Abstract
Background
Inflammation is a long-established hallmark of liver fibrosis and carcinogenesis. Eosinophils are emerging as crucial components of the inflammatory process influencing cancer development. The role of blood eosinophils in patients with hepatocellular carcinoma receiving systemic treatment is an unexplored field.
Objective
The objective of this study was to analyse the prognostic role of the baseline eosinophil count in patients with sorafenib-treated hepatocellular carcinoma.
Patients and Methods
A training cohort of 92 patients with advanced- or intermediate-stage sorafenib-treated hepatocellular carcinoma and two validation cohorts of 65 and 180 patients were analysed. Overall survival and progression-free survival in relation to baseline eosinophil counts were estimated by the Kaplan–Meier method. Univariate and multivariate analyses were performed.
Results
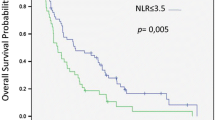
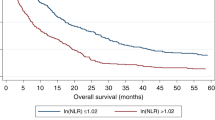
A negative prognostic impact of low baseline eosinophil counts (< 50*109/L) was demonstrated in all cohorts (training cohort: hazard ratio = 50.1, 95% confidence interval 11.6–216.5, p < 0.0001 for low vs high eosinophil counts; first validation cohort: hazard ratio = 4.55, 95% confidence interval 1.24–16.65, p = 0.022; second validation cohort: hazard ratio = 3.21, 95% confidence interval 1.83–5.64, p < 0.0001). Moreover, low eosinophil counts had a negative prognostic role in patients progressing on or intolerant to sorafenib who received second-line regorafenib, but not capecitabine or best supportive care.
Conclusions
Our analysis identified baseline blood eosinophil counts as a new prognostic factor in patients with sorafenib-treated hepatocellular carcinoma. Concerning second-line therapies, eosinophil counts were associated with survival outcomes only in regorafenib-treated patients, suggesting a possible predictive role in this setting.




Similar content being viewed by others
Change history
28 October 2020
An Online First version of this article was made available online at <ExternalRef><RefSource>https://springer.longhoe.net/article/10.1007/s11523-020-00757-3</RefSource><RefTarget Address="https://springer.longhoe.net/article/10.1007/s11523-020-00757-3" TargetType="URL"/></ExternalRef> on 12 October 2020. Errors were subsequently identified in the article, and the following corrections should be noted.
References
International Agency for Research on Cancer, World Health Organization. Cancer today. 2019. Available from: https://gco.iarc.fr/today/home. Accessed 30 Sept 2020.
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68(6):394–424.
Llovet JM, Ricci S, Mazzaferro V, Hilgard P, Gane E, Blanc J-F, et al. Sorafenib in advanced hepatocellular carcinoma. N Engl J Med. 2008;359(4):378–90.
Cheng AL, Kang Y-K, Chen Z, Tsao C-J, Qin S, Kim JS, et al. Efficacy and safety of sorafenib in patients in the Asia-Pacific region with advanced hepatocellular carcinoma: a phase III randomised, double-blind, placebo-controlled trial. Lancet Oncol. 2009;10(1):25–34.
European Association for the Study of the Liver. EASL clinical practice guidelines: management of hepatocellular carcinoma. J Hepatol. 2018;69:182–236.
Heimbach JK, Kulik LM, Finn RS, Sirlin CB, Abecassis MM, Roberts LR, et al. AASLD guidelines for the treatment of hepatocellular carcinoma. Hepatology. 2018;67:358–80.
Kudo M, Finn RS, Qin S, Han K-H, Ikeda K, Piscaglia F, et al. Lenvatinib versus sorafenib in first-line treatment of patients with unresectable hepatocellular carcinoma: a randomised phase 3 non-inferiority trial. Lancet. 2018;391(10126):1163–73.
Bruix J, Qin S, Merle P, Granito A, Huang Y-H, Bodoky G, et al. Regorafenib for patients with hepatocellular carcinoma who progressed on sorafenib treatment (RESORCE): a randomised, double-blind, placebo controlled, phase 3 trial. Lancet. 2017;389(10064):56–66.
Zhu AX, Kang Y-K, Yen C-J, Finn RS, Galle PR, Llovet JM, et al. Ramucirumab after sorafenib in patients with advanced hepatocellular carcinoma and increased α-fetoprotein concentrations (REACH-2): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2019;20(2):282–96.
Abou-Alfa GK, Meyer T, Cheng A-L, El-Khoueiry AB, Rimassa L, Ryooet B-Y, et al. Cabozantinib in patients with advanced and progressing hepatocellular carcinoma. N Engl J Med. 2018;379(1):54–63.
Tovoli F, Casadei-Gardini A, Benevento F, Piscaglia F. Immunotherapy for hepatocellular carcinoma: a review of potential new drugs based on ongoing clinical studies as of 2019. Dig Liver Dis. 2019;51(8):1067–73.
El-Khoueiry AB, Sangro B, Yau T, Crocenzi TS, Kudo M, Hsuet C, et al. Nivolumab in patients with advanced hepatocellular carcinoma (CheckMate 040): an open-label, non-comparative, phase 1/2 dose escalation and expansion trial. Lancet. 2017;389(10088):2492–502.
Zhu AX, Finn RS, Edeline J, Cattan S, Ogasawara S, Palmeret D, et al. Pembrolizumab in patients with advanced hepatocellular carcinoma previously treated with sorafenib (KEYNOTE-224): a non-randomised, open-label phase 2 trial. Lancet Oncol. 2018;19(7):940–52.
Finn RS, Ryoo B-Y, Merle P, Kudo M, Bouattour M, Lim H-Y, et al. Results of KEYNOTE-240: phase 3 study of pembrolizumab (Pembro) vs. best supportive care (BSC) for second line therapy in advanced hepatocellular carcinoma (HCC). J Clin Oncol. 2019;37(15_Suppl.):4004.
Yau T, Park JW, Finn RS, Cheng A-L, Mathurin P, Edeline J, et al. CheckMate 459: a randomized, multi-center phase III study of nivolumab (NIVO) vs sorafenib (SOR) as first-line (1L) treatment in patients (pts) with advanced hepatocellular carcinoma (aHCC). Ann Oncol. 2019;30(5_Suppl.):v874–5.
Finn RS, Qin S, Ikeda M, Galle PR, Ducreux M, Kim T-Y, et al. Atezolizumab plus bevacizumab in unresectable hepatocellular carcinoma. N Engl J Med. 2020;382(20):1894–905.
Ghouri YA, Mian I, Rowe JH. Review of hepatocellular carcinoma: epidemiology, etiology, and carcinogenesis. J Carcinog. 2017;16:1.
Sanghera C, Teh JJ, Pinato DJ. The systemic inflammatory response as a source of biomarkers and therapeutic targets in hepatocellular carcinoma. Liver Int. 2019;39(11):2008–233.
Ogunwobi OO, Harricharran T, Huaman J, Galuza A, Odumuwagun O, Tanet Y, et al. Mechanisms of hepatocellular carcinoma progression. World J Gastroenterol. 2019;25(19):2279–93.
Hernandez-Gea V, Toffanin S, Friedman SL, Llovet JM. Role of the microenvironment in the pathogenesis and treatment of hepatocellular carcinoma. Gastroenterology. 2013;144(3):512–27.
Budhu A, Wang XW. The role of cytokines in hepatocellular carcinoma. J Leukocyte Biol. 2006;80(6):1197–213.
Budhu A, Forgues M, Ye Q-H, Jia H-L, He P, Zanetti KA, et al. Prediction of venous metastases, recurrence, and prognosis in hepatocellular carcinoma based on a unique immune response signature of the liver microenvironment. Cancer Cell. 2006;10(2):99–111.
Zhou H, Huang H, Shi J, Zhao Y, Dong Q, Jia H, et al. Prognostic value of interleukin 2 and interleukin 15 in peritumoral hepatic tissues for patients with hepatitis B-related hepatocellular carcinoma after curative resection. Gut. 2010;59(12):1699–708.
Bruix J, Cheng AL, Meinhardt G, Nakajima K, De Sanctis Y, Llove L. Prognostic factors and predictors of Sorafenib benefit in patients with hepatocellular carcinoma: analysis of two phase III studies. J Hepatol. 2017;67(5):999–1008.
Da Fonseca LG, Barroso-Sousa R, Bento Ada S, Blanco BP, Valente GL, Pfiffer TE, et al. Pre-treatment neutrophil-to-lymphocyte ratio affects survival in patients with advanced hepatocellular carcinoma treated with sorafenib. Med Oncol. 2014;31:264.
Howell J, Pinato DJ, Ramaswami R, Arizumi T, Ferrari C, Gibbin A, et al. Integration of the cancer-related inflammatory response as a stratifying biomarker of survival in hepatocellular carcinoma treated with sorafenib. Oncotarget. 2017;8:36161–700.
Lué A, Serrano MT, Bustamante FJ, Iñarrairaegui M, Arenas JI, Testillano M, et al. Neutrophil-to-lymphocyte ratio predicts survival in European patients with hepatocellular carcinoma administered sorafenib. Oncotarget. 2017;8:103077–86.
Casadei Gardini A, Scarpi E, Faloppi L, Scartozzi M, Silvestris N, Santini D, et al. Immune inflammation indicators and implication for immune modulation strategies in advanced hepatocellular carcinoma patients receiving sorafenib. Oncotarget. 2016;7(41):67142–9.
Varricchi G, Galdiero MR, Loffredo S, Lucarini V, Marone G, Mattei F, et al. Eosinophils: the unsung heroes in cancer? Oncoimmunology. 2017;7(2):e1393134.
Zahoor H, Barata PC, Jia X, Martin A, Allman KD, Wood LS, et al. Patterns, predictors and subsequent outcomes of disease progression in metastatic renal cell carcinoma patients treated with nivolumab. J Immunother Cancer. 2018;6(1):107.
Rosner S, Kwong E, Shoushtari AN, Friedman CF, Betof AS, Bradye MS, et al. Peripheral blood clinical laboratory variables associated with outcomes following combination nivolumab and ipilimumab immunotherapy in melanoma. Cancer Med. 2018;7(3):690–7.
Wei Y, Zhang X, Wang G, Zhou Y, Luo M, Wang S, et al. The impacts of pretreatment circulating eosinophils and basophils on prognosis of stage I–III colorectal cancer. Asia Pac J Clin Oncol. 2018;14(5):e243–e251251.
Tanizaki J, Haratani K, Hayashi H, Chiba Y, Nakamura Y, Yonesaka K, et al. Peripheral blood biomarkers associated with clinical outcome in non-small cell lung cancer patients treated with nivolumab. J Thorac Oncol. 2018;13(1):97–105.
Holub K, Biete A. Impact of systemic inflammation biomarkers on the survival outcomes of cervical cancer patients. Clin Transl Oncol. 2019;21(7):836–44.
Holub K, Conill C. Unveiling the mechanisms of immune evasion in pancreatic cancer: may it be a systemic inflammation responsible for dismal survival? Clin Transl Oncol. 2020;22(1):81–90.
Ownby HE, Roi LD, Isenberg RR, Brennan MJ. Peripheral lymphocyte and eosinophil counts as indicators of prognosis in primary breast cancer. Cancer. 1983;52(1):126–30.
Utsunomiya A, Ishida T, Inagaki A, Ishii T, Yano H, Komatsu H, et al. Clinical significance of a blood eosinophilia in adult T-cell leukemia/lymphoma: a blood eosinophilia is a significant unfavorable prognostic factor. Leuk Res. 2007;31(7):915–20.
Hude I, Sasse S, Bröckelmann PJ, von Tresckow B, Momotow J, Engert A, et al. Leucocyte and eosinophil counts predict progression-free survival in relapsed or refractory classical Hodgkin lymphoma patients treated with PD1 inhibition. Br J Haematol. 2018;181(6):837–40.
Steel JL, Kim KH, Dew MA, Unruh ML, Antoni MH, Olek MC, et al. Cancer-related symptom clusters, eosinophils, and survival in hepatobiliary cancer: an exploratory study. J Pain Symptom Manag. 2010;39(5):859–71.
Casadei Gardini A, Marisi G, Dadduzio V, Gramantieri L, Faloppi L, Ulivi P, et al. Association of NOS3 and ANGPT2 gene polymorphisms with survival in patients with hepatocellular carcinoma receiving sorafenib: results of the multicenter prospective INNOVATE study. Clin Cancer Res. 2020;26(17):4485–93.
Casadei Gardini A, Foca F, Scartozzi M, Silvestris N, Tamburini E, Faloppi L, et al. Metronomic capecitabine versus best supportive care as second-line treatment in hepatocellular carcinoma: a retrospective study. Sci Rep. 2017;7:42499.
Pelizzaro F, Sammarco A, Dadduzio V, Pastorelli D, Giovanis P, Soldà C, et al. Capecitabine in advanced hepatocellular carcinoma: a multicenter experience. Dig Liver Dis. 2019;51(12):1713–9.
McDonald JH. Fisher’s exact test of independence. In: Handbook of biological statistics. 3rd ed. Baltimore, Maryland: Sparky House Publishing; 2014.
Gunduz S, Mutlu H, Uysal M, Coskun HS, Bozcuk H, et al. Prognostic value of hematologic parameters in patients with metastatic renal cell carcinoma using tyrosine kinase inhibitors. Asian Pac J Cancer Prev. 2014;15(8):3801–4.
Martens A, Wistuba-Hamprecht K, Geukes Foppen M, Yuan J, Postow MA, Wong P, et al. Baseline peripheral blood biomarkers associated with clinical outcome of advanced melanoma patients treated with ipilimumab. Clin Cancer Res. 2016;22(12):2908–18.
Weide B, Martens A, Hassel JC, Berking C, Postow MA, Bisschop K, et al. Baseline biomarkers for outcome of melanoma patients treated with pembrolizumab. Clin Cancer Res. 2016;22(22):5487–96.
Wang HK, Wan FN, Gu WJ, Zhu Y, Dai B, Shi G-H, et al. Eosinophil percentage elevation as a prognostic factor for overall survival in patients with metastatic renal cell carcinoma treated with tyrosine kinase inhibitor. Oncotarget. 2016;7(42):68943–53.
Gebhardt C, Sevko A, Jiang H, Lichtenberger R, Reith M, Tarnanidis K, et al. Myeloid cells and related chronic inflammatory factors as novel predictive markers in melanoma treatment with ipilimumab. Clin Cancer Res. 2015;21(24):5453–9.
Davis BP, Rothenberg ME. Eosinophils and cancer. Cancer Immunol Res. 2014;2(1):1–8.
Ramirez GA, Yacoub MR, Ripa M, Mannina D, Cariddi A, Saporiti N, et al. Eosinophils from physiology to disease: a comprehensive review. Biomed Res Int. 2018;2018:9095275.
Carretero R, Sektioglu IM, Garbi N, Salgado OC, Beckhove P, Hämmerling GJ, et al. Eosinophils orchestrate cancer rejection by normalizing tumor vessels and enhancing infiltration of CD8(+) T cells. Nat Immunol. 2015;16(6):609–17.
Shomura M, Kagawa T, Shiraishi K, Hirose S, Arase Y, Koizumi J, et al. Skin toxicity predicts efficacy to sorafenib in patients with advanced hepatocellular carcinoma. World J Hepatol. 2014;6(9):670–6.
Wang P, Tan G, Zhu M, Li W, Zhai B, Sun X, et al. Hand-foot skin reaction is a beneficial indicator of sorafenib therapy for patients with hepatocellular carcinoma: a systemic review and meta-analysis. Expert Rev Gastroenterol Hepatol. 2018;12(1):1–8.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
No external funding was used in the conduct of this study or the preparation of this article.
Conflict of Interest
Andrea Casadei-Gardini: consultant for Bayer, EISAI, AstraZeneca and Ipsen. Mario Scartozzi: advisory board for EISAI. Fabio Piscaglia: Esaote (Genova, Italia): Institutional Research Contract; Honoraria: Astrazeneca (Milano, Italia): advisory board, Bayer (Leverkusen, Germania): speaker bureau, advisory board, Bracco (Milano, Italia): speaker bureau, BMS (UK): speaker bureau, La Force Guerbet (Minneapolis, USA): speaker bureau, EISAI (Milano, Italia): speaker bureau, advisory board, GE (Milwaukee, USA): consultant, Tiziana Life Sciences (UK) advisory board, Siemens Healthcare (Germany) advisory board, Alkermes (USA) advisory board, IPSEN (Italy and Greece) speaker bureau). Giulia Orsi, Francesco Tovoli, Vincenzo Dadduzio, Caterina Vivaldi, Oronzo Brunetti, Luca Ielasi, Fabio Conti, Giulia Rovesti, Laura Gramantieri, Mario Domenico Rizzato, Irene Pecora, Antonella Argentiero, Federica Teglia, Sara Lonardi, Francesca Salani, Alessandro Granito, Vittorina Zagonel, Giorgia Marisi, Giuseppe Cabibbo, Francesco Giuseppe Foschi, Francesca Benevento, Alessandro Cucchetti and Stefano Cascinu have no conflicts of interest that are directly relevant to the content of this article.
Ethics Approval
The present research was performed in accordance with the Declaration of Helsinki (6th revision, 2008) and the study protocol was reviewed and approved by the local ethics committee (CEIIAV: comitato etico IRST IRCCS AVR), study number IRST B041, protocol number 5482/v.1 intern code: L3P1192.
Consent to Aarticipate
The study did not involve direct human participants, but only medical records of patients. All participants provided written informed consent to authorise the use of their medical data.
Consent for Publication
The authors declare that the manuscript has not been published previously, in whole or in part, and is not under consideration for publication elsewhere. All authors have approved the manuscript and consent to its publication.
Availability of Data and Material
The datasets generated and/or analysed during the current study are available from the corresponding author on reasonable request.
Author Contributions
Study concept and design: ACG, GO. Data collection: GO, FT, VD, CV, OB, LI, FC, GR, LG, MDR, IP, AA, FT, SL, FS, AG, VZ, GM, GC, FGF, AC, FP, SC, MS, ACG. Experiments and procedures: ACG, GO. Writing and critical revision of the draft: GO, FT, VD, CV, OB, LI, FC, GR, LG, MDR, IP, AA, FT, SL, FS, AG, VZ, GM, GC, FGF, FB, AC, FP, SC, MS, ACG.Study concept and design: ACG, GO. Data collection: GO, FT, VD, CV, OB, LI, FC, GR, LG, MDR, IP, AA, FT, SL, FS, AG, VZ, GM, GC, FGF, AC, FP, SC, MS, ACG. Experiments and procedures: ACG, GO. Writing and critical revision of the draft: GO, FT, VD, CV, OB, LI, FC, GR, LG, MDR, IP, AA, FT, SL, FS, AG, VZ, GM, GC, FGF, FB, AC, FP, SC, MS, ACG.
Additional information
The original article has been updated: Due to Author affiliation update.
Rights and permissions
About this article
Cite this article
Orsi, G., Tovoli, F., Dadduzio, V. et al. Prognostic Role of Blood Eosinophil Count in Patients with Sorafenib-Treated Hepatocellular Carcinoma. Targ Oncol 15, 773–785 (2020). https://doi.org/10.1007/s11523-020-00757-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11523-020-00757-3