Abstract
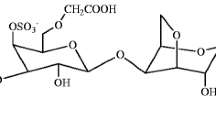
A new type of solid polymer electrolyte based on iota (ɩ)-carrageenan derivative has been investigated. ɩ-Carrageenan was reacted with monochloroacetic acid to produce carboxymethyl ɩ-carrageenan. The produced powders were characterized by 1H nuclear magnetic resonance and attenuated total reflection Fourier transform infrared spectroscopy to confirm the substitution of targeted functional in ɩ-carrageenan. The polymer electrolytes films were prepared by solution casting technique. Thermal gravimetric analysis result shows that the modified powder and film were stable up to 300 °C. X-ray diffraction analysis shows that the chemical modification of ɩ-carrageenan increases the amorphous phase of polymer electrolyte film. Meanwhile, the scanning electron microscope observation reveals that the carboxymethyl ɩ-carrageenan film becomes smoother than ɩ-carrageenan film which supports the ionic conductivity enhancement. The ionic conductivity of ɩ-carrageenan film increases from 2.19 × 10−8 to 4.87 × 10−6 S cm−1 after chemical modification of ɩ-carrageenan to carboxymethyl ɩ-carrageenan.










Similar content being viewed by others
References
Raphael E, Avellaneda CO, Manzolli B, Pawlicka A (2010) Electrochim Acta 55:1455–1459
Samsudin AS, Khairul WM, Isa MIN (2012) J Non-Cryst Solids 358:1104–1112
Vieira DF, Avellaneda CO, Pawlicka A (2007) Electrochim Acta 53:1404–1408
Majid SR, Arof AK (2005) Physica B 355:78–82
Mobarak NN, Ramli N, Ahmad A, Rahman MYA (2012) Solid State Ionics 224:51–57
Rodríguez J, Navarrete E, Dalchiele EA, Sánchez L, Ramos-Barrado JR, Martín F (2013) J Power Sources 237:270–276
Khiar ASA, Puteh R, Arof AK (2006) Physica B 373:23–27
Campo VL, Kawano DF, Silva DB Jr, Carvalho I (2009) Carbohydr Polym 77:167–180
Leong KH, Chung LY, Noordin MI, Mohamad K, Nishikawa M, Onuki Y, Morishita M, Takayama K (2011) Carbohydrate Poly 83:1507–1515
Yang Y, Hu H, Zhou CH, Xu S, Sebo B, Zhao XZ (2011) J Power Sources 196:2410–2415
Otomo J et al (2003) Protonic conduction of CsH2PO4 and its composite with silica in dry and humid atmospheres. Solid State Ionics 156(3–4):357–369
Bandara TMWJ, Dissanayake MAKL, Albinson I, Mellander B-E (1998) J Non-Cryst Solids 241:140–148
Huber B, Rossrucker L (2013) Solid State Ionics 247–248:15–21
Handy S (2011) Applications of ionic liquids in science and technology (2011): InTech, Chapters published September 22, under CC BY-NC-SA 3.0 license
Pavia DL, Lampman GM, Kriz GS (2001) Introduction to spectroscopy: a guide for students of organic chemistry. Harcourt College Publishers, San Diego
Freile-Pelegrín Y, Azamar JA, Robledo D (2011) J Aquatic Food Product Tech 20:73–83
Aranilla CT, Nagasawa N, Bayquen A, Rosa AD (2012) Carbohydrate Poly 87:1810–1816
Fan L, Wang L, Gao S, Wu P, Li M, **e W, Liu S, Wang W (2011) Carbohydrate Poly 86:1167–1174
Fernández-Carretero FJ, Compañ V, Riande E (2007) J Power Sources 173:68–76
Su'ait MS, Ahmad A, Hamzah H, Rahman MYA (2011) Electrochim Acta 57:123–131
Jaffe DA, Rose NJ (1991) Spectrochim Acta A 47:1695–1705
Medhat Ibrahim EK (2004) Acta Chim Slov 51:453–460
Nhlapo NS, Focke WW, Vuorinen E (2012) Thermochim Acta 546:113–119
Noor SAM, Ahmad A, Talib IA, Rahman MYA (2010) Ionics 16:161–170
Costa MM, Terezo AJ, Matos AL, Moura WA, Giacometti JA, Sombra ASB (2010) Physica B 405:4439–4444
Wang S, Jeung S, Min K (2010) Poly 51:2864–2871
Shuhaimi NEA, Teo LP, Woo HJ, Majid SR, Arof AK (2012) Poly Bull 69:807–826
Koh JCH, Ahmad ZA, Mohamad AA (2012) Ionics 18:359–364
Edward R, Barker J (1976) Pure Appl Chem 46:157–170
Rajantharan RS (2011) Investigation on the effects of ionic liquid and ionic mixture in biodegradable polymer electrolytes. Faculty of Engineering and Science, Universiti Tunku Abdul Rahman, Thesis of Master
**a Y, Fujieda T, Tatsumi K, Prosini PP, Sakai T (2001) J Power Sources 92:234–243
Acknowledgments
The authors would like to thank UKM and Fisheries Department for the provision of grants UKM-DLP-2012-021 and STGL-007-2010, respectively.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Jumaah, F.N., Mobarak, N., Ahmad, A. et al. Derivative of iota-carrageenan as solid polymer electrolyte. Ionics 21, 1311–1320 (2015). https://doi.org/10.1007/s11581-014-1306-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11581-014-1306-x