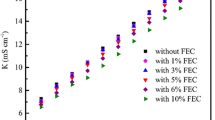
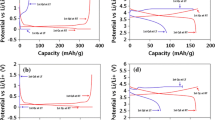
Abstract
Increasing the charging cutoff voltage is an effective method for improving the energy density of lithium-ion batteries. However, the conventional carbonate-based electrolyte with LiPF6 is unstable when the end-of-charge voltage up to 4.5 V (vs. Li/Li+), resulting in poor cycling stability of LIBs. In this work, when 1.0 wt% LiDFP is added into the conventional electrolyte for LiNi0.4Co0.4Mn0.2O2/graphite pouch cells, the capacity retention of the cell is significantly improved from 22.9 to 87.6% after 100 cycles, even a high capacity retention of 81.0% is maintained after 200 cycles. The results of spectroscopic and electrochemical techniques indicate that the passivation film induced by LiDFP can be formed simultaneously at both cathode and anode surfaces. The improvement of cell high-voltage performance can be credited to the addition of LiDFP, which effectively inhibited the dissolution of transition metals and the side reaction of electrolyte on the cathode and anode surfaces.




Similar content being viewed by others
References
Gu S, Cui Y, Wen K, Chen S, Zhao J (2020) 3-cyano-5-fluorobenzenzboronic acid as an electrolyte additive for enhancing the cycling stability of Li1.2Mn0.54Ni0.13Co0.13O2 cathode at high voltage. J Alloys Compd 829:154491
Li X, Shi H, Zhang L, Chen J, Lü P (2020) Novel synthesis of SiOx/C composite as high-capacity lithium-ion battery anode from silica-carbon binary xerogel. Chin J Chem Eng 28:579–583
Zhou F, Yang X, Liu J, Liu J, Hu R, Ouyang L, Zhu M (2021) Boosted lithium storage cycling stability of TiP2 by in-situ partial self-decomposition and nano-spatial confinement. J Power Sources 485:229337
Zhang M, Hu R, Liu J, Ouyang L, Liu J, Yang L, Zhu M (2017) A ZnGeP2/C anode for lithium-ion and sodium-ion batteries. Electrochem Commun 77:85–88
Zhou F, Ouyang L, Liu J, Yang X, Zhu M (2020) Chemical bonding black phosphorus with TiO2 and carbon toward high-performance lithium storage. J Power Sources 449:227549
Tao F, Yan X, Liu J, Lei H, Chen L (2016) Effects of PVP-assisted Co3O4 coating on the electrochemical and storage properties of LiNi0.6Co0.2Mn0.2O2 at high cut-off voltage. Electrochim Acta 210:548–556
Pham H, Mirolo M, Tarik M, Kazzi M, Trabesinger S (2020) Multifunctional electrolyte additive for improved interfacial stability in Ni-rich layered oxide full-cells. Energy Storage Mater 33:216–229
Wang C, Yu L, Fan W, Liu J, Ouyang L, Yang L, Zhu M (2017) 3,3′-(Ethylenedioxy)dipropiononitrile as an electrolyte additive for 4.5 V LiNi1/3Co1/3Mn1/3O2/graphite cells. ACS Appl Mater Interfaces 9:9630–9639
Han Y, Yoo J, Yim T (2016) Distinct reaction characteristics of electrolyte additives for high-voltage lithium-ion batteries: tris(trimethylsilyl) phosphite, borate, and phosphate. Electrochim Acta 215:455–465
Imholt L, Röser S, Börner M, Streipert B, Rad B, Winter M, Cekic-Laskovi I (2017) Trimethylsiloxy based metal complexes as electrolyte additives for high voltage application in lithium ion cells. Electrochim Acta 235:332–339
Li Y, Wang K, Chen J, Zhang W, Luo X, Hu Z, Zhang Q, **ng L, Li W (2020) Stabilized high-voltage cathodes via an F-rich and Si-containing electrolyte additive. ACS Appl Mater Interfaces 12:28169–28178
Wang C, Tang S, Zuo X, **ao X, Liu J, Nan J (2015) 3-(1,1,2,2-Tetrafluoroethoxy)-1,1,2,2-tetrafluoropropane as a high voltage solvent for LiNi1/3Co1/3Mn1/3O2/graphite cells. J Electrochem Soc 162:A1997–A2003
Wang C, Zuo X, Zhao M, **ao X, Yu L, Nan J (2016) 1H,1H,5H-Perfluoropentyl-1,1,2,2-tetrafluoroethylether as a co-solvent for high voltage LiNi1/3Co1/3Mn1/3O2/graphite cells. J Power Sources 307:772–781
Xu K (2004) Nonaqueous liquid electrolytes for lithium-based rechargeable batteries. Chem Rev 104:4303–4417
Markevich E, Baranchugov V, Aurbach D (2006) On the possibility of using ionic liquids as electrolyte solutions for rechargeable 5 V Li ion batteries. Electrochem Commun 8:1331
Kang Y, Park I, Park M, Choi W, Lee S, Mun J, Choi B, Koh M, Kim D, Park K (2020) L-Tryptophan: antioxidant as a film-forming additive for a high-voltage cathode. Langmuir 36:2823–2828
Sun Y, Huang J, **ang H, Liang X, Feng Y, Yu Y (2020) 2-(Trifluoroacetyl) thiophene as an electrolyte additive for high-voltage lithium-ion batteries using LiCoO2 cathode. J Mater Sci Technol 55:198–202
Liu Q, Yang G, Liu S, Han M, Wang Z, Chen L (2019) Trimethyl borate as film-forming electrolyte additive to improve high-voltage performances. ACS Appl Mater Interfaces 11:17435–17443
Yang B, Zhang H, Yu L, Fan W, Huang D (2016) Lithium difluorophosphate as an additive to improve the low temperature performance of LiNi0.5Co0.2Mn0.3O2/graphite cells. Electrochim Acta 221:107–114
Wang C, Yu L, Fan W, Liu R, Liu J, Ouyang L, Yang L, Zhu M (2018) Enhanced high-voltage cyclability of LiNi0.5Co0.2Mn0.3O2-based pouch cells via lithium difluorophosphate introducing as electrolyte additive. J Alloys Compd 755:1–9
Wang C, Yu L, Fan W, Liu J, Ouyang L, Yang L, Zhu M (2018) Lithium difluorophosphate as a promising electrolyte lithium additive for high-voltage lithium-ion batteries. ACS Appl Energy Mater 1:2647–2656
Liu Q, Ma L, Du C, Dahn J (2018) Effects of the LiPO2F2 additive on unwanted lithium plating in lithium-ion cells. Electrochim Acta 263:237–248
Zhao W, Zheng G, Lin M, Zhao W, Li D, Guan X, Ji Y, Ortiz G, Yang Y (2018) Toward a stable solid-electrolyte-interfaces on nickel-rich cathodes: LiPO2F2 salt-type additive and its working mechanism for LiNi0.5Mn0.25Co0.25O2 cathodes. J Power Sources 380:149–157
Zheng H, **ang H, Jiang F, Liu Y, Sun Y, Liang X, Feng Y, Yu Y (2020) Lithium difluorophosphate-based dual-salt low concentration electrolytes for lithium metal batteries. Adv Energy Mater 10:2001440
Shi P, Zhang L, **ang H, Liang X, Sun Y, Xu W (2018) Lithium difluorophosphate as a dendrite-suppressing additive for lithium metal batteries. ACS Appl Mater Interfaces 10:22201–22209
Prakasha K, Madasamy K, Kathiresan M, Prakash A (2019) Ethylviologen hexafluorophosphate as electrolyte additive for high-voltage nickel-rich layered cathode. J Phys Chem C 123:28604–28610
Zheng Y, Xu N, Chen S, Liao Y, Zhong G, Zhang Z, Yang Y (2020) Construction of a stable LiNi0.8Co0.1Mn0.1O2 (NCM811) cathode interface by a multifunctional organosilicon electrolyte additive. ACS Appl Energy Mater 3:2837–2845
Zuo X, Zhao M, Ma X, **ao X, Liu J, Nan J (2017) Effect of diphenyl disulfide as an additive on the electrochemical performance of Li1.2Mn0.54Ni0.13Co0.13O2/graphite batteries at elevated temperature. Electrochim Acta 245:705–714
Hao J, Li X, Zeng X, Li D, Mao J, Guo Z (2020) Deeply understanding the Zn anode behaviour and corresponding improvement strategies in different aqueous Zn-based batteries. Energy Environ Sci 13:3917–3949
Hao J, Yuan L, Ye C, Chao D, Davey K, Guo Z, Qiao S (2021) Boosting zinc electrode reversibility in aqueous electrolytes by using low-cost antisolvents. Angew Chem 133:7442–7451
Funding
This project funded by the China Postdoctoral Science Foundation (2020M682662), the Natural Science Foundations of Guangdong (No. 2018A030313423), and the Foundation for Innovative Research Groups of the National Natural Science Foundation of China (No. NSFC51621001).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wang, C., Liu, M., Huang, D. et al. High-voltage LiNi0.4Co0.4Mn0.2O2/graphite pouch battery cycled at 4.5 V with a LiDFP-based electrolyte . Ionics 27, 4135–4142 (2021). https://doi.org/10.1007/s11581-021-04193-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11581-021-04193-9