Abstract
Background
Stenosing tenosynovitis or trigger finger is a common clinical condition regularly treated with steroid injections. Varied success is reported at early time points following injection. We present a prospective, randomized IRB-approved study to confirm these findings at a long-term follow-up.
Methods
Adult patients presenting with symptoms of stenosing tenosynovitis who agreed to participate were randomized into two groups. Group 1 received an initial injection of triamcinolone and local anesthetic mixture. Group 2 received the same initial injection and an additional staged injection at 6 weeks. The patients were then followed beyond 2 years. If Group 1 patients were still symptomatic at 6 weeks, another injection was given. An additional injection or surgery was defined as treatment failure. DASH scores were obtained at baseline, 3, 6, and 12 months.
Results
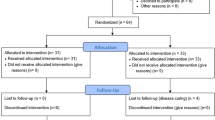
Ninety-seven patients (101 trigger digits) were enrolled in the prospective trial. Fifty-six digits were randomized to the one-injection group versus 45 digits randomized to the two-injection group (“intention to treat analysis”—ITT). After accounting for crossover between the groups, 42 patients received one-injection versus fifty-nine patients receiving two injections (“actual” analysis).
Overall failure was the same between the two groups. However, a higher surgery rate was noted for patients having undergone two injections versus one injection [47 % versus 27 % (p < 0.013), ITT]. Diabetes was associated with a higher surgery rate at 1 year within the group of overall failures [56 % versus 37 % (p = 0.0505), ITT]. High baseline DASH score (>40) was associated with a median time of 10 months for failure and 6 months for surgery as per a Kaplan–Meier survival analysis (p < 0.005 and p < 0.001, respectively, ITT).
Conclusions
As overall failure of steroid injection for trigger finger is not improved with staged, two-injection treatment, we recommend a single injection initial treatment for trigger finger with a second injection given in cases of recurrence or failure. Diabetes was a risk factor for needing surgery if failure occurred within 1 year. The baseline DASH score is helpful in predicting which patients have a higher chance of failing as well as needing surgery.
Level of evidence
Prospective, randomized trial, level I.




Similar content being viewed by others
References
Baumgarten KM, Gerlach D, Boyer MI. Corticosteroid injection in diabetic patients with trigger finger. A prospective, randomized, controlled double-blinded study. J Bone Joint Surg Am. 2007;89(12):2604–11.
Blyth MJ, Ross DJ. Diabetes and trigger finger. J Hand Surg (Br). 1996;21(2):244–5.
Chammas M, Bousquet P, Renard E, Poirier JL, Jaffiol C, Allieu Y. Dupuytren’s disease, carpal tunnel syndrome, trigger finger, and diabetes mellitus. J Hand Surg [Am]. 1995;20(1):109–14.
Clark DD, Ricker JH, MacCollum MS. The efficacy of local steroid injection in the treatment of stenosing tenovaginitis. Plast Reconstr Surg. 1973;51(2):179–80.
Fahey JJ, Bollinger JA. Trigger-finger in adults and children. J Bone Joint Surg Am. 1954;36-A(6):1200–18.
Fleisch SB, Spindler KP, Lee DH. Corticosteroid injections in the treatment of trigger finger: a level I and II systematic review. J Am Acad Orthop Surg. 2007;15(3):166–71.
Freiberg A, Mulholland RS, Levine R. Nonoperative treatment of trigger fingers and thumbs. J Hand Surg [Am]. 1989;14(3):553–8.
Kerrigan CL, Stanwix MG. Using evidence to minimize the cost of trigger finger care. J Hand Surg [Am]. 2009;34(6):997–1005. doi:10.1016/j.jhsa.2009.02.029.
Kolind-Sorensen V. Treatment of trigger fingers. Acta Orthop Scand. 1970;41(4):428–32.
Lapidus PW. Stenosing tenovaginitis. Surg Clin North Am. 1953 Oct:1317–47 PMD 13102191.
Lapidus PW, Guidotti FP. Stenosing tenovaginitis of the wrist and fingers. Clin Orthop Relat Res. 1972;83:87–90.
Maneerit J, Sriworakun C, Budhraja N, Nagavajara P. Trigger thumb: results of a prospective randomised study of percutaneous release with steroid injection versus steroid injection alone. J Hand Surg (Br). 2003;28(6):586–9.
Marks MR, Gunther SF. Efficacy of cortisone injection in treatment of trigger fingers and thumbs. J Hand Surg [Am]. 1989;14(4):722–7.
Murphy D, Failla JM, Koniuch MP. Steroid versus placebo injection for trigger finger. J Hand Surg [Am]. 1995;20(4):628–31. Erratum in: J, Hand Surg [Am] 1995 Nov;20(6):1075.
Peters-Veluthamaningal C, van der Windt DA, Winters JC, Meyboom-de Jong B. Corticosteroid injection for trigger finger in adults. Cochrane Database Syst Rev. 2009;1, CD005617. doi:10.1002/14651858.CD005617.
Rhoades CE, Gelberman RH, Manjarris JF. Stenosing tenosynovitis of the fingers and thumb. Results of a prospective trial of steroid injection and splinting. Clin Orthop Relat Res. 1984;190:236–8.
Sato ES, Gomes Dos Santos JB, Belloti JC, Albertoni WM, Faloppa F. Treatment of trigger finger: randomized clinical trial comparing the methods of corticosteroid injection, percutaneous release and open surgery. Rheumatology (Oxford). 2012;51(1):93–9. doi:10.1093/rheumatology/ker315.
Speed CA. Fortnightly review: corticosteroid injections in tendon lesions. BMJ. 2001;323(7309):382–6.
Taras JS, Raphael JS, Pan WT, Movagharnia F, Sotereanos DG. Corticosteroid injections for trigger digits: is intrasheath injection necessary? J Hand Surg [Am]. 1998;23(4):717–22.
Wang AA, Hutchinson DT. The effect of corticosteroid injection for trigger finger on blood glucose level in diabetic patients. J Hand Surg [Am]. 2006;31(6):979–81.
Acknowledgments
The authors would like to acknowledge the work of James Dowdell, BS for his help in the collection of the data presented in this study.
Conflict of Interest
Charles F. Leinberry declares he has no conflict of interest.
Emran Sheikh declares he has no conflict of interest.
Mithchell Maltenfort declares he has no conflict of interest.
Will Sayde declares he has no conflict of interest.
John D Peters declares he has no conflict of interest.
Statement of Human and Animal Rights
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2008 (5).
Statement of Informed Consent
All patients enrolled in this study gave informed consent to participate and no identifying factors were used.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Sheikh, E., Peters, J.D., Sayde, W. et al. A prospective randomized trial comparing the effectiveness of one versus two (staged) corticosteroid injections for the treatment of stenosing tenosynovitis. HAND 9, 340–345 (2014). https://doi.org/10.1007/s11552-014-9603-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11552-014-9603-4