Abstract
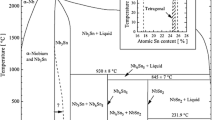
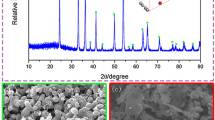
Chemical vapor synthesis was used to prepare Nb nanopowders with NbCl5 vapor as a raw material and Na vapor as a reductant. Thermodynamic analysis indicated that Na had a strong ability to reduce NbCl5. X-ray diffraction (XRD), field emission scanning electron microscopy (FESEM) with energy dispersive spectrometry (EDS), and transmission electron microscopy (TEM) were used to analyze and characterize the product powders. These powders contained two phases: Nb and NaCl. Pure-phase Nb powders were synthesized by vacuum evaporation. The spherical Nb particles had an average size of 30 to 120 nm. In addition, the effects of operating conditions, such as the evaporation temperature, the reaction temperature, and the argon flow rate, on the Nb particle size were investigated.












Similar content being viewed by others
References
J.S. Yoon, J.Y. Yang, J.M. Lee, and S.J. Hong: Mater. Trans., 2013, vol. 54, pp. 114-118.
Y. Freeman, G.F. Alapatt, W.R. Harrell, and P. Lessner: J. Electrochem. Soc., 2012, vol. 159, pp. 1646-1651.
E.Y. Jeong and C. **sub: Electrochem. Commun., 2011, vol. 13, pp. 298-301.
M. Trainer: Mater. Chem. Phys., 2003, vol. 80, pp. 264-268.
J. Zhu, K. Huang, J.G. Hou, and H.M. Zhu: T. Nonferr. Metal. Soc., 2014, vol. 24, pp. 3987-3993.
W.H. Yin: Rare Metal. Mat. Eng., 1998, vol. 1, pp. 1-8.
M.A. Hunter: J. Met., 1953, vol. 5, pp. 130-132.
T.H. Okabe, S. Iwata, M. Imagunbai, and M. Maeda: ISIJ Int., 2003, vol. 43, pp. 1882-1889.
T.H. Okabe, N. Sato, Y. Mitsuda, and S. Ono: Mater. Trans., 2003, vol. 44, pp. 2646–2653.
T.H. Okabe, S. Iwata, M. Imagunbai, Y. Mitsuda, and M. Maeda: ISIJ Int., 2004, vol. 44, pp. 285–293.
M. Baba, Y. Ono, and R.O. Suzuki: J. Phys. Chem. Sol., 2005, vol. 66, pp. 466–470.
T.H. Okabe, I. Park, K.T. Jacob, and Y. Waseda: J. Alloys Compd., 1999, vol. 288, pp. 200–210.
K. Ono and R.O. Suzuki: J. Metals, 2002, vol. 54, pp. 59–61.
K. Ono and R.O. Suzuki: Bull. Jpn. Inst. Metal, 2002, vol. 41, pp. 28–31.
R.O. Suzuki: J. Phys. Chem. Sol., 2005, vol. 66, pp. 461-465.
X.Y. Yan and D.J. Fray: J. Mater. Res., 2003, vol. 18, pp. 346–356.
Y.W. Lv: Rare. Metal Mat. Eng., 2017, vol. 9, pp. 2499-2503.
G.Q. Zhou, L. Hu, L.Z. Wei, and F.P. Zhang: J. Funct. Mater., 2017, vol. 4, pp. 4110-4115.
Z.Y. Shen, L.M. He, G.H. Huang, R.D. Mu, J.W. Gu, and W.Z. Liu: Acta Metall. Sin., 2016, vol. 12, pp. 1579-1585.
T. Ishigaki, S. Tatsuoka, K. Sato, K. Yanagisawa, K. Yamaguchi, and S. Nishida: Int. J. Refract. Met. hard Mater., 2018, vol. 71, pp. 227-231.
H. Lamprey and R.L. Ripley: J. Electrochem. Soc., 1962, vol. 109, pp. 713-716.
Otsuka K, Yamamoto H, Yoshizawa A (1984) J. Chem. Soc. Jpn.: Chem. Ind. Chem. 6:869-878
K.Y. Park, H.D. Jang, and C.S. Choi: Aerosol Sci. Tech., 1998, vol. 28, pp. 215-223.
Y.J. Suh, H.D. Jang, H.K. Chang, D.W. Hwang, and H.C. Kim: Mater. Res. Bull., 2005, vol. 71, pp. 2100-2109.
F. Mirhoseini, A. Bateni, and S. Firoozi: Powder Technol., 2012, vol. 228, pp. 158-162.
H.Y. Sohn and S. Paldey: Metall. Mater. Trans. B, 1998, vol. 29, pp. 457-464.
H.Y. Sohn and S. Paldey: Metall. Mater. Trans. B, 1998, vol. 29, pp. 465-469.
ACKNOWLEDGMENTS
This work was financially supported by Primary Research and Development Project of Anhui Province (Grant Nos. 1804a09020076; 201904a05020040), the Doctor’s Start-up Research Foundation of Anhui University of Science and Technology (Grant No. 11285), the Pre-Research Project of NSFC of Anhui University of Science and Technology (Grant No. 12357), the University-level Key Projects of Anhui University of Science and Technology (Grant No. 12880), the Research Foundation of the Institute of Environment-friendly Materials and Occupational Health of Anhui University of Science and Technology (Wuhu) (Grant No. ALW2020YF13), and the National Natural Science Foundation of China (Grant No. 52071002).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Manuscript submitted on March 7, 2020, accepted September 22, 2020.
Rights and permissions
About this article
Cite this article
Du, C., Pan, C., Pan, Y. et al. Chemical Vapor Synthesis of Ultrafine Niobium Powder Via Sodiothermic Reduction of Chloride. Metall Mater Trans B 51, 2576–2584 (2020). https://doi.org/10.1007/s11663-020-01983-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11663-020-01983-w