Abstract
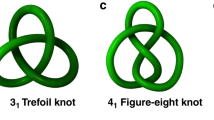
Using the strategy of ligand fine-tuning by steric hindrance, we successfully obtained Solomon links (\(4_1^2\)) and figure-eight knots (41) with half-sandwich organometallic unit and amino-acid embedded ligands. The two curved bidentate ligands exhibit subtle differences, whereas they result in totally distinct entanglement modes. An alcoholysis reaction with the ligands leads to the formation of a molecular tweezer. Notably, unsymmetrical ligands were utilized in the self-assembly process to explore the formation of directional molecules, and the reactions exhibited selectivity due to comprehensive π interactions and multiple hydrogen bonds. The topologies and behavior of the above structures were confirmed through single-crystal X-ray diffraction, nuclear magnetic resonance techniques and mass spectrometry.

Similar content being viewed by others
References
Dietrich-Buchecker CO, Sauvage JP, Kintzinger JP. Tetrahedron Lett, 1983, 24: 5095–5098
Gil-Ramírez G, Leigh DA, Stephens AJ. Angew Chem Int Ed, 2015, 54: 6110–6150
Sawada T, Yamagami M, Ohara K, Yamaguchi K, Fujita M. Angew Chem Int Ed, 2016, 55: 4519–4522
Xu L, Zhang WB. Sci China Chem, 2018, 61: 3–16
Oka Y, Masai H, Terao J. Angew Chem Int Ed, 2023, 62: e202217002
Qiu XT, Yao R, Zhou WF, Liu MD, Liu Q, Song YL, Young DJ, Zhang WH, Lang JP. Chem Commun, 2018, 54: 4168–4171
Shen YR, Gao X, Cui Z, ** GX. Chin J Chem, 2021, 39: 3303–3308
Pentecost CD, Chichak KS, Peters AJ, Cave GW, Cantrill SJ, Stoddart JF. Angew Chem Int Ed, 2007, 46: 218–222
Ponnuswamy N, Cougnon FBL, Pantoş GD, Sanders JKM. J Am Chem Soc, 2014, 136: 8243–8251
Beves JE, Danon JJ, Leigh DA, Lemonnier JF, Vitorica-Yrezabal IJ. Angew Chem Int Ed, 2015, 54: 7555–7559
Song YH, Singh N, Jung J, Kim H, Kim EH, Cheong HK, Kim Y, Chi KW. Angew Chem Int Ed, 2016, 55: 2007–2011
Chichak KS, Cantrill SJ, Pease AR, Chiu SH, Cave GWV, Atwood JL, Stoddart JF. Science, 2004, 304: 1308–1312
Guo X, Lin E, Gao J, Mao T, Yan D, Cheng P, Ma S, Chen Y, Zhang Z. Angew Chem Int Ed, 2021, 60: 2974–2979
Castle T, Evans ME, Hyde ST. New J Chem, 2008, 32: 1484–1492
Zhao X, Wang H, Li B, Zhang W, Li X, Zhao W, Janiak C, Heard AW, Yang XJ, Wu B. Angew Chem Int Ed, 2022, 61: e202115042
Dang LL, Zhang TT, Chen T, Zhao Y, Gao X, Aznarez F, Ma LF, ** GX. Angew Chem Int Ed, 2023, 62: e202301516
Liu XR, Cui PF, Guo ST, Lin YJ, ** GX. J Am Chem Soc, 2023, 51: 7779–7809
Horner KE, Miller MA, Steed JW, Sutcliffe PM. Chem Soc Rev, 2016, 45: 6432–6448
Ashbridge Z, Fielden SDP, Leigh DA, Pirvu L, Schaufelberger F, Zhang L. Chem Soc Rev, 2022, 51: 7779–7809
Jamieson EMG, Modicom F, Goldup SM. Chem Soc Rev, 2018, 47: 5266–5311
Caprice K, Pál D, Besnard C, Galmés B, Frontera A, Cougnon FBL. J Am Chem Soc, 2021, 143: 11957–11962
Rodríguez-Rubio A, Savoini A, Modicom F, Butler P, Goldup SM. J Am Chem Soc, 2022, 144: 11927–11932
McConnell AJ, Wood CS, Neelakandan PP, Nitschke JR. Chem Rev, 2015, 115: 7729–7793
Wang Y, Gong J, Wang X, Li WJ, Wang XQ, He X, Wang W, Yang HB. Angew Chem Int Ed, 2022, 61: e202210542
Liu JX, Chen K, Redshaw C. Chem Soc Rev, 2023, 52: 1428–1455
Goldup SM. Nat Chem, 2016, 8: 404–406
Marcos V, Stephens AJ, Jaramillo-Garcia J, Nussbaumer AL, Woltering SL, Valero A, Lemonnier JF, Vitorica-Yrezabal IJ, Leigh DA. Science, 2016, 352: 1555–1559
Mitra R, Zhu H, Grimme S, Niemeyer J. Angew Chem Int Ed, 2017, 56: 11456–11459
Martinez-Cuezva A, Saura-Sanmartin A, Alajarin M, Berna J. ACS Catal, 2020, 10: 7719–7733
Mo X, Deng Y, Lai SKM, Gao X, Yu HL, Low KH, Guo Z, Wu HL, Au-Yeung HY, Tse ECM. J Am Chem Soc, 2023, 145: 6087–6099
Zhang L, Qiu Y, Liu WG, Chen H, Shen D, Song B, Cai K, Wu H, Jiao Y, Feng Y, Seale JSW, Pezzato C, Tian J, Tan Y, Chen XY, Guo QH, Stern CL, Philp D, Astumian RD, Goddard Iii WA, Stoddart JF. Nature, 2023, 613: 280–286
Jiao Y, Stoddart JF. Matter, 2021, 4: 2582–2584
Zhang ZH, Andreassen BJ, August DP, Leigh DA, Zhang L. Nat Mater, 2022, 21: 275–283
Spence GT, Beer PD. Acc Chem Res, 2013, 46: 571–586
Lewis JEM, Beer PD, Loeb SJ, Goldup SM. Chem Soc Rev, 2017, 46: 2577–2591
Inthasot A, Tung ST, Chiu SH. Acc Chem Res, 2018, 51: 1324–1337
Gao WX, Feng HJ, Guo BB, Lu Y, ** GX. Chem Rev, 2020, 120: 6288–6325
Alexander JW, Briggs GB. Ann Math, 1926, 28: 562–586
Zhang HN, Lin YJ, ** GX. Chem Asian J, 2021, 16: 1918–1924
Inomata Y, Sawada T, Fujita M. Chem, 2020, 6: 294–303
Inomata Y, Sawada T, Fujita M. J Am Chem Soc, 2021, 143: 16734–16739
Cui Z, Mu QS, Gao X, ** GX. J Am Chem Soc, 2023, 145: 725–731
Itabashi H, Datta S, Tsukuda R, Hollamby MJ, Yagai S. Chem Sci, 2023, 14: 3270–3276
Gao WX, Feng HJ, Lin YJ, ** GX. J Am Chem Soc, 2019, 141: 9160–9164
Zhang HN, Yu WB, Lin YJ, ** GX. Angew Chem Int Ed, 2021, 60: 15466–15471
Acknowledgements
This work was supported by the National Natural Science Foundation of China (22031003, 21720102004) and Shanghai Science Technology Committee (19DZ2270100).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest The authors declare no conflict of interest.
Additional information
Supporting information The supporting information is available online at http://chem.scichina.com and http://springer.longhoe.net/journal/11426. The supporting materials are published as submitted, without typesetting or editing. The responsibility for scientific accuracy and content remains entirely with the authors.
Supporting Information
Rights and permissions
About this article
Cite this article
Mu, QS., Gao, X., Cui, Z. et al. Selective construction of molecular Solomon links and figure-eight knots by fine-tuning unsymmetrical ligands. Sci. China Chem. 66, 2885–2891 (2023). https://doi.org/10.1007/s11426-023-1675-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-023-1675-0