Abstract
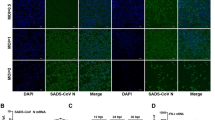
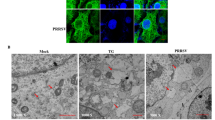
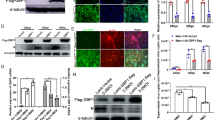
Although the functional parameters of microRNAs (miRNAs) have been explored to some extent, the roles of these molecules in coronavirus infection and the regulatory mechanism of miRNAs in virus infection are still unclear. Transmissible gastroenteritis virus (TGEV) is an enteropathgenic coronavirus and causes high morbidity and mortality in suckling piglets. Here, we demonstrated that microRNA-27b-3p (miR-27b-3p) suppressed TGEV replication by directly targeting porcine suppressor of cytokine signaling 6 (SOCS6), while TGEV infection downregulated miR-27b-3p expression in swine testicular (ST) cells and in piglets. Mechanistically, the decrease of miR-27b-3p expression during TGEV infection was mediated by the activated inositol-requiring enzyme 1 (IRE1) pathway of the endoplasmic reticulum (ER) stress. Further studies showed that when ER stress was induced by TGEV, IRE1 acted as an RNase activated by autophosphorylation and unconventionally spliced mRNA encoding a potent transcription factor, X-box-binding protein 1 (Xbp1s). Xbp1s inhibited the transcription of miR-27 and ultimately reduced the production of miR-27b-3p. Therefore, our findings indicate that TGEV inhibits the expression of an anti-coronavirus microRNA through the IRE1 pathway and suggest a novel way in which coronavirus regulates the host cell response to infection.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bail, S., Swerdel, M., Liu, H., Jiao, X., Goff, L.A., Hart, R.P., and Kiledjian, M. (2010). Differential regulation of microRNA stability. RNA 16, 1032–1039.
Bartel, D.P. (2004). MicroRNAs: genomics, biogenesis, mechanism, and function. Cell 116, 281–297.
Berry, C., Lal, M., and Binukumar, B.K. (2018). Crosstalk between the unfolded protein response, microRNAs, and insulin signaling pathways: in search of biomarkers for the diagnosis and treatment of type 2 diabetes. Front Endocrinol 9, 210.
Bhattacharyya, S., Sen, U., and Vrati, S. (2014). Regulated IRE1-dependent decay pathway is activated during Japanese encephalitis virus-induced unfolded protein response and benefits viral replication. J Gen Virol 95, 71–79.
Bookout, A.L., Cummins, C.L., Mangelsdorf, D.J., Pesola, J.M., and Kramer, M.F. (2006). High-throughput real-time quantitative reverse transcription PCR. Curr Protoc Mol Biol 73.
Buck, A.H., Perot, J., Chisholm, M.A., Kumar, D.S., Tuddenham, L., Cognat, V., Marcinowski, L., Dolken, L., and Pfeffer, S. (2010). Post-transcriptional regulation of miR-27 in murine cytomegalovirus infection. RNA 16, 307–315.
Calfon, M., Zeng, H., Urano, F., Till, J.H., Hubbard, S.R., Harding, H.P., Clark, S.G., and Ron, D. (2002). IRE1 couples endoplasmic reticulum load to secretory capacity by processing the XBP-1 mRNA. Nature 415, 92–96.
Cameron, J.E., Fewell, C., Yin, Q., McBride, J., Wang, X., Lin, Z., and Flemington, E.K. (2008a). Epstein-Barr virus growth/latency III program alters cellular microRNA expression. Virology 382, 257–266.
Cameron, J.E., Yin, Q., Fewell, C., Lacey, M., McBride, J., Wang, X., Lin, Z., Schaefer, B.C., and Flemington, E.K. (2008b). Epstein-Barr virus latent membrane protein 1 induces cellular microRNA miR-146a, a modulator of lymphocyte signaling pathways. J Virol 82, 1946–1958.
Credle, J.J., Finer-Moore, J.S., Papa, F.R., Stroud, R.M., and Walter, P. (2005). On the mechanism of sensing unfolded protein in the endoplasmic reticulum. Proc Natl Acad Sci USA 102, 18773–18784.
Ding, L., Ni, J., Yang, F., Huang, L., Deng, H., Wu, Y., Ding, X., and Tang, J. (2017). Promising therapeutic role of miR-27b in tumor. Tumour Biol 39, 101042831769165.
Doyle, L.P., and Hutchings, L.M. (1946). A transmissible gastroenteritis in pigs. J Am Vet Med Assoc 108, 257–259.
Du, H., Cui, S., Li, Y., Yang, G., Wang, P., Fikrig, E., and You, F. (2018). miR-221 negatively regulates innate anti-viral response. PLoS ONE 13, e0200385.
Fehr, A.R., and Perlman, S. (2015). Coronaviruses: an overview of their replication and pathogenesis. Methods Mol Biol 1282, 1–23.
Fink, S.L., Jayewickreme, T.R., Molony, R.D., Iwawaki, T., Landis, C.S., Lindenbach, B.D., and Iwasaki, A. (2017). IRE1α promotes viral infection by conferring resistance to apoptosis. Sci Signal 10, eaai7814.
Forster, S.C., Tate, M.D., and Hertzog, P.J. (2015). MicroRNA as type I interferon-regulated transcripts and modulators of the innate immune response. Front Immunol 6, 334.
Fung, T.S., Huang, M., and Liu, D.X. (2014). Coronavirus-induced ER stress response and its involvement in regulation of coronavirus-host interactions. Virus Res 194, 110–123.
Fung, T.S., and Liu, D.X. (2014). Coronavirus infection, ER stress, apoptosis and innate immunity. Front Microbiol 5, 296.
Gatto, G., Rossi, A., Rossi, D., Kroening, S., Bonatti, S., and Mallardo, M. (2008). Epstein-Barr virus latent membrane protein 1 trans-activates miR-155 transcription through the NF-κB pathway. Nucleic Acids Res 36, 6608–6619.
Hassan, I.H., Zhang, M.S., Powers, L.S., Shao, J.Q., Baltrusaitis, J., Rutkowski, D.T., Legge, K., and Monick, M.M. (2012). Influenza A viral replication is blocked by inhibition of the inositol-requiring enzyme 1 (IRE1) stress pathway. J Biol Chem 287, 4679–4689.
Hassler, J., Cao, S.S., and Kaufman, R.J. (2012). IRE1, a double-edged sword in pre-miRNA slicing and cell death. Dev Cell 23, 921–923.
Heindryckx, F., Binet, F., Ponticos, M., Rombouts, K., Lau, J., Kreuger, J., and Gerwins, P. (2016). Endoplasmic reticulum stress enhances fibrosis through IRE1α-mediated degradation of miR-150 and XBP-1 splicing. EMBO Mol Med 8, 729–744.
Huang, J., Wang, F., Argyris, E., Chen, K., Liang, Z., Tian, H., Huang, W., Squires, K., Verlinghieri, G., and Zhang, H. (2007). Cellular microRNAs contribute to HIV-1 latency in resting primary CD4+ T lymphocytes. Nat Med 13, 1241–1247.
Kai, Z.S., and Pasquinelli, A.E. (2010). MicroRNA assassins: factors that regulate the disappearance of miRNAs. Nat Struct Mol Biol 17, 5–10.
Lecellier, C.H., Dunoyer, P., Arar, K., Lehmann-Che, J., Eyquem, S., Himber, C., Saib, A., and Voinnet, O. (2005). A cellular microRNA mediates antiviral defense in human cells. Science 308, 557–560.
Lee, A.H., Iwakoshi, N.N., and Glimcher, L.H. (2003). XBP-1 regulates a subset of endoplasmic reticulum resident chaperone genes in the unfolded protein response. Mol Cell Biol 23, 7448–7459.
Ma, Y., Wang, C., Xue, M., Fu, F., Zhang, X., Li, L., Yin, L., Xu, W., Feng, L., and Liu, P. (2018). The coronavirus transmissible gastroenteritis virus evades the type I interferon response through IRE1alpha-mediated manipulation of the microRNA miR-30a-5p/SOCS1/3 axis. J Virol 92.
Mahajan, V.S., Drake, A., and Chen, J. (2009). Virus-specific host miRNAs: antiviral defenses or promoters of persistent infection? Trends Immunol 30, 1–7.
Mallick, B., Ghosh, Z., and Chakrabarti, J. (2009). MicroRNome analysis unravels the molecular basis of SARS infection in bronchoalveolar stem cells. PLoS ONE 4, e7837.
O’Connell, R.M., Taganov, K.D., Boldin, M.P., Cheng, G., and Baltimore, D. (2007). MicroRNA-155 is induced during the macrophage inflammatory response. Proc Natl Acad Sci USA 104, 1604–1609.
O’Neill, L.A., Sheedy, F.J., and McCoy, C.E. (2011). MicroRNAs: the fine-tuners of Toll-like receptor signalling. Nat Rev Immunol 11, 163–175.
Peng, X., Gralinski, L., Ferris, M.T., Frieman, M.B., Thomas, M.J., Proll, S., Korth, M.J., Tisoncik, J.R., Heise, M., Luo, S., et al. (2011). Integrative deep sequencing of the mouse lung transcriptome reveals differential expression of diverse classes of small RNAs in response to respiratory virus infection. mBio 2.
Potenza, N., Papa, U., Mosca, N., Zerbini, F., Nobile, V., and Russo, A. (2011). Human microRNA hsa-miR-125a-5p interferes with expression of hepatitis B virus surface antigen. Nucleic Acids Res 39, 5157–5163.
Pritchard, G. (1987). Transmissible gastroenteritis in endemically infected breeding herds of pigs in East Anglia, 1981–85. Vet Record 120, 226–230.
Rivera-Ramírez, E., Méndez, L.B., Ortíz-Rivera, A., Rodríguez-Cotto, R.I., and Jiménez-Vélez, B. (2019). A novel role of PM extracts on the post-transcriptional control of pro-inflammatory mediators, IL-6 and CXCL8. Atmosphere 10, 270.
van Rooij, E., and Olson, E.N. (2007). MicroRNAs: powerful new regulators of heart disease and provocative therapeutic targets. J Clin Invest 117, 2369–2376.
Xue, M., Fu, F., Ma, Y., Zhang, X., Li, L., Feng, L., and Liu, P. (2018). The PERK arm of the unfolded protein response negatively regulates transmissible gastroenteritis virus replication by suppressing protein translation and promoting type I interferon production. J Virol 92.
Yu, Z., and Hecht, N.B. (2008). The DNA/RNA-binding protein, translin, binds microRNA122a and increases its in vivo stability. J Androl 29, 572–579.
Zhang, K., Liu, H., Song, Z., Jiang, Y., Kim, H., Samavati, L., Nguyen, H. M., and Yang, Z.Q. (2020). The UPR transducer IRE1 promotes breast cancer malignancy by degrading tumor suppressor microRNAs. iScience 23, 101503.
Zhao, X., Song, X., Bai, X., Fei, N., Huang, Y., Zhao, Z., Du, Q., Zhang, H., Zhang, L., and Tong, D. (2016). miR-27b attenuates apoptosis induced by transmissible gastroenteritis virus (TGEV) infection via targeting runt-related transcription factor 1 (RUNX1). PeerJ 4, e1635.
Zheng, H., Xu, L., Liu, Y., Li, C., Zhang, L., Wang, T., Zhao, D., Xu, X., and Zhang, Y. (2018). MicroRNA-221-5p inhibits porcine epidemic diarrhea virus replication by targeting genomic viral RNa and activating the NF-κB pathway. Int J Mol Sci 19, 3381.
Acknowledgements
This work was supported by the Heilongjiang Postdoctoral fund (LBH-Z18207), the National Natural Science Foundation of China (31802198), the Fundamental Research Funds for the Provincial Universities (2018-KYYWF-0553), the National Key Research and Development Program of China (2017YFC0908001) and the Spark Research Fund from the Fourth Affiliated Hospital of Harbin Medical University (HYDSYXH201914).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Compliance and ethics The author(s) declare that they have no conflict of interest.
Electronic supplementary material
11427_2021_1967_MOESM1_ESM.docx
Supplement Fig 1. The promoter sequence of pri-miR-27b. The transcription start site (TSS) was C and was marked in bold.
Rights and permissions
About this article
Cite this article
Wang, C., Xue, M., Wu, P. et al. Coronavirus transmissible gastroenteritis virus antagonizes the antiviral effect of the microRNA miR-27b via the IRE1 pathway. Sci. China Life Sci. 65, 1413–1429 (2022). https://doi.org/10.1007/s11427-021-1967-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-021-1967-x