Abstract
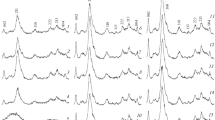
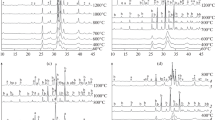
The formation of dipeptides from amino acids can be driven by hydroxyapatite at a relatively low temperature in air. For example, the formation of (Ala)2 from Ala is induced on hydroxyapatite at 110°C with considerable yield. Typically, condensing agents, high temperatures (>250°C) or high pressures (>25 MPa) are required to drive the condensation of amino acids. Similar effects are observed in the condensation of Gly, Glu and Asp. Experiments demonstrate that hydroxyapatite is an effective inorganic catalytic agent, reducing the activation barrier for the formation of dipeptides by more than 50%. HAP promotes condensation by adsorbing amino acid monomers in an organized manner, which decreases the distance between amino and carboxyl groups on neighboring molecules and extends the contact time of the reaction groups. This work provides a chemical understanding of the primitive condensation of amino acids and reveals a mechanism for enhancement of mineral catalysts. It is important that the conditions used for hydroxyapatite-assisted dipeptide formation are not harsh and can be readily achieved, revealing a possible mechanism for the chemical evolution of biomolecules over geologic ages.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Eigen M. Molecular self organisation and evolution. Naturwissenschaften, 1971, 58: 465–523
Matsuno K. Natural self-organization of polynucleotides and polypeptides in protobiogenesis: Appearance of a protohypercycle. Bio-Systems, 1982, 15: 1–11
Kauffman S A. Autocatalytic sets of proteins. J Theor Biol, 1986, 119: 1–24
Orgel L E. Mini review: RNA catalysis and the origins of life. Ibid, 1986, 123: 127–149
Bada J L, Lazcano A. Some like it hot, but not the first biomolecules. Science, 2002, 296: 1982–1983
Brack A. From interstellar amino acids to prebiotic catalytic peptides: A review. Chem Biodiv, 2007, 4: 665–679
Schoonen M, Smirnov A, Cohn C. A perspective on the role of minerals in prebiotic synthesis. Ambio, 2004, 33: 539–551
Lahav N. Minerals and the origin of life — hypotheses and experiments in heterogeneous chemistry. Heterogen Chem Rev, 1994, 1: 159–179
Joshi P C, Pitsch S, Ferris J P. Homochiral selection in the montmorillonite-catalyzed and uncatalyzed prebiotic synthesis of RNA. Chem Commun, 2000: 2497–2498
Bujdák J, Eder A, Yongyai Y, et al. Investigation on the mechanism of peptide chain prolongation on montmorillonite. J Inorg Biochem, 1996, 61: 69–78
Bujdák J, Son H L, Rode B M. Montmorillonite catalyzed peptide bond formation: The effect of exchangeable cations. J Inorg Biochem, 1996, 63: 119–124
Basiuk V A, Gromovoy T Y, Golovaty V G, et al. Mechanisms of amino acid polycondensation on silica and alumina surfaces. Orig Life Evol Biosphere, 1990, 20: 483–498
Ferris J P, Hill A R Jr, Liu R H, et al. Synthesis of long prebiotic oligomers on mineral surfaces. Nature, 1996, 381: 59–61
Imai E, Honda H, Hatori K, et al. Elongation of oligopeptides in a simulated submarine hydrothermal system. Science, 1999, 283: 831–833
Dorozhkin S V, Epple M. Biological and medical significance of calcium phosphates. Angew Chem Int Ed, 2002, 41: 3130–3146
Koutsopoulos S, Dalas E. Inhibition of hydroxyapatite formation in aqueous solutions by amino acids with hydrophobic side groups. Langmuir, 2000, 16: 6739–6744
Pan H, Tao J, Xu X, et al. Adsorption processes of gly and glu amino acids on hydroxyapatite surfaces at the atomic level. Langmuir, 2007, 23: 8972–8981
Tanaka H, Miyajima K, Nakagaki M, et al. Interactions of aspartic acid, alanine and lysine with hydroxyapatite. Chem Pharmac Bull, 1989, 37: 2897–2901
Churchill H, Teng H, Hazen R M. Correlation of pH-dependent surface interaction forces to amino acid adsorption: Implications for the origin of life. Am Mineral, 2004, 89: 1048–1055
Fujisawa R, Wada Y, Nodasaka Y, et al. Acidic amino acid-rich sequences as binding sites of osteonectin to hydroxyapatite crystals. Biochim Biophys Acta, 1996, 1292: 53–60
Hill A R Jr, Böhler C, Orgel L E. Polymerization on the rocks: Negatively — charged α-amino acids. Orig Life Evol Biosphere, 1998, 28: 235–243
Capriotti L A, Beebe T P Jr, Schneider J P. Hydroxyapatite surface-induced peptide folding. J Am Chem Soc, 2007, 129: 5281–5287
Kafarov V. Cybernetic Methods in Chemistry and Chemical Engineering. Moscow: Mir Pub, 1976.
Kawamura K, Nishi T, Sakiyama T. Consecutive elongation of alanine oligopeptides at the second time range under hydrothermal conditions using a microflow reactor system. J Am Chem Soc, 2005, 127: 522–523
Ferris J P. Chemical markers of prebiotic chemistry in hydrothermal systems. Orig Life Evol Biosphere, 1992, 22: 109–134
Amend J P, Shock E L. Energetics of amino acid synthesis in hydrothermal ecosystems. Science, 1998, 281: 1659–1662
Lahav N, White D, Chang S. Peptide formation in the prebiotic era: Thermal condensation of glycine in fluctuating clay environments. Science, 1978, 201: 67–69
Saetia S, Liedl K R, Eder A H, et al. Evaporation cycle experiments—a simulation of salt-induced peptide synthesis under possible prebiotic conditions. Orig Life Evol Biosphere, 1993, 23: 167–176
Rode B M, Son H L, Suwannachot Y. The combination of salt induced peptide formation reaction and clay catalysis: A way to higher peptides under primitive earth conditions. Orig Life Evol Biosphere, 1999, 29: 273–286
Hauptmann S, Dufner H, Brickmann J, et al. Potential energy function for apatites. Phys Chem Chem Phys, 2003, 5: 635–639
Berendsen H, van der Spoel D, van Drunen R. Gromacs: A message-passing parallel molecular dynamics implementation. Comp Phys Commun, 1995, 91: 43–56
Lindahl E, Hess B, van der Spoel D. Gromacs 3.0: A package for molecular simulation and trajectory analysis. J Mol Model, 2001, 7: 306–317
Frenkel D, Smit B. Understanding Molecular Simulations. San Diego: Academic Press, 1996
Orgel L E. The origin of polynucleotide-directed protein synthesis. J Mol Evol, 1989, 29: 465–474
Rode B M, Schwendinger M G. Copper-catalyzed amino acid condensation in water—a simple possible way of prebiotic peptide formation. Orig Life Evol Biosphere, 1990, 20: 401–410
Nagayama M, Takaoka O, Inomata K, et al. Diketopiperazine-mediated peptide formation in aqueous solution. Orig Life Evol Biosphere, 1990, 20: 249–257
Cox J S, Seward T M. The reaction kinetics of alanine and glycine under hydrothermal conditions. Geochim Cosmochim Acta, 2007, 71: 2264–2284
Rode B M, Eder A H, Yongyai Y. Amino acid sequence preferences of the salt-induced peptide formation reaction in comparison to archaic cell protein composition. Inorg Chim Acta, 1997, 254: 309–314
Rimola A, Sodupe M, Ugliengo P. Aluminosilicate surfaces as promoters for peptide bond formation: An assessment of bernal’s hypothesis by ab initio methods. J Am Chem Soc, 2007, 129: 8333–8344
Rimola A, Tosoni S, Sodupe M, et al. Does silica surface catalyze peptide bond formation? New insights from first-principles calculations. Chem Phys Chem, 2006, 7: 157–163
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Wu, J., Zhang, Z., Yu, X. et al. Mechanism of promoted dipeptide formation on hydroxyapatite crystal surfaces. Chin. Sci. Bull. 56, 633–639 (2011). https://doi.org/10.1007/s11434-010-4314-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11434-010-4314-x