Abstract
Purpose
Due to its rarity and biological heterogeneity, guidelines for primary appendiceal tumor (PAT) are based on scarce evidence, resulting in no strong recommendations. The present study explored prognosis-related factors, including the timing of lymph node dissection (LND), in PAT patients after curative resection (CR) to determine the optimal surgical therapies.
Methods
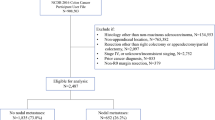
We retrospectively collected and analyzed data from 404 patients with PATs who underwent CR at 43 tertiary hospitals from 2000 to 2017. This manuscript is based on revised manuscript during review process. Please, change the bold characters to normal characters in the manuscript.
Results
After propensity score matching, there were no marked differences in the recurrence-free survival (RFS) or overall survival (OS) between the primary and secondary LND groups (P = 0.993 and 0.728). A multivariate analysis showed that lymph node metastasis (LNM) was an independent factor for the RFS (hazard ratio [HR] 2.59; 95% confidence interval [CI] 1.09–6.13; P = 0.031) and OS (HR 4.70; 95% CI 1.40–15.76; P = 0.012). There were significant associations between the LNM rates and tumor depth (P < 0.0001) and the histological type (P = 0.006). There was no LNM in patients with low-grade appendiceal mucinous neoplasm (LAMN) or well-differentiated mucinous adenocarcinoma (G1) or patients with any Tis or T1 PATs.
Conclusions
LNM was an independent prognostic predictor in PATs after CR with LND. Tumor depth and histological type were not prognostic predictors but were LNM predictors. Secondary LND based on the pathological findings of resected specimens is considered an acceptable surgical management without a worse prognosis than primary LND, and it may be omitted in LAMN+G1 or in any Tis and T1 PATs.



Similar content being viewed by others
Data Availability
The datasets generated and/or analyzed during the current study are available from the corresponding authors on reasonable request, but no information infringing on the privacy of the participants will be given.
References
Fournier K, Rafeeq S, Taggart M, Kanaby P, Ning J, Chen HC, Overman M, Raghav K, Eng C, Mansfield P, Royal R. Low-grade Appendiceal Mucinous Neoplasm of Uncertain Malignant Potential (LAMN-UMP): Prognostic Factors and Implications for Treatment and Follow-up. Ann Surg Oncol 2017;24:187-193.
Misdraji J, Carr NJ, Pai RK. Appendiceal mucinous neoplasm, In: Carneiro F, Chan JKC, Cheung AN et al (eds). World Health Organization Classification of Tumours: Digestive System Tumours, 5th ed. IARC Press: Lyon, 2019, pp 140-155
Carr NJ, Cecil TD, Mohamed F, Sobin LH, Sugarbaker PH, González-Moreno S, Taflampas P, Chapman S, Moran BJ. A Consensus for Classification and Pathologic Reporting of Pseudomyxoma Peritonei and Associated Appendiceal Neoplasia: The Results of the Peritoneal Surface Oncology Group International (PSOGI) Modified Delphi Process. Am J Surg Pathol 2016;40:14-26.
Amin MB, Edge SB, Greene FL (eds). AJCC Cancer Staging Manual. 8th ed. Springer, New York, 2017.
Overman MJ, Asare EA, Compton CC. Appendix—carcinoma. In: Amin M, editor. AJCC Cancer Staging Manual. 8th ed. Springer; Chicago, 2017, pp 237–250.
Matsui S, Murata K, Fukunaga Y, Takeda T, Fujii M, Yamaguchi T, Kagawa Y, Mizushima T, Ohno Y, Yao T, Doki Y, Sugihara K. Analysis of clinicopathological characteristics of appendiceal tumors in Japan: a multicenter collaborative retrospective clinical study — a Japanese Nationwide Survey. Dis Colon Rectum 2020;63:1403-1410.
Sueda T, Murata K, Takeda T, Kagawa Y, Hasegawa J, Komori T, Noura S, Ikeda K, Tsujie M, Ohue M, Ota H, Ikenaga M, Hata T, Matsuda C, Mizushima T, Yamamoto H, Sekimoto M, Nezu R, Mori M, Doki Y. Survival outcomes of appendiceal mucinous neoplasms by histological type and stage: Analysis of 266 cases in a multicenter collaborative retrospective clinical study. Ann Gastroenterol Surg 2019;3:291-300.
Landry CS, Woodall C, Scoggins CR, Mc Masters KM, Martin RC 2nd. Analysis of 900 appendiceal carcinoid tumors for a proposed predictive staging system. Arch Surg 2008;143:664-670.
National Comprehensive Cancer Network. Clinical practice guidelines in oncology (NCCN guidelines): colon cancer: version 2, 2017.
UICC. TNM Classification of Malignant Tumours, 8th ed. Wiley, New York, 2017.
Nitecki SS, Wolff BG, Schlinkert R, Sarr MG. The natural history of surgically treated primary adenocarcinoma of the appendix. Ann Surg 1994;219:51-57.
Nash GM, Smith JD, Tang L, Weiser MR, Temple LK, O’Reilly E, Saltz LB, Guillem JG, Paty PB. Lymph Node Metastasis Predicts Disease Recurrence in a Single-Center Experience of 70 Stages 1-3 Appendix Cancers: A Retrospective Review. Ann Surg Oncol 2015;22:3613-3617.
González-Moreno S, Sugarbaker PH. Right hemicolectomy does not confer a survival advantage in patients with mucinous carcinoma of the appendix and peritoneal seeding. Br J Surg 2004;91:304-311
Day RW, Chang YH, Stucky CC, Gray R, Pockaj B, Wasif N. A Predictive Model for Nodal Metastases in Patients With Appendiceal Cancers. Ann Surg 2019 274:155–166.
Foster JM, Gupta PK, Carreau JH, Grotz TE, Blas JV, Gatalica Z, Nath S, Loggie BW. Right hemicolectomy is not routinely indicated in pseudomyxoma peritonei. Am Surg 2012;78:171-177
Sugarbaker PH. When and When Not to Perform a Right Colon Resection with Mucinous Appendiceal Neoplasms. Ann Surg Oncol 2017;24:729-732.
Andersson RE, Petzold MG. Nonsurgical treatment of appendiceal abscess or phlegmon: a systematic review and meta-analysis. Ann Surg 2007;246:741-748.
Deelder JD, Richir MC, Schoorl T, Schreurs WH. How to treat an appendiceal inflammatory mass: operatively or nonoperatively? J Gastrointest Surg 2014;18:641-645.
Augestad KM, Bakaki PM, Rose J, Crawshaw BP, Lindsetmo RO, Dørum LM, Koroukian SM, Delaney CP. Metastatic spread pattern after curative colorectal cancer surgery. A retrospective, longitudinal analysis. Cancer Epidemiol 2015;39:734-744.
Uen YH, Lu CY, Tsai HL, Yu FJ, Huang MY, Cheng TL, Lin SR, Wang JY. Persistent presence of postoperative circulating tumor cells is a poor prognostic factor for patients with stage I−III colorectal cancer after curative resection. Ann Surg Oncol 2008;15:2120-2128.
Cady B. Basic principles in surgical oncology. Arch Surg 1997;132:338-346.
Kawada K, Taketo MM. Significance and mechanism of lymph node metastasis in cancer progression. Cancer Res 2011;71:1214-1218.
**e X, Zhou Z, Song Y, Li W, Diao D, Dang C, Zhang H. The Management and Prognostic Prediction of Adenocarcinoma of Appendix. Sci Rep 2016;6:39027.
Acknowledgements
The following doctors cooperated in this study: M. Ikeda (Hyogo College of Medicine), M. Itabashi (Tokyo Women’s Medical University), Y. Ide (Yao Municipal Hospital), M. Ito (National Cancer Center Hospital East), A. Iwashita (Fukuoka University Chikushi Hospital), S. Okamura (Suita Municipal Hospital), E. Oki (Kyushu University), H. Ozawa (Tochigi Cancer Center), T. Kato (Osaka National Hospital), Y. Kanemitsu (National Cancer Center Hospital), M. Shiozawa (Kanagawa Cancer Center), T. Shioda (Kobe City Nishi-Kobe Medical Center), A. Shiomi (Shizuoka Cancer Center Hospital), I. Takemasa (Sapproro Medical University), H. Taniguchi (Aichi Cancer Center Hospital), M. Nakanishi (Kyoto Prefectural University of Medicine), T. Furuhata (St. Marianna Medical University Toyoko Hospital), S. Morita (Toyonaka Municipal Hospital), M. Yasui (Osaka International Cancer Institute), K. Yamazaki (Shizuoka Cancer Center Hospital), H. Yokomizo (Tokyo Women’s Medical University Medical Center East), K. Yoshimatsu (Saiseikai Kurihashi Hospital), T. Komori (Osaka General Medical Center), M. Tsujie (Sakai City Medical Center), H. Ohta (Ikeda City Hospital), M. Ikenaga (Higashiosaka City Medical Center), H. Mizuno (Nippon Life Hospital), A. Ogawa (Tane General Hospital), K. Yasumasa (Japan Community Health care Organization Osaka Hospital), K. Tamagawa (Otemae Hospital), H. Takemoto (Kinki Central Hospital), T. Shingai (Saiseikai Senri Hospital), Y. Suzuki (Osaka Police Hospital), M. Fukunaga (Hyogo Prefectural Nishinomiya Hospital), Y. Fukunaga (Cancer Institute Hospital), Y. Ohno (Osaka University), T. Mizushima (Osaka University), and Y. Doki (Osaka University).
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 20 kb)
Rights and permissions
About this article
Cite this article
Takeyama, H., Murata, K., Takeda, T. et al. Clinical Significance of Lymph Node Dissection and Lymph Node Metastasis in Primary Appendiceal Tumor Patients After Curative Resection: a Retrospective Multicenter Cohort Study. J Gastrointest Surg 26, 128–140 (2022). https://doi.org/10.1007/s11605-021-05070-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11605-021-05070-6