Abstract
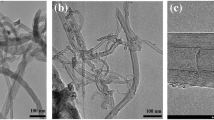
Catalysts based on Ni and Fe nanoparticles deposited selectively on carbon nanotubes were investigated before and after the dry methane reforming. Three catalysts were synthesized and evaluated, varying the concentration of Ni inside and Fe outside the carbon tubes. BET analysis revealed that the acid treatment opened the ends of the nanotubes and resulted in a higher surface area. Transmission electron microscopy (TEM) showed 24 layers with inner diameter ranging from 4 to 6 nm and outer diameter ranging from 16 to 22 nm. Raman spectroscopy confirmed that after calcination at high temperature the structure of the nanotubes was maintained. X-ray diffraction (XRD) analysis of the catalysts confirmed the presence of NiO (2.6–3.2 nm) and Fe2O3 (4.3 nm) crystallites. The catalytic tests presented high activity in dry methane reform (DRM). The catalysts 10Ni@CNT and 10Ni@CNT/5Fe presented conversions of CH4 (63 and 67%) and CO2 (72 and 88%), respectively, at 800 °C, under atmospheric pressure. Analysis after the reaction showed an increasing ratio of ID/IG, which indicates the formation of defects. The Raman analysis showed that even after calcination at high temperatures the structures of the nanotubes were mostly preserved, and TEM images showed that during the reaction, there were formation of nanotubes occurring randomly.
Graphical Abstract














Similar content being viewed by others
Data availability
The datasets generated during the current study are available from the corresponding author.
References
Abdullah B, Ghani NAA, Vo D-VN (2017) Recent advances in dry reforming of methane over Ni-based catalysts. J Clean Prod 162:170–185. https://doi.org/10.1016/j.jclepro.2017.05.176
Al-Fatesh AS, Arafat Y, Kasim SO, Ibrahim AA, Abasaeed AE, Fakeeha AH (2021) In situ auto-gasification of coke deposits over a novel Ni-Ce/W-Zr catalyst by sequential generation of oxygen vacancies for remarkably stable syngas production via CO2-reforming of methane. Appl Catal B 280:119445. https://doi.org/10.1016/j.apcatb.2020.119445
Alipour Z, Rezaei M, Meshkani F (2014) Effect of Ni loadings on the activity and coke formation of MgO-modified Ni/Al2O3 nanocatalyst in dry reforming of methane. J Energy Chem 23:633–638. https://doi.org/10.1016/S2095-4956(14)60194-7
Augusto E (2016) Acordo de Paris.http://www.mma.gov.br/clima/convencao-das-nacoes-unidas/acordo-de-paris. Accessed 1 Aug 2018
Bian Z, Das S, Wai MH, Hongmanorom P, Kawi S (2017) A review on bimetallic nickel-based catalysts for CO2 reforming of methane. Chem Phys Chem 18(22):3117–3134. https://doi.org/10.1002/cphc.201700529
Costa S, Borowiak-Palen E, Kruszyńska M, Bachmatiuk A, Kaleńczuk RJ (2008) Characterization of carbon nanotubes by Raman spectroscopy. Mater Sci - Poland 26(2):433–441
Díaz K, García V, Matos J (2007) Activated carbon supported Ni–Ca: influence of reaction parameters on activity and stability of catalyst on methane reformation. Fuel 86(9):1337–1344. https://doi.org/10.1016/J.FUEL.2006.05.011
Drif A, Bion N, Brahmi R, Ojala S, Pirault-Roy L, Turpeinen E, Seelam PK, Keiski RL, Epron F (2015) Study of the dry reforming of methane and ethanol using Rh catalysts supported on doped alumina. Appl Catal A 504:576–584. https://doi.org/10.1016/j.apcata.2015.02.019
Figueira CE, Moreira Júnior PF, Giudici R, Alves RMB, Schmal M (2018) Nanoparticles of Ce, Sr, Co in and out the multi-walled carbon nanotubes applied for dry reforming of methane. Appl Catal A 550:297–307. https://doi.org/10.1016/j.apcata.2017.11.019
Grace T, Yu L, Gibson C, Tune D, Alturaif H, Othman ZA, Shapter J (2016) Investigating the effect of carbon nanotube diameter and wall number in carbon nanotube/silicon heterojunction solar cells. Nanomaterials 6(3):5–9. https://doi.org/10.3390/nano6030052
Haghighi M, Sun Z-q, Wu J-h, Bromly J, Wee HL, Ng E, Wang Y, Zhang D-k (2007) On the reaction mechanism of CO2 reforming of methane over a bed of coal char. Proc Combust Inst 31(2):1983–1990. https://doi.org/10.1016/j.proci.2006.07.029
Inui T (2001) Rapid catalytic reforming of methane with CO2 and its application to other reactions. Appl Organomet Chem 15(2):87–94
Izquierdo U, Barrio VL, Requies J, Cambra JF, Güemez MB, Arias PL (2013) Tri-reforming: a new biogas process for synthesis gas and hydrogen production. Int J Hydrog Energy 38(18):7623–7631. https://doi.org/10.1016/j.ijhydene.2012.09.107
Jabbour K, El Hassan N, Davidson A, Massiani P, Casale S (2015) Characterizations and performances of Ni/diatomite catalysts for dry reforming of methane. Chem Eng J 264:351–358. https://doi.org/10.1016/j.cej.2014.11.109
Kalai DY (2015) Dry reforming of methane: catalyst development and thermodynamic analysis. Master thesis. University of Stavanger. http://hdl.handle.net/11250/301310
Kim SM, Abdala PM, Margossian T, Hosseini D, Foppa L, Armutlulu A, van Beek W, Comas-Vives A, Copéret C, Müller C (2017) Cooperativity and dynamics increase the performance of NiFe dry reforming catalysts. J Am Chem Soc 139(5):1937–1949. https://doi.org/10.1021/jacs.6b11487
Koike M, Li D, Nakagawa Y, Tomishige K (2012) A. Highly active and coke-resistant steam reforming catalyst comprising uniform nickel-iron alloy nanoparticles. ChemSusChem 5(12):2312–2314. https://doi.org/10.1002/cssc.201200507
Lobo AO, Martin AA, Antunes EF, Trava-Airoldi VJ, Corat EJ (2005) Caracterização de materiais carbonosos por espectroscopia Raman. Revista Brasileira de Aplicações de Vácuo 24(2):98–103. https://doi.org/10.17563/rbav.v24i2.99
Ma Q, Wang D, Wu M, Zhao T-S (2013) Effect of catalytic site position: Nickel nanocatalyst selectively loaded inside or outside carbon nanotubes for methane dry reforming. Fuel 108:430–438. https://doi.org/10.1016/j.fuel.2012.12.028
Mariotti D, Švrček V, Mathur A, Dickinson C, Matsubara K, Kondo M (2013) Carbon nanotube growth activated by quantum-confined silicon nanocrystals. J Phys D Appl Phys 46(12):122001. https://doi.org/10.1088/0022-3727/46/12/122001
Monthioux M, Flahaut E, Laurent C, Escoffier W, Raquet B, Bacsa W, Puech P, Machado B, Serp P (2014) Properties of carbon nanotubes. In: Bhushan B, Luo D, Schricker S, Sigmund W, Zauscher S (eds) Handbook of Nanomaterials Properties. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-31107-9_24
Nakagawa K, Kikuchi M, Nishitani-Gamo M, Oda H, Gamo H, Ogawa K, Ando T (2008) CO2 Reforming of CH4 over co/oxidized diamond catalyst. Energy Fuels 22(6):3566–3570. https://doi.org/10.1021/ef800265x
Rouibah K, Barama A, Benrabaa R, Guerrero-Caballero J, Kane T, Vannier R-N, Rubbens A, Löfberg A (2017) Dry reforming of methane on nickel-chrome, nickel-cobalt and nickel-manganese catalysts. Int J Hydrog Energy 42(50):29725–29734. https://doi.org/10.1016/j.ijhydene.2017.10.049
Sengupta J, Jacob C (2010) The effect of Fe and Ni catalysts on the growth of multiwalled carbon nanotubes using chemical vapor deposition. J Nanopart Res Dordrecht 12(2):457–465. https://doi.org/10.1007/s11051-009-9667-1
Sing KSW, Everett DH, Haul RAW, Moscou L, Pierotti RA, Rouquerol J, Siemieniewska T (2014) Assessment of mesoporosity. In: Press A (ed) Adsorption by powders and porous solids, 2nd edn. Elsevier, Montpeiller, pp 269–302. https://doi.org/10.1002/9783527610044.hetcat0065
Tessonier JP, Ersen O, Weinberg G, Huu-Pham C, Su DS, Schlogl R (2009) Selective deposition of metal nanoparticles inside or outside multiwalled carbon nanotubes. Am Chem Soc Nano 3(8):2081–2089
Theofanidis SA, Galvita VV, Poelman H, Marin GB (2015) Enhanced carbon-resistant dry reforming Fe-Ni catalyst: role of Fe. ACS Catal 5(5):3028–3039. https://doi.org/10.1021/acscatal.5b00357
Theofanidis SA, Galvita VV, Sabbe M, Poelman H, Detavernier C, Marin GB (2017) Controlling the stability of a Fe–Ni reforming catalyst: structural organization of the active components. Appl Catal B 209:405–416. https://doi.org/10.1016/j.apcatb.2017.03.025
Usman M, Daud WMAW, Abbas HF (2015) Dry reforming of methane: influence of process parameters - a review. Renew Sustain Energy Rev 45:710–744. https://doi.org/10.1016/j.rser.2015.02.026
Wang X, Vinodgopal K, Dai G (2019). Synthesis of carbon nanotubes by catalytic chemical vapor deposition. In: Saleh HE, El-Sheikh SMM. Perspective of Carbon Nanotubes. Intech Open. https://doi.org/10.5772/intechopen.86995
Wei J, Iglesia E (2004) Mechanism and site requirements for activation and chemical conversion of methane on supported Pt clusters and turnover rate comparisons among noble metals. J Phys Chem B 108(13):4094–4103. https://doi.org/10.1021/jp036985z
Wu T, Zhang Q, Cai W, Zhang P, Song X, Sun Z, Gao L (2015) Phyllosilicate evolved hierarchical Ni- and Cu–Ni/SiO2 nanocomposites for methane dry reforming catalysis. Appl Catal A 503:94–102. https://doi.org/10.1016/j.apcata.2015.07.012
Yousefi SR, Ghanbari D, Salavati-Niasari M, Hassanpour M (2016) Photo-degradation of organic dyes: simple chemical synthesis of Ni(OH)2 nanoparticles, Ni/Ni(OH)2 and Ni/NiO magnetic nanocomposites. J Mater Sci 27:1244–1253
Yousefi SR, Masjedi-Arani M, Morassaei MS, Salavati-Niasari M, Moayedi H (2019) Hydrothermal synthesis of DyMn2O5/Ba3Mn2O8 nanocomposite as a potential hydrogen storage material. Int J Hydrog Energy 44(43):24005–24016. https://doi.org/10.1016/j.ijhydene.2019.07.113
Zhang J, Li F (2015) Coke-resistant Ni@SiO2 catalyst for dry reforming of methane. Appl Catal B 176–177:513–521. https://doi.org/10.1016/j.apcatb.2015.04.039
Zhu Y, Chen K, Yi C, Mitra S, Barat R (2018) Dry reforming of methane over palladium–platinum on carbon nanotube catalyst. Chem Eng Commun 205(7):888–896. https://doi.org/10.1080/00986445.2017.1423065
Funding
The authors are grateful for the financial support of CAPES (Capes 88882.333487/2019–01) for the scholarship (Vinicius) received during this work.
Author information
Authors and Affiliations
Contributions
CEK has provided scientific suggestions and some results and edited the first and final draft, VMS has provided the major results, and MS has prepared the final manuscript and proposed the idea.
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
All authors have given explicit consent to publish this work.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: George Z. Kyzas
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kozonoe, C.E., Santos, V.M. & Schmal, M. Investigating the stability of Ni and Fe nanoparticle distribution and the MWCNT structure in the dry reforming of methane. Environ Sci Pollut Res 30, 111382–111396 (2023). https://doi.org/10.1007/s11356-023-30205-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-023-30205-4