945. https://doi.org/10.1155/2021/8859945
P. Oberbek, T. Bolek, A. Chlanda, S. Hirano, S. Kusnieruk, J. Rogowska-Tylman, G. Nechyporenko, V. Zinchenko, W. Swieszkowski, and T. Puzyn, Characterization and Influence of Hydroxyapatite Nanopowders on Living Cells, Beilstein J. Nanotechnol., 2018, 9, p 3079–3094. https://doi.org/10.3762/bjnano.9.286
L.L. Song, Y.Q. Qu, Y.P. Tang, X. Chen, H.H. Lo, L.Q. Qu, Y.X. Yun, V.K.W. Wong, R.L. Zhang, H.M. Wang, M.H. Liu, W. Zhang, H.X. Zhang, J.T.W. Chan, C.R. Wang, J.H. Wu, and B.Y.K. Law, Hyperoside Alleviates Toxicity of β-Amyloid Via Endoplasmic Reticulum-Mitochondrial Calcium Signal Transduction Cascade in APP/PS1 Double Transgenic Alzheimer’s Disease Mice, Redox Biol., 2023, 61, p 102637. https://doi.org/10.1016/j.redox.2023.102637
C. Giorgi, F. Baldassari, A. Bononi, M. Bonora, E. De Marchi, S. Marchi, S. Missiroli, S. Patergnani, A. Rimessi, J.M. Suski, M.R. Wieckowski, and P. Pinton, Mitochondrial Ca2+ and Apoptosis, Cell Calcium, 2012, 52(1), p 36–43. https://doi.org/10.1016/j.ceca.2012.02.008
Z. Wang, C. Wang, C. Li, Y. Qin, L. Zhong, B. Chen, Z. Li, H. Liu, F. Chang, and J. Wang, Analysis of Factors Influencing Bone Ingrowth into Three-Dimensional Printed Porous Metal Scaffolds: A Review, J. Alloy Compd., 2017, 717, p 271–285. https://doi.org/10.1016/j.jallcom.2017.05.079
C. Gao, M. Yao, S. Peng, W. Tan, and C. Shuai, Pre-Oxidation Induced In Situ Interface Strengthening in Biodegradable Zn/nano-SiC Composites Prepared by Selective Laser Melting, J. Adv. Res., 2022, 38, p 143–155. https://doi.org/10.1016/j.jare.2021.09.014
V. Mandal, P. Tripathi, A. Kumar, S.S. Singh, and J. Ramkumar, A Study on Selective Laser Melting (SLM) of TiC and B4C Reinforced IN718 Metal Matrix Composites (MMCs), J. Alloy Compd., 2022, 901, p 17. https://doi.org/10.1016/j.jallcom.2021.163527
Acknowledgments
The authors thank Mr. Zhang Chunyu and Ms. Yan Yu of Guangdong Jianchi Biotechnology Company Limited, Dr. Eskandar Fereiduni of McMaster University, and all of the research staff members at the Department of Oral Implantology, Guanghua School of Stomatology, Sun Yat-sen University. This research was funded by the Foshan Science and Technology Innovation Project (No. 2018IT100212) and the Guangdong Provincial Science and Technology Major Project (No. 2017B090912004).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
No potential conflict of interest was reported by the authors.
Ethics Approval
All methods were performed in accordance with the National Research Council’s Guide for the Care and Use of Laboratory Animals. Rabbits included in this study were provided from the Animal Experiment Center of Sun Yat-sen University with the approval number of SYSU-IACUC-2021-000725.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lin, Y., Balbaa, M., Zeng, W. et al. Osteogenic Properties of Titanium Alloy Ti6Al4V-Hydroxyapatite Composites Fabricated by Selective Laser Melting. J. of Materi Eng and Perform (2023). https://doi.org/10.1007/s11665-023-08632-8
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11665-023-08632-8