Abstract
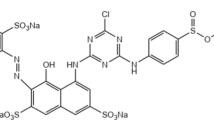
Azo dyes are made in significant amounts annually and released into the environment after being employed in the industry. There are some reports about the toxic effects of these dyes on several organisms. Thus, the textile dye Reactive Black 5 (RB5) has been examined for its cytotoxic effects on the human serum albumin (HSA) structure. Molecular interaction between RB5 and HSA indicated the combination of docking methods, molecular dynamic simulation, and multi-spectroscopic approaches. HSA’s intrinsic fluorescence was well quenched with enhancing RB5 level, confirming complex formation. Molecular dynamics (MD) simulation was done to study the cytotoxic effects of RB5 and HSA conformation. Molecular modeling revealed that the RB5-HSA complex was stabilized by hydrogen bonds and van der Waals interactions. The results of molecular docking revealed that the binding energy of RB5 to HSA was − 27.94 kJ/mol. The change in secondary structure causes the annihilation of hydrogen bonding networks and the reduction of biological activity. This research can indicate a suitable molecular modeling interaction of RB5 and HAS and broaden our knowledge for azo dye toxicity under natural conditions.






Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article and its supplementary information files.
References
Andersen HC (1980) Molecular dynamics simulations at constant pressure and/or temperature. J Chem Phys 72:2384–2393
Asemi-Esfahani Z, Shareghi B, Farhadian S, Momeni L (2021) Effect of naphthol yellow S as a food dye on the lysozyme structure and its mechanisms of action. J Mol Liq 332:115846
Asgharzadeh S, Shareghi B, Farhadian S, Tirgir F (2019) Effect of free L-cysteine on the structure and function of α-chymotrypsin. J Mol Liq 280:79–86
Banerjee S, Choudhury SD, Dasgupta S, Basu S (2008) Photoinduced electron transfer between hen egg white lysozyme and anticancer drug menadione. J Lumin 128:437–444
Carliell C, Barclay S, Naidoo N, Buckley C, Mulholland D, Senior E (1995) Microbial decolourisation of a reactive azo dye under anaerobic conditions. Water Sa 21:61–69
Carter DC, Ho JX (1994) Structure of serum albumin, Advances in protein chemistry. Elsevier, pp. 153–203
Chatterjee S, Lim S-R, Woo SH (2010) Removal of Reactive Black 5 by zero-valent iron modified with various surfactants. Chem Eng J 160:27–32
Chi Z, Liu R, Teng Y, Fang X, Gao C (2010) Binding of oxytetracycline to bovine serum albumin: spectroscopic and molecular modeling investigations. J Agric Food Chem 58:10262–10269
Chi Z, Zhao J, Li W, Araghi A, Tan S (2017) In vitro assessment of phthalate acid esters-trypsin complex formation. Chemosphere 185:29–35
Hiou M, Li H (2003) Adsorption behavior of reactive dye in aqueous solution on chemical cross-linked chitosan beads. Chemosphere 50:1095-1105
Choi H-D, Shin M-C, Kim D-H, Jeon C-S, Baek K (2008) Removal characteristics of reactive black 5 using surfactant-modified activated carbon. Desalination 223:290–298
Chowdhury MH, Aslan K, Malyn SN, Lakowicz JR, Geddes CD (2006) Metal-Enhanced Chemiluminescence J Fluorescence 16:295–299
Chung K-T, Stevens SE, Cerniglia CE (1992) The reduction of azo dyes by the intestinal microflora. Crit Rev Microbiol 18:175–190
Dehkordi SH, Farhadian S, Ghasemi M (2021) The interaction between the azo dye tartrazine and α-chymotrypsin enzyme: molecular dynamics simulation and multi-spectroscopic investigations. J Mol Liq 117931
Delavari B, Saboury AA, Atri MS, Ghasemi A, Bigdeli B, Khammari A, Maghami P, Moosavi-Movahedi AA, Haertlé T, Goliaei B (2015) Alpha-lactalbumin: a new carrier for vitamin D3 food enrichment. Food Hydrocolloids 45:124–131
Divsalar A, Saboury AA, Ahadi L, Zemanatiyar E, Mansouri-Torshizi H (2010) Investigation of effects of newly synthesized Pt (II) complex against human serum albumin and leukemia cell line of K562. BMB Rep 43:766–771
Donovan JW (1969) Changes in ultraviolet absorption produced by alteration of protein conformation. J Biol Chem 244:1961–1967
El Bouraie M, El Din WS (2016) Biodegradation of Reactive Black 5 by Aeromonas hydrophila strain isolated from dye-contaminated textile wastewater. Sustain Environ Res 26:209–216
Eslami-Farsani R, Shareghi B, Farhadian S, Momeni L (2020): Experimental and theoretical investigations on the interaction of glucose molecules with myoglobin in the aqueous solution using theoretical and experimental methods. J Biomol Struct Dynamics 1–12
Farhadian S, Shareghi B, Momeni L, Abou-Zied OK, Sirotkin VA, Tachiya M, Saboury AA (2018) Insights into the molecular interaction between sucrose and α-chymotrypsin. Int J Biol Macromol 114:950–960
Farhadian S, Shareghi B, Asgharzadeh S, Rajabi M, Asadi H (2019a) Structural characterization of α‑chymotrypsin after binding to curcumin: spectroscopic and computational analysis of their binding mechanism. J Mol Liq 111111
Farhadian S, Shareghi B, Tirgir F, Reiisi S, Dehkordi NG, Momeni L, Heidari E (2019b) Design, synthesis, and anti-gastric cancer activity of novel 2, 5-diketopiperazine. J Mol Liq 294:111585
Farhadian S, Hashemi-Shahraki F, Asadpour S, Shareghi B, Shakerian B, Rafatifard M, Firooz AR (2022) Malachite Green, the hazardous materials that can bind to Apo-transferrin and change the iron transfer. Int J Biol Macromol 194:790–799
Ghalandari B, Divsalar A, Saboury AA, Haertlé T, Parivar K, Bazl R, Eslami-Moghadam M, Amanlou M (2014) Spectroscopic and theoretical investigation of oxali–palladium interactions with β-lactoglobulin. Spectrochim Acta Part A Mol Biomol Spectrosc 118:1038–1046
Ghalandari B, Divsalar A, Saboury AA, Parivar K (2015) β-Lactoglobulin nanoparticle as a chemotherapy agent carrier for oral drug delivery system. J Iran Chem Soc 12:613–619
Gharaghani S, Khayamian T, Ebrahimi M (2013) Molecular dynamics simulation study and molecular docking descriptors in structure-based QSAR on acetylcholinesterase (AChE) inhibitors. SAR QSAR Environ Res 24:773–794
Ghosh KS, Sahoo BK, Dasgupta S (2008) Spectrophotometric studies on the interaction between (−)-epigallocatechin gallate and lysozyme. Chem Phys Lett 452:193–197
Greluk M, Hubicki Z, Wawrzkiewicz M, Wołowicz A (2011) Application of weakly basic anion exchanger for removal of Remazol Black B. Challenges Modern Technol 2
He W, Dou H, Zhang L, Wang L, Wang R, Chang J (2014) Spectroscopic study on the interaction of trypsin with bicyclol and analogs. Spectrochim Acta Part A Mol Biomol Spectrosc 118:510–519
He XM, Carter DC (1992) Atomic structure and chemistry of human serum albumin. Nature 358:209
Hou J, Liang Q, Shao S (2017) The binding of four licorice flavonoids to bovine serum albumin by multi-spectroscopic and molecular docking methods: structure–affinity relationship. J Appl Spectrosc 84:177–187
Hu Y-J, Liu Y, Shen X-S, Fang X-Y, Qu S-S (2005a) Studies on the interaction between 1-hexylcarbamoyl-5-fluorouracil and bovine serum albumin. J Mol Struct 738:143–147
Hu Y-J, Liu Y, Zhang L-X, Zhao R-M, Qu S-S (2005b) Studies of interaction between colchicine and bovine serum albumin by fluorescence quenching method. J Mol Struct 750:174–178
Jafari-Arvari H, Saei-Dehkordi SS, Farhadian S (2021) Evaluation of interactions between food colorant, tartrazine, and Apo-transferrin using spectroscopic analysis and docking simulation. J Mol Liq 339:116715
Khatun S, Qais FA (2018) Characterization of the binding of triprolidine hydrochloride to hen egg white lysozyme by multi-spectroscopic and molecular docking techniques. J Mol Liq 269:521–528
Krieger E, Joo K, Lee J, Lee J, Raman S, Thompson J, Tyka M, Baker D, Karplus K (2009) Improving physical realism, stereochemistry, and side-chain accuracy in homology modeling: four approaches that performed well in CASP8. Proteins: Structure. Function Bioinform 77:114–122
Lakowicz JR, Weber G (1973) Quenching of fluorescence by oxygen. Probe for structural fluctuations in macromolecules. Biochemistry 12:4161–4170
Levine WG (1991) Metabolism of azo dyes: implication for detoxication and activation. Drug Metab Rev 23:253–309
Liang H, Gao H, Kong Q, Chen Z (2006) Adsorption equilibrium and kinetics of tetrahydrofuran+ water solution mixture on zeolite 4A. J Chem Eng Data 51:119–122
Manimaran D, Sulthana A, Elangovan N (2018) Reactive black 5 induced developmental defects via potentiating apoptotic cell death in Zebrafish (Danio rerio) embryos. Pharm Pharmacol Int J 6:449–452
Mazuryk O, Gajda-Morszewski P, Flejszar M, Łabuz P, van Eldik R, Stochel G, Brindell M (2020) Influence of aqueous extracts of urban airborne particulate matter on the structure and function of human serum albumin. Environment Pollut 114667
McCann J, Choi E, Yamasaki E, Ames BN (1975) Detection of carcinogens as mutagens in the Salmonella/microsome test: assay of 300 chemicals. Proc Natl Acad Sci 72:5135–5139
Min J, Meng-**a X, Dong Z, Yuan L, **ao-Yu L, **ng C (2004) Spectroscopic studies on the interaction of cinnamic acid and its hydroxyl derivatives with human serum albumin. J Mol Struct 692:71–80
Mohammadi M, Shareghi B, Akbar Saboury A, Farhadian S (2019) Spermine as a possible endogenous allosteric activator of carboxypeptidase A: multispectroscopic and molecular simulation studies. J Biomol Struct Dynamics 1–13
Momeni L, Mahmodian S, Shareghi B, Saboury AA, Farhadian S (2017) The functional and structural stabilization of trypsin by sucrose. Int J Biol Macromol 99:343–349
Momeni L, Shareghi B, Farhadian S, Vaziri S, Saboury AA, Raisi F (2018) A molecular simulation and spectroscopic approach to the binding affinity between trypsin and 2-propanol and protein conformation. Int J Biol Macromol 119:477–485
Moradi M, Divsalar A, Saboury A, Ghalandari B, Harifi A (2015) Inhibitory effects of deferasirox on the structure and function of bovine liver catalase: a spectroscopic and theoretical study. J Biomol Struct Dyn 33:2255–2266
Morris GM, Huey R, Lindstrom W, Sanner MF, Belew RK, Goodsell DS, Olson AJ (2009) AutoDock4 and AutoDockTools4: Automated docking with selective receptor flexibility. J Comput Chem 30:2785–2791
Moussavi G, Mahmoudi M (2009) Removal of azo and anthraquinone reactive dyes from industrial wastewaters using MgO nanoparticles. J Hazard Mater 168:806–812
Noman M, Shahid M, Ahmed T, Niazi MBK, Hussain S, Song F, Manzoor I (2020) Use of biogenic copper nanoparticles synthesized from a native Escherichia sp. as photocatalysts for azo dye degradation and treatment of textile effluents. Environment Pollut 257, 113514
Novotný Č, Dias N, Kapanen A, Malachová K, Vándrovcová M, Itävaara M, Lima N (2006) Comparative use of bacterial, algal and protozoan tests to study toxicity of azo-and anthraquinone dyes. Chemosphere 63:1436–1442
O’mahony T, Guibal E, Tobin J (2002) Reactive dye biosorption by Rhizopus arrhizus biomass. Enzyme Microb Technol 31 456-463
Pan X, Qin P, Liu R, Wang J (2011) Characterizing the interaction between tartrazine and two serum albumins by a hybrid spectroscopic approach. J Agric Food Chem 59:6650–6656
Papadopoulou A, Green RJ, Frazier RA (2005) Interaction of flavonoids with bovine serum albumin: a fluorescence quenching study. J Agric Food Chem 53:158–163
Parrott JL, Bartlett AJ, Balakrishnan VK (2016) Chronic toxicity of azo and anthracenedione dyes to embryo-larval fathead minnow. Environ Pollut 210:40–47
Patra C, Gupta R, Bedadeep D, Narayanasamy S (2020) Surface treated acid-activated carbon for adsorption of anionic azo dyes from single and binary adsorptive systems: a detail insight. Environ Pollut 266:115102
Platzek T, Lang C, Grohmann G, Gi U, Baltes W (1999) Formation of a carcinogenic aromatic amine from an azo dye by human skin bacteria in vitro. Hum Exp Toxicol 18:552–559
Poklar N, Vesnaver G (2000) Thermal denaturation of proteins studied by UV spectroscopy. J Chem Educ 77:380
Prato-Garcia D, Cervantes FJ, Buitrón G (2013) Azo dye decolorization assisted by chemical and biogenic sulfide. J Hazard Mater 250:462–468
Raeessi-Babaheydari E, Farhadian S, Shareghi B (2021) Comparative studies on the interaction of ascorbic acid with gastric enzyme using multispectroscopic and docking methods. J Mole Struct131270
Rajabi M, Shareghi B, Farhadian S, Momeni L (2019) Evaluation of maltose on conformation and activity parameters of trypsin. J Biomol Struct Dynamics 1–6
Rajaguru P, Fairbairn LJ, Ashby J, Willington M, Turner S, Woolford L, Chinnasamy N, Rafferty JA (1999) Genotoxicity studies on the azo dye Direct Red 2 using the in vivo mouse bone marrow micronucleus test. Mutation Res/gen Toxicol Environment Mutagen 444:175–180
Ross PD, Subramanian S (1981) Thermodynamics of protein association reactions: forces contributing to stability. Biochemistry 20:3096–3102
Sadeghi-Kaji S, Shareghi B, Saboury AA, Farhadian S (2019a) Spermine as a porcine pancreatic elastase activator: spectroscopic and molecular simulation studies. J Biomole Struct Dynamics 1–11
Sadeghi-kaji S, Shareghi B, Saboury AA, Farhadian S (2019a) Spectroscopic and molecular docking studies on the interaction between spermidine and pancreatic elastase. Int J Biol Macromol 131:473–483
Sadeghi-kaji S, Shareghi B, Saboury AA, Farhadian S, Hemmati R (2019b) A molecular investigation into the interaction of SiO2 nanoparticles with elastase by multispectroscopic techniques and kinetic studies. Int J Biol Macromol 134:216–222
Shaul GM, Holdsworth TJ, Dempsey CR, Dostal KA (1991) Fate of water soluble azo dyes in the activated sludge process. Chemosphere 22:107–119
Shen L, Xu H, Huang F, Li Y, **ao H, Yang Z, Hu Z, He Z, Zeng Z, Li Y (2015) Investigation on interaction between Ligupurpuroside A and pepsin by spectroscopic and docking methods. Spectrochim Acta Part A Mol Biomol Spectrosc 135:256–263
Singla V, Verma A, Parashar A (2018) A molecular dynamics based study to estimate the point defects formation energies in graphene containing STW defects. Materials Research Express 6:015606
Srinivasan S, Sadasivam SK, Gunalan S, Shanmugam G, Kothandan G (2019) Application of docking and active site analysis for enzyme linked biodegradation of textile dyes. Environ Pollut 248:599–608
Tang L, Cai Y, Yang G, Liu Y, Zeng G, Zhou Y, Li S, Wang J, Zhang S, Fang Y (2014) Cobalt nanoparticles-embedded magnetic ordered mesoporous carbon for highly effective adsorption of rhodamine B. Appl Surf Sci 314:746–753
Van Der Spoel D, Lindahl E, Hess B, Groenhof G, Mark AE, Berendsen HJ (2005) GROMACS: fast, flexible, and free. J Comput Chem 26:1701–1718
Verma A, Parashar A (2017) The effect of STW defects on the mechanical properties and fracture toughness of pristine and hydrogenated graphene. Phys Chem Chem Phys 19:16023–16037
Verma A, Parashar A (2018a) Molecular dynamics based simulations to study failure morphology of hydroxyl and epoxide functionalised graphene. Comput Mater Sci 143:15–26
Verma A, Parashar A (2018b) Molecular dynamics based simulations to study the fracture strength of monolayer graphene oxide. Nanotechnology 29:115706
Verma A, Parashar A (2018c) Reactive force field based atomistic simulations to study fracture toughness of bicrystalline graphene functionalised with oxide groups. Diam Relat Mater 88:193–203
Verma A, Parashar A (2018d) Structural and chemical insights into thermal transport for strained functionalised graphene: a molecular dynamics study. Materials Research Express 5:115605
Verma A, Parashar A, Packirisamy M (2018) Atomistic modeling of graphene/hexagonal boron nitride polymer nanocomposites: a review. Wiley Interdisciplinary Reviews: Computational Molecular Science 8:e1346
Verma A, Kumar R, Parashar A (2019) Enhanced thermal transport across a bi-crystalline graphene–polymer interface: an atomistic approach. Phys Chem Chem Phys 21:6229–6237
Vignesh G, Manojkumar Y, Sugumar K, Arunachalam S (2015) Spectroscopic investigation on the interaction of some polymer–cobalt (III) complexes with serum albumins. J Lumin 157:297–302
Wang Z, Xue M, Huang K, Liu Z (2011) Textile dyeing wastewater treatment, Advances in treating textile effluent. IntechOpen
Xu T, Guo X, Zhang L, Pan F, Lv J, Zhang Y, ** H (2012) Multiple spectroscopic studies on the interaction between olaquindox, a feed additive, and bovine serum albumin. Food Chem Toxicol 50:2540–2546
Yue Y, Chen X, Qin J, Yao X (2009) Spectroscopic investigation on the binding of antineoplastic drug oxaliplatin to human serum albumin and molecular modeling. Colloids Surf, B 69:51–57
Zhang M-F, Xu Z-Q, Ge Y-S, Jiang F-L, Liu Y (2012) Binding of fullerol to human serum albumin: spectroscopic and electrochemical approach. J Photochem Photobiol, B 108:34–43
Acknowledgements
This research was financially supported by the University of Kashan, Shahrekord University (Shahrekord branch) as well as SaNa Zist-pardaz Co., Iran.
Author information
Authors and Affiliations
Contributions
All authors contributed toward data analysis, drafting, and revising the paper and agreed to be responsible for all the aspects of this work. Conceived and designed the experiments: Z.Z., and S.F. Performed the experiments: N.F.D., and S.F. Analyzed and interpreted the data: S.F., and Z.Z. Contributed reagents/materials/analysis tools: Z.Z., and S.F. Wrote the paper: Z.Z., S.F. N.F.D., and N.G.D. Final approval of the manuscript: Z.Z., S.F., N.F.D., and N.G.D.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Mohamed M. Abdel-Daim
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Farajzadeh-Dehkordi, N., Zahraei, Z., Farhadian, S. et al. The interactions between Reactive Black 5 and human serum albumin: combined spectroscopic and molecular dynamics simulation approaches. Environ Sci Pollut Res 29, 70114–70124 (2022). https://doi.org/10.1007/s11356-022-20736-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-20736-7