Abstract
Gold nanorods (AuNRs) are rod-shaped nanoparticles (NPs) with special optical properties that allow their application in several areas including photothermal therapy, diagnosis, drug and gene delivery, cellular imaging, and biosensors. Their high potential for many applications increases the possibility of release in aquatic environments, which can cause risks to organisms. In this study, we evaluated toxic effects of AuNRs on cladoceran and fish (Ceriodaphnia dubia and Danio rerio) and their recovery after post-exposure periods. The EC50 of 0.03 mg L−1 was found for C. dubia in the acute exposure. There was a significant decrease in the number of neonates produced and in the filtration rate of C. dubia after sub-lethal exposure to AuNRs. The toxic mechanism of these NPs to cladocerans was attributed to increases in the reactive oxygen species (ROS) generation. After 4 h of recovery in clean medium, C. dubia were able to reestablish the filtration rate. Enzymatic biomarkers for D. rerio showed significant increases in the activity of superoxide dismutase, catalase, and lipid peroxidation after sub-lethal exposure to AuNRs. These biomarkers were recovered after 168 h in clean water. These results are pivotal on the comprehension of AuNR toxicity to aquatic organisms and are useful in assessing this novel nanomaterial impacts on aquatic biota.





Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
ABNT (2017) Associação Brasileira de Normas Técnicas. Ecotoxicologia Aquática – Toxicidade Crônica – Método de ensaio com Ceriodaphnia spp (Crustacea, Cladocera) NBR 13373:2017. (Brazilian Association of Technical Standards. Aquatic Ecotoxicology - Chronic toxicity – Test method with Ceriodaphnia spp). https://www.abntcatalogo.com.br/norma.aspx?ID=359921
Arndt DA, Moua M, Chen J, Klaper RD (2013) Core structure and surface functionalization of carbon nanomaterials alter impacts to Daphnid mortality, reproduction, and growth: acute assays do not predict chronic exposure impacts. Environ Sci Technol 47:9444–9452 https://pubs.acs.org/doi/abs/10.1021/es4030595
Beers RF, Sizer JW (1952) A spectrophotometric method of measuring the breakdown of hydrogen peroxide by catalase. J Biol Chem 195:133–140 https://pdfs.semanticscholar.org/f824/22e2e58932030184e840cdceed0e6b3ae8df.pdf
Bern L (1994) Particle selection over a broad size range by crustacea zooplankton. Freshw biol 35:105–112. https://doi.org/10.1111/j.1365-2427.1994.tb00870.x
Beutler E (1984) Red cell metabolism: manual of biochemical methods. Grune & Stratton INC, Michigan
Bird RP, Draper HH (1984) Comparative studies on different methods of malonaldehyde determination. Methods Enzymol 105:299–305. https://doi.org/10.1016/S0076-6879(84)05038-2
Bozich JS, Lohse SE, Torelli MD, Murphy CJ, Hamers RJ, Klaper RD (2014) Surface chemistry, charge and ligand type impact the toxicity of gold nanoparticles to Daphnia magna. Environ Sci Nano 1:260–270 http://pubs.rsc.org/en/content/articlepdf/2014/en/c4en00006d
Chupani L, Niksirat H, Velíšek J, Stará A, Hradilová Š, Kolařík J, Panáček A, Zusková E (2018) Chronic dietary toxicity of zinc oxide nanoparticles in common carp (Cyprinus carpio L.): tissue accumulation and physiological responses. Ecotoxicol Environ Saf 147:110–116. https://doi.org/10.1016/j.ecoenv.2017.08.024
Congur G, Sayar F, Erdem A, Piskin E (2013) Voltammetric and impedimetric DNA detection at single-use graphite electrodes modified with gold nanorods. Colloids Surf B Biointerfaces 112:61–66. https://doi.org/10.1016/j.colsurfb.2013.07.040
Craigh PM, Wood CM, McClelland GB (2007) Oxidative stress response and gene expression with acute copper exposure in zebrafish (Danio rerio). Am J Physiol Regul Integr Comp Physiol 293:1882–1892. https://doi.org/10.1152/ajpregu.00383.2007
Dedeh A, Ciutat A, Treguer-Delapierre M, Bourdineaud JP (2015) Impact of gold nanoparticles on zebrafish exposed to a spiked sediment. Nanotoxicology 9:71–80 https://www.ncbi.nlm.nih.gov/pubmed/24559428
Di Giulio RT, Meyer JN (2008) Reactive oxygen species and oxidative stress. In: Di Giulio RT, Hinton DE (eds) The Toxicology of fishes. CRC Press, Boca Raton, pp 273–326
Di Giulio RT, Washburn PC, Wenning RJ (1989) Biochemical responses in aquatic animals: a review of determinants of oxidative stress. Environ Toxicol Chem 8:1103–1123 https://setac.onlinelibrary.wiley.com/doi/pdf/10.1002/etc.5620081203
Dominguez GA, Lohse SE, Torelli MD, Murphy CJ, Hamers RJ, Orr G, Klaper RD (2015) Effects of charge and surface ligand properties of nanoparticles on oxidative stress and gene expression within the gut of Daphnia magna. Aquat Toxicol 162:1–9 https://www.ncbi.nlm.nih.gov/pubmed/25734859
Feng J, Guo Y, Gao Y, Zhu L (2016) Effects of hypoxia on the physiology of zebrafish (Danio rerio): initial responses, acclimation and recovery. Bull Environ Contam Toxicol 96:43–48. https://doi.org/10.1007/s00128-015-1668-4
Ferry JL, Craig P, Hexel C, Sisco P, Frey R, Pennington PL, Fulton MH, Scott GI, Decho AW, Kashiwada S, Murphy CJ, Shaw TJ (2009) Transfer of gold nanoparticles from the water column to the estuarine food web. Nat Nanotechnol 4:441–444 https://www.nature.com/articles/nnano.2009.157
García-Cambero JP, García MN, López GD, Herranz AL, Cuevas L, Pérez-Pastrana E, Cuadal JS, Castelltort MR, Calvo AC (2013) Converging hazard assessment of gold nanoparticles to aquatic organisms. Chemosphere 93:1194–1200. https://doi.org/10.1016/j.chemosphere.2013.06.074
Gauld DT (1951) The grazing rate of planktonic copepods. J Mar Biol Assoc UK 29:695–705. https://doi.org/10.1017/S0025315400052875
Halliwell B (2006) Reactive species and antioxidants. Redox biology is a fundamental theme of aerobic life. Plant Physiol 141:312–322. https://doi.org/10.1104/pp.106.077073
Hao L, Chen L (2006) Oxidative stress responses in different organs of carp (Cyprinus carpio) with exposure to ZnO nanoparticles. Ecotoxicol Environ Saf 80:103–110. https://doi.org/10.1016/j.ecoenv.2012.02.017
Hirayama J, Cho S, Sassone-Corsi P (2007) Circadian control by the reduction/oxidation pathway: catalase represses light-dependent clock gene expression in the zebrafish. PNAS 140:15747–15752. https://doi.org/10.1073/pnas.0705614104
Huang XH, El-Sayed IH, Qian W, El-Sayed MA (2006) Cancer cell imaging and photothermal therapy in the near-infrared region by using gold nanorods. J Am Chem Soc 128:2115–2120 https://pubs.acs.org/doi/abs/10.1021/ja057254a
Huang HC, Barua S, Kay DB, Rege K (2009) Simultaneous enhancement of photothermal stability and gene delivery efficacy of gold nanorods using polyelectrolytes. ACS Nano 3:2941–2952 https://pubs.acs.org/doi/abs/10.1021/nn900947a
Iswarya V, Manivannan J, De A, Paul S, Roy R, Johnson JB, Kundu R, Chandrasekaran N, Mukherjee A, Mukherjee A (2016) Surface cap** and size-dependent toxicity of gold nanoparticles on different trophic levels. Environ Sci Pollut Res 23:4844–4858 https://springer.longhoe.net/article/10.1007%2Fs11356-015-5683-0
Kaya H, Aydın F, Gürkan M, Yılmaz S, Ates M, Demir V, Arslan Z (2015) Effects of zinc oxide nanoparticles on bioaccumulation and oxidative stress in different organs of tilapia (Oreochromis niloticus). Environ Toxicol Pharmacol 40:936–947. https://doi.org/10.1016/j.etap.2015.10.001
Khan FR, Kennaway GM, Croteau MN, Dybowska A, Smith BD, Nogueira AJA, Rainbow PS, Luoma SN, Valsami-Jones E (2014) In vivo retention of ingested Au NPs by Daphnia magna: no evidence for trans-epithelial alimentary uptake. Chemosphere 100:97–104. https://doi.org/10.1016/j.chemosphere.2013.12.051
Larguinho M, Correia D, Diniz MS, Baptista PV (2014) Evidence of one-way flow bioaccumulation of gold nanoparticles across two trophic levels. J Nanopart Res 16:2549–2560 https://springer.longhoe.net/article/10.1007%2Fs11051-014-2549-1
Lovern SB, Owen HA, Klaper R (2008) Electron microscopy of gold nanoparticle intake in the gut of Daphnia magna. Nanotoxicology 2:43–48. https://doi.org/10.1080/17435390801935960
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275 https://www.ncbi.nlm.nih.gov/pubmed/14907713
Lushchak VI (2011a) Environmentally induced oxidative stress in aquatic animals. Aquat Toxicol 101:13–30. https://doi.org/10.1016/j.aquatox.2010.10.006
Lushchak VI (2011b) Adaptive response to oxidative stress: bacteria, fungi, plants and animals. Comp Biochem Physiol-C 153:175–190. https://doi.org/10.1016/j.cbpc.2010.10.004
Mansano AS, Souza JP, Cancino-Bernardi J, Venturini FP, Marangoni VS, Zucolotto V (2018) Toxicity of copper oxide nanoparticles to Neotropical species Ceriodaphnia silvestrii and Hyphessobrycon eques. Environ Pollut 243:723–733. https://doi.org/10.1016/j.envpol.2018.09.020
Marangoni VS, Cancino-Bernardi J, Zucolotto V (2016) Synthesis, physico-chemical properties, and biomedical applications of gold nanorods - a review. J Biomed Nanotechnol 12:1136–1158 https://www.ncbi.nlm.nih.gov/labs/articles/27319210
McWillian RA, Bair DJ (2002) Postexposure feeding depression: a new toxicity endpoint for use in laboratory studies with Daphnia magna. Environ Toxicol Chem 21:1198–1205. https://doi.org/10.1002/etc.5620210612
OECD (1992) Guideline for Testing of Chemicals, Fish, Acute Toxicity Test 203. OECD Publishing. http://www.oecd.org/chemicalsafety/risk-assessment/1948241.pdf.
OECD (2004) Guideline for testing of chemicals, Daphnia sp., acute imobilisation test 202. OECD Publishing. https://www.oecd-ilibrary.org/docserver/9789264069947-en.pdf
Ohkawa H, Ohishi N, Yagi K (1979) Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem 95:351–358. https://doi.org/10.1016/0003-2697(79)90738-3
Okuno T, Kato S, Hatakeyama Y, Okajima J, Maruyama S, Sakamoto M, Mori S, Kodama T (2013) Photothermal therapy of tumors in lymph nodes using gold nanorods and near-infrared laser light. J Control Release 172:879–884. https://doi.org/10.1016/j.jconrel.2013.10.014
Perez-Juste J, Pastoriza-Santos I, Liz-Marzan LM, Mulvaney P (2005) Gold nanorods: synthesis, characterization and applications. Coord Chem Rev 249:1870–1901. https://doi.org/10.1016/j.ccr.2005.01.030
Sancho E, Fernández-Vega C, Villarroel MJ, Andreu-Moliner E, Ferrando DM (2009) Physiological effects of tricyclazole on zebrafish (Danio rerio) and post-exposure recovery. Comp Biochem Physiol C: Toxicol Pharmacol 150:25–32. https://doi.org/10.1016/j.cbpc.2009.02.004
Sau TK, Murphy CJ (2004) Seeded high yield synthesis of short Au nanorods in aqueous solution. Langmuir 20:6414–6420 https://pubs.acs.org/doi/abs/10.1021/la049463z
Shaw BJ, Handy RD (2011) Physiological effects of nanoparticles on fish: a comparison of nanometals versus metal íons. Environ Internat 37:1083–1097 https://www.ncbi.nlm.nih.gov/pubmed/21474182
Smith AM, Mancini MC, Nie SM (2009) Bioimaging second window for in vivo imaging. Nat Nanotechnol 4:710–711 https://www.nature.com/articles/nnano.2009.326
Souza JP, Venturini FP, Santos F, Zucolotto V (2018) Chronic toxicity in Ceriodaphnia dubia induced by graphene oxide. Chemosphere 190:218–224. https://doi.org/10.1016/j.chemosphere.2017.10.018
Souza JP, Mansano AS, Venturini FP, Santos F, Zucolotto V (2019) Antioxidant metabolism of zebrafish after sub-lethal exposure to graphene oxide and recovery. Fish Physiol Biochem 45:1289–1297. https://doi.org/10.1007/s10695-019-00678-7
Teles M, Fierro-Castro C, Na-Phatthalung P, Tvarijonaviciute A, Trindade T, Soares AMVM, Tort L, Oliveira M (2016) Assessment of gold nanoparticle effects in a marine teleost (Sparus aurata) using molecular and biochemical biomarkers. Aquat Toxicol 177:125–135. https://doi.org/10.1016/j.aquatox.2016.05.015
Valerio-García RC, Carbajal-Hernández AL, Martínez-Ruíz EB, Jarquín-Díaz VH, Haro-Pérez C, Martínez-Jerónimo F (2017) Exposure to silver nanoparticles produces oxidative stress and affects macromolecular and metabolic biomarkers in the goodeid fish Chapalichthys pardalis. Sci Total Environ 583:308–318. https://doi.org/10.1016/j.scitotenv.2017.01.070
Venturini FP, Moraes FD, Cortella LRX, Rossi PA, Cruz C, Moraes G (2015) Metabolic responses of trichlorfon (Masoten) on the neotropical freshwater fish pacu (Piaractus mesopotamicus). Fish Physiol Biochem 41:299–309 https://springer.longhoe.net/article/10.1007%2Fs10695-014-9983-y
Venturini FP, Moraes FD, Rossi PA, Avilez IM, Shiogiri NS, Moraes G (2019) A multi-biomarker approach to lambda-cyhalothrin effects on the freshwater teleost matrinxã Brycon amazonicus: single-pulse exposure and recovery. Fish Physiol Biochem 45:341–353. https://doi.org/10.1007/s10695-018-0566-1
Verma A, Stellacci F (2010) Effect of surface properties on nanoparticle-cell interactions. Small 6:12–21. https://doi.org/10.1002/smll.200901158
Wang Z, **e D, Liu H, Bao Z, Wang Y (2016) Toxicity assessment of precise engineered gold nanoparticles with different shapes in zebrafish embryos. RSC Adv 6:33009–33013 http://pubs.rsc.org/en/content/articlelanding/2016/ra/c6ra00632a#!divAbstract
Wang F, Guan W, Xu L, Ding Z, Ma H, Ma A, Terry N (2019) Effects of nanoparticles on algae: adsorption, distribution, ecotoxicity and fate. Appl Sci 9:1534–1547. https://doi.org/10.3390/app9081534
Acknowledgments
Centro Nacional de Pesquisa em Energia e Materiais - Brazilian Nanotechnology National Laboratory CNPEM-LNNano (TEM-18453).
Funding
This study was supported by the Fundação de Amparo à Pesquisa do Estado de São Paulo-FAPESP (Proc. No. 2014/13493-8) and Conselho Nacional de Desenvolvimento Científico e Tecnológico–CNPq (Proc. No. 150555/2018-0).
Author information
Authors and Affiliations
Contributions
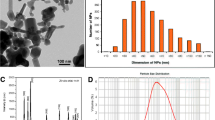
ASM analyzed the reactive oxygen species generation on C. dubia. FPV analyzed the enzymatic activity of zebrafish and performed the English review of the manuscript. VSM synthesized the AuNRs and performed images of transmission electron microscopy. PMPL characterized the AuNRs including hydrodynamic size and zeta potential in Milli-Q water and exposure media. BPCS cultivated and maintained C. dubia in laboratory and performed experiments of survival and reproduction. BD cultivated zebrafish in laboratory and achieved sub-lethal experiments. JPS performed all analyses on C. dubia and zebrafish, proposed the experimental design, discussed and interpreted the results, and was a major contributor in writing the manuscript. VZ proposed to experimental design, reviewed, and discussed the results. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical approval
All zebrafish experiments were approved by the Ethics Committee for Animal Research of the Physics Institute of São Carlos of the University of São Paulo, under protocol number 01/2014.
Consent to publication
Not applicable.
Consent to participate
Not applicable.
Competing interest
The authors declare that they have no competing interest.
Additional information
Responsible Editor: Philippe Garrigues
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Souza, J.P., Mansano, A.S., Venturini, F.P. et al. Toxicity of gold nanorods on Ceriodaphnia dubia and Danio rerio after sub-lethal exposure and recovery. Environ Sci Pollut Res 28, 25316–25326 (2021). https://doi.org/10.1007/s11356-021-12423-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-021-12423-w