Abstract
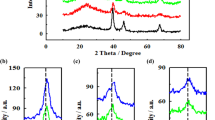
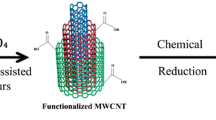
The composition and atomic structure of nanoalloy electrocatalysts play an important role in the ethanol oxidation reaction (EOR). To study this and develop an electrochemically active electrocatalyst towards ethanol electro-oxidation, a second oxophilic metal has generally been initiated as an alloy, capable of enhancing the electrocatalytic activity of platinum. Given the above, the present investigation deals with nanoparticles of PtRu with different bimetallic compositions supported on multi-walled carbon nanotube (MWCNT) (C) for their activity on the ethanol electro-oxidation. The structure, morphology, and bonding of the electrocatalysts were studied by XRD, SEM, TEM, EDXRF, and FTIR. Electrochemical methods like cyclic voltammetry (CV), linear sweep voltammetry (LSV), chronoamperometry (CA), and electrochemical impedance spectroscopy (EIS) were used to explore the catalytic activity towards ethanol electro-oxidation. This study focused on establishing the relationship between the electrocatalysts’ composition, atomic structure, and catalytic activity for the EOR. All the results of physical and electrochemical studies revealed an intriguing composition–structure–activity relationship for the PtRu electrocatalysts under EOR experimental conditions. In particular, the alloy with a Pt/Ru ratio of ∼ 84 : 16 was found to exhibit a maximum EOR activity as a function of the bimetallic composition. Notably, the catalytic activities of the C/PtRu electrocatalysts showed a significant improvement during the EOR. The results show a new perspective in the development of C/PtRu electrocatalysts regarding ethanol oxidation reaction which is important for the rational design and synthesis of active nanoalloy electrocatalysts for direct ethanol fuel cell (DEFC).

























Similar content being viewed by others
Data availability
Data will be made available on request.
References
Singh S, Datta J (2021) Influence of Nafion template on the kinetics of anodic Pt based pluri-metallic electrocatalyst for ethanol electro-oxidation. J Electroanal Chem 882:114974–114987. https://doi.org/10.1016/j.jelechem.2020.114974
Zhong JP, Hou C, Li L, Waqas M, Fan YJ, Shen XC, Chen W, Wan LY, Liao HG, Sun SG (2020) A novel strategy for synthesizing Fe, N, and S tridoped graphene-supported Pt nanodendrites toward highly efficient methanol oxidation. J Catal 381:275–228. https://doi.org/10.1016/j.jcat.2019.11.002
Wang RX, Fan YJ, Wang L, Wu LN, Sun SN, Sun SG (2015) Pt nanoelectrocatalysts on a polyindole-functionalized carbon nanotube composite with high performance for methanol electrooxidation. J Power Sources 287:341–348. https://doi.org/10.1016/j.jpowsour.2015.03.181
Fan JJ, Fan YJ, Wang RX, **ang S, Tanga HG, Sun SG (2017) A novel strategy for the synthesis of sulfur-doped carbon nanotubes as a highly efficient Pt electrocatalyst support toward the methanol oxidation reaction. J Mater Chem A 5:19467–19475. https://doi.org/10.1039/C7TA05102F
Rousseau S, Contanceau C, Lamy C, Leger JM (2006) Direct ethanol fuel cell (DEFC): electrical performances and reaction products distribution under operating conditions with different platinum-based anodes. J Power Sources 158:18–24. https://doi.org/10.1016/j.jpowsour.2005.08.027
Lopez V, Fabiola V, Mason T, Shearing PR, Brett DJL (2020) Carbon monoxide poisoning and mitigation strategies for polymer electrolyte membrane fuel cells – a review. Prog Energy Combust Sci 79:100842–100912. https://doi.org/10.1016/j.pecs.2020.100842
Zhong J, Li L, Waqas M, Wang X, Fan Y, Qi J, Yang B, Rong C, Chen W, Sun S (2019) Deep eutectic solvent-assisted synthesis of highly efficient PtCu alloy nanoclusters on carbon nanotubes for methanol oxidation reaction. Electrochim Acta 322:134677. https://doi.org/10.1016/j.electacta.2019.134677
**ang S, Wang L, Huang CC, Fan YJ, Tang HG, Wei L, Sun SG (2018) Concave cubic PtLa alloy nanocrystals with high-index facets: controllable synthesis in deep eutectic solvents and their superior electrocatalytic properties for ethanol oxidation. J Power Sources 399:422–428. https://doi.org/10.1016/j.jpowsour.2018.07.102
Datta J, Singh S, Sengupta S, Mukherjee S, Mukherjee M (2011) Significant role of Ru-oxide present in the Pt-Ru alloy electrocatalyst for ethanol electro-oxidation in acid medium. Mater Manuf Process 26:261–271. https://doi.org/10.1080/10426914.2010.520796
Datta J, Singh S, Das S, Bandyopadhyay NR (2009) A comprehensive study on the effect of Ru addition to Pt electrodes for direct ethanol fuel cell. Bull Mater Sci 32(6):643–652 https://www.ias.ac.in/article/fulltext/boms/032/06/0643-0652. Accessed 2009
Sengupta S, Singh S, Datta J (2009) Promoting role of unalloyed Sn in PtSn binary electrocatalysts for ethanol electro-oxidation. Mater Chem Phys 116:223–228. https://doi.org/10.1016/j.matchemphys.2009.03.016
Sen Gupta S, Singh S, Datta J (2010) Temperature effect on the electrode kinetics of ethanol electro-oxidation on Sn modified Pt electrocatalyst through voltammetry and impedance spectroscopy. Mater Chem Phys 120:682–690. https://doi.org/10.1016/j.matchemphys.2009.12.023
Shen SY, Zhao TS, Xu JB (2010) Carbon supported PtRh electrocatalysts for ethanol oxidation in alkaline direct ethanol fuel cell. Int J Hydrog Energy 35:12911–12917. https://doi.org/10.1016/j.ijhydene.2010.08.107
Akhairi MAF, Kamarudin SK (2016) Electrocatalysts in direct ethanol fuel cell (DEFC): an overview. Int J Hydrog Energy 41:4214–4228. https://doi.org/10.1016/j.ijhydene.2015.12.145
Silva EL, Cuna VMRO, Radtke C, Machado G, Tancredi N, Malfatti CF (2016) Influence of the support on PtSn electrocatalysts behavior: ethanol electro-oxidation performance and in-situ ATR-FTIRS studies. Appl Catal 193:170–179. https://doi.org/10.1016/j.apcatb.2016.04.021
Lee K, Zhang J, Wang H, Wilkinson DP (2006) Progress in the synthesis of carbon nanotube- and nanofiber-supported Pt electrocatalysts for PEM fuel cell catalysis. J Appl Electrochem 36:507–522. https://doi.org/10.1007/s10800-006-9120-4
Parreira LS, Silva JCM, Simoes FR, Cordeiro MAL, Satao RH, Leite ER, Santos MCD (2017) PtSn electrocatalyst supported on MWCNT-COOH: investigating the ethanol oxidation reaction. Chem Electro Chem 4:1950–1958
Centi G, Perathoner S (2009) The role of nanostructure in improving the performance of electrodes for energy storage and conversion. Eur J Inorg Chem 26:3851–3878. https://doi.org/10.1002/ejic.2009002751
Kim J, Lee SW, Carlton C, Shao-Horn Y (2011) Pt-covered multiwall carbon nanotubes for oxygen reduction in fuel cell applications. J Phys Chem Lett 2:1332–1336. https://doi.org/10.1021/jz200531z
Guo JW, Zhao TS, Prabhuram J, Chen R, Wong CW (2005) Preparation and characterization of a PtRu/C nano electrocatalyst for direct methanol fuel cells. Electrochem Acta 51:754–763. https://doi.org/10.1016/j.electacta.2005.05.056
Young RA, Willes DB (1982) Profile shape functions in Rietveld refinements. J Appl Crystallogr 15:430–438. https://doi.org/10.1021/acs.cgd.0c00956
Ghosh M, Pradhan S, Mandal S, Roy A, Chakrabarty S, Chakrabarti G, Pradhan SK (2022) Enhanced antibacterial activity of a novel protein-arginine deiminase type-4 (PADI4) inhibitor after conjugation with a biocompatible nanocarrier. J Drug Deliv Sci Technol 74:103549. https://doi.org/10.1016/j.jddst.2022.103549
Ghosh M, Mandal S, Roy A, Chakrabarty S, Chakrabarti G, Pradhan SK (2020) Enhanced antifungal activity of fluconazole conjugated with Cu-Ag-ZnO nanocomposite. Mater Sci Eng C 106:110160–110170. https://doi.org/10.1016/j.msec.2019.110160
Ghosh M, Mandal S, Dutta S, Paladhi A, Ray S, Hira SK, Pradhan SK (2021) Synthesis of drug conjugated magnetic nanocomposite with enhanced hypoglycemic effects. Mater Sci Eng C 120:111697–111741. https://doi.org/10.1016/j.msec.2020.111697
Ghosh M, Mandal S, Roy A, Paladhi A, Mondal P, Hira SK, Mukhopadhyay SK, Pradhan SK (2021) Synthesis and characterization of a novel drug conjugated copper-silver-titanium oxide nanocomposite with enhanced antibacterial activity. J Drug Deliv Sci Technol 62:102384–102395. https://doi.org/10.1016/j.msec.2020.111697
Ghosh M, Mandal S, Roy A, Mondal P, Mukhopadhyay SK, Chakrabarty S, Chakrabarti G, Pradhan SK (2021) Synthesis and characterization of a novel nanocarrier for biocompatible targeting of an antibacterial therapeutic agent with enhanced activity. J Drug Deliv Sci Technol 66:102821–102830. https://doi.org/10.1016/j.msec.2020.111697
Ghosh M, Mondal M, Mandal S, Roy A, Chakrabarty S, Chakrabarti G, Pradhan SK (2020) Enhanced photocatalytic and antibacterial activities of mechanosynthesized TiO2-Ag nanocomposite in wastewater treatment. J Mol Struct 1211:128076–128087. https://doi.org/10.1016/j.msec.2020.111697
Bunaciu AA, Udristioiu EG, Aboul-Enein HY (2015) X-ray diffraction: instrumentation and applications. Reviews in Anal Chem 45:289–299. https://doi.org/10.1080/10408347.2014.949616
Tripković AV, Popović KDJ, Lović JD (2007) Kinetic study of methanol oxidation on Pt2Ru3/C electrocatalyst in the alkaline media. J Serb Chem Soc 72:1095–1101. https://doi.org/10.2998/JSC0711095T
Antolini E, Cardellini F (2001) Formation of carbon supported PtRu alloys: an XRD analysis. J Alloys Compd 315:118–122. https://doi.org/10.1016/S0925-8388(00)01260-3
Yang G, Sun Y, Lv P, Zhen F, Cao X, Chen X, Wang Z, Yuan Z, Kong X (2016) Preparation of Pt–Ru/C as an oxygen-reduction electrocatalyst in microbial fuel cells for wastewater treatment electrocatalysts. J Catal 6:150–160. https://doi.org/10.3390/catal6100150
Momma K, Izumi F (2011) VESTA 3 for three-dimensional visualization of crystal, volumetric and morphology data. J. Appl. Cryst. 44:1272–1276. https://doi.org/10.1080/10408347.2014.949616
Momma K, Izumi F (2008) VESTA: a three-dimensional visualization system for electronic and structural analysis. J Appl Cryst 41:653–658. https://doi.org/10.1107/S0021889808012016
Ogihara Y, Yano H, Matsumoto T, Tryk DA, Iiyama A, Uchida A (2017) In situ FTIR analysis of CO-tolerance of a Pt-Fe alloy with stabilized Pt skin layers as a hydrogen anode electrocatalyst for polymer electrolyte fuel cells. Catalysis 7:8–20. https://doi.org/10.3390/catal7010008
Elgrishi N, Rountree KJ, McCarthy BD, Rountree ES, Thomas T, Eisenhart DJL (2018) A practical beginner’s guide to cyclic voltammetry. J Chem Educ 95:197–206. https://doi.org/10.1021/acs.jchemed.7b00361
Chen B, Bruekner TM, Altarawneh RM, Pickup PG (2018) Composition dependence of ethanol oxidation at ruthenium-tin oxide/carbon supported platinum electrocatalysts. J Electrochem Soc 165:J3019–J3025. https://doi.org/10.1149/2.0041815jes
Singh S, Datta J (2011) Kinetic investigations and product analysis for optimizing platinum loading in direct ethanol fuel cell (DEFC) electrodes. Ionics 17:785–798. https://doi.org/10.1021/cen-v074n029.p062
Zhao Y, Yang X, Zhan L, Ou S, Tian J (2011) High electrocatalytic activity of PtRu nanoparticles supported on starch-functionalized multi-walled carbon nanotubes for ethanol oxidation. J Mater Chem 21:4257. https://doi.org/10.1039/C0JM03892J
Yang X, Zheng J, Zhen M, Meng X, Jiang F, Wang T, Shu C, Jiang L, Wang C (2012) A linear molecule functionalized multi-walled carbon nanotubes with well dispersed PtRu nanoparticles for ethanol electro-oxidation. Appl Catal B: Environ 121–122:57–64. https://doi.org/10.1016/j.apcatb.2012.03.027
Acknowledgements
This work was partially carried out using the facilities of UGC-DAE CSR. The authors would also like to express their heartfelt thanks to Dr. Ankan Dutta Chowdhury for providing the instrumental facility regarding electrochemical studies.
Funding
The authors acknowledge the financial support from UGC-DAE CSR through a Collaborative Research Scheme (CRS) project number CRS/2021-22/02/515 funded to Dr. Susmita Singh.
Author information
Authors and Affiliations
Contributions
Susmita Singh: conceptualization, resources, writing—original draft, writing—review and editing, supervision, validation, and project administration
Moupiya Ghosh: investigation, methodology, formal analysis, and writing—original draft
Mainak Bose: investigation, methodology, and writing—review and editing
Sinthia Saha: investigation, methodology, and writing—review and editing
Anushna Dutta: investigation, methodology, and writing—review and editing
Corresponding author
Ethics declarations
Ethical approval
This declaration is not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Singh, S., Ghosh, M., Bose, M. et al. A Study on significant role of Ru on MWCNT-supported PtRu electrocatalysts for ethanol electro-oxidation. Ionics 29, 5417–5430 (2023). https://doi.org/10.1007/s11581-023-05218-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11581-023-05218-1