Abstract
Background, aim, and scope
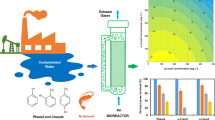
Two new high phenol-degrading strains, Micrococcus sp. and Alcaligenes faecalis JH 1013, were isolated. The two isolates could grow aerobically in mineral salts medium containing phenol as a sole carbon source at concentration of 3,000 mg L−1. It was found that the binary mixed culture of the two isolates possessed good potential for phenol removal.
Material and methods
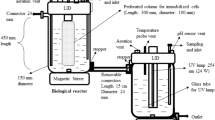
Phenol biodegradation using the binary mixed culture of the two isolates was studied. The optimal conditions were determined to be temperature 32°C, pH 7.0, inoculum size 10.0%, and agitation rate 150 rpm in the synthetic wastewater. In addition, the kinetics of the cell growth and phenol degradation by the binary mixed culture were also investigated using Haldane model over a wide range of initial phenol concentrations from 20 to 2,400 mg L−1.
Results
The experimental data indicated that the binary mixed culture had pretty high phenol degradation potential, which could thoroughly degrade the phenol in the synthetic wastewater containing phenol 2,400 mg L−1 within 72 h under aerobic condition. Under the optimal conditions, the phenol concentration was reduced speedily from 1,000 to below 0.28 mg L−1 in the presence of the binary mixed culture, and the phenol degradation rate reached 99.97% after 16 h. It was well below the standard value 0.28 mg L−1 as described by Chinese Environmental Protection Agency. It was clear that the Haldane kinetic model adequately described the dynamic behavior of phenol degradation by the binary mixed culture with kinetic constants of q max = 0.45 h−1, K sq = 64.28 mg L−1, and K iq = 992.79 mg L−1. The phenol concentration to avoid substrate inhibition had been inferred theoretically to be 252.62 mg L−1.
Conclusions
Phenol, as the only carbon source, could be degraded by the binary mixed culture at high initial phenol concentrations. Phenol exhibited inhibitory behavior, and the growth kinetics of the binary mixed culture could be correlated well by the simple Haldane’s inhibitory model. The kinetics parameters were invariably required for the design and simulation of batch and continuous bioreactor treating phenolic wastewaters.






Similar content being viewed by others
Abbreviations
- μ :
-
Specific growth rate (h−1)
- S :
-
Phenol concentration (mg L−1)
- K iμ :
-
Inhibition constant for cell growth (mg L−1)
- K sμ :
-
Half-saturation constant for cell growth (mg L−1)
- μ max :
-
Maximum specific growth rate (h−1)
- t :
-
Time (h)
- X :
-
Cell concentration (mg L−1)
- X 0 :
-
Cell concentration in lag phase (mg L−1)
- Δt :
-
Time in exponential phase
- q :
-
Specific substrate consumption rate (h−1)
- q max :
-
Maximum substrate consumption rate (h−1)
- K sq :
-
Half-saturation constant for substrate consumption (mg L−1)
- K iq :
-
Inhibition constant for substrate consumption (mg L−1)
- Y 0 :
-
Yield coefficient (mg mg−1)
- K i :
-
Inhibition constant (mg L−1)
- K s :
-
Half-saturation constant (mg L−1)
- R 2 :
-
Correlation coefficient
References
Agarry SE, Solomon BO (2008) Kinetics of batch microbial degradation of phenols by indigenous Pseudomonas fluorescence. Int J Environ Sci Tech 5:223–232
Agarry SE, Solomon BO, Layokun SK (2008) Kinetics of batch microbial degradation of phenols by indigenous binary mixed culture of Pseudomonas aeruginosa and Pseudomonas fluorescence. Afr J Biotechnol 7:2417–2423
Agency for Toxic Substances and Disease Registry (ATSDR) (2006) Toxicological profile for Phenol. Department of Health and Human Services, Public Health Service, Atlanta
Alemzadeh I, Vossoughi F, Houshmandi M (2002) Phenol biodegradation by rotating biological contactor. Biochem Eng J 11:19–23
APHA-AWWA-WEF (1995) Standard method for the examination of water and wastewater, 19th edn. Am Publ Hlth Assoc, Washington DC
Arutchelvan V, Kanakasabai V, Elangovan R, Nagarajan S, Muralikrishnan V (2006) Kinetics of high strength phenol degradation using Bacillus brevis. J Hazard Mater B129:216–222
Bai J, Wen J-P, Li H-M, Jiang Y (2007) Kinetic modeling of growth and biodegradation of phenol and m-cresol using Alcaligenes faecalis. Process Biochem 42:510–517
Carmona M, De Lucas A, Valverde JL, Velasco B, Rodríguez JF (2006) Combined adsorption and ion exchange equilibrium of phenol on Amberlite IRA-420. Chem Eng J 117:155–160
Collins LD, Daugulis AJ (1997) Biodegradation of phenol at high concentrations in two-phase partitioning batch and fed-batch reactors. Biotechnol Bioeng 55:155–162
Garcĭa IG, Venceslada JLB, Peňa PRJ, Gómez ER (1997) Biodegradation of phenol compounds in vinasse using Aspergillius terreus and Geotrichum candidum. Water Res 31:2005–2011
Geng A, Soh AEW, Lim CJ, Loke LCT (2006) Isolation and characterization of a phenol-degrading bacterium from an industrial activated sludge. Appl Microbiol Biotechnol 71:728–735
Giti E, Mehdi H, Nasser G (2005) Development of a microtitre plate method for determination of phenol utilization, biofilm formation and respiratory activity by environmental bacterial isolates. Int Biodeter Biodegr 56:231–235
Hamad A, Aidan A, Fayed M, Mehrvar M (2005) Experimental investigation of phenolic wastewater treatment using combined activated carbon and UV processes. Clean Technol Environ Policy 7:177–181
Jiang Y, Wen J, Li H, Yang S, Hu Z (2005) The biodegradation of phenol at high concentration by the yeast Candida tropicalis. Biochem Een J 24:243–247
Jiang Y, Wen J, Bai J, Jia X, Hu Z (2007) Biodegradation of phenol at high initial concentration by Alcaligenes faecalis. J Hazard Mater 147:672–676
Kapley A, Tolmare A, Purohit HJ (2001) Role of oxygen in the utilization of phenol by Pseudomonas CF600 in continuous culture. World J Microbiol Biotechnol 17:801–804
Kauffmann C, Petersen BR, Bjerrum MJ (1999) Enzymatic removal of phenols from aqueous solutions by Coprinus cinereus peroxidase and hydrogen peroxide. J Biotechnol 73:71–74
Kujawski W, Warszawski A, Ratajczak W, Porębski T, Capała W, Ostrowska I (2004) Removal of phenol from wastewater by different separation techniques. Desalination 163:287–296
Kumar A, Kumar S, Kumar S (2005) Biodegradation kinetics of phenol and catechol using Pseudomonas putida MTCC 1194. Biochem Eng J 22:151–159
Lazarova Z, Boyadzhieva S (2004) Treatment of phenol-containing aqueous solutions by membrane-based solvent extraction in coupled ultrafiltration modules. Chem Eng J 100:129–138
Lesllie Grady CP, Daigger GT, Lim HC (1999) Biological wastewater treatment. Marcel Dekker, New York
Marrot B, Barrios-Martinez A, Moulin P, Roche N (2006) Biodegradation of high phenol concentration by activated sludge in an immersed membrane bioreactor. Biochem Eng J 30:174–183
Monteiro ÁAMG, Boaventura RAR, Rodrigues AE (2000) Phenol biodegradation by Pseudomonas putida DSM 548 in a batch reactor. Biochem Eng J 6:45–49
Nuhoglu A, Yalcin B (2005) Modeling of phenol removal in a batch reactor. Process Biochem 40:1233–1239
Pawlowsky U, Howell JA (1973) Mixed culture biooxidation of phenol. I. Determination of kinetic parameters. Biotechnol Bioeng 15:889–896
Rocha LL, De Aguiar Cordeiro R, Cavalcante RM, Do Nascimento RF, Martins SCS, Santaella ST, Melo VMM (2007) Isolation and characterization of phenol-degrading yeasts from an oil refinery wastewater in Brazil. Mycopathologia 164:183–188
Saravanan P, Pakshirajan K, Saha P (2008) Growth kinetics of an indigenous mixed microbial consortium during phenol degradation in a batch reactor. Bioresour Technol 99:205–209
Singh RK, Kumar S, Kumar S, Kumar A (2008) Biodegradation kinetic studies for the removal of p-cresol from wastewater using Gliomastix indicus MTCC 3869. Biochem Eng J 40:293–303
Tomaszewska M, Mozia S, Morawski AW (2004) Removal of organic matter by coagulation enhanced with adsorption on PAC. Desalination 161:79–87
Wang S-J, Loh K-C (1999) Modeling the role of metabolic intermediates in kinetics of phenol biodegradation. Enzyme Microb Technol 25:177–184
Wu J, Rudy K, Sapark J (2000) Oxidation of aqueous phenol by ozone and peroxidase. Adv Environ Res 4:339–346
Zheng C, Zhou J, Wang J, Wang J, Qu B (2008) Isolation and characterization of a nitrobenzene degrading yeast strain from activated sludge. J Hazard Mater 160:194–199
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Hailong Wang
Rights and permissions
About this article
Cite this article
Zeng, HY., Jiang, H., **a, K. et al. Characterization of phenol degradation by high-efficiency binary mixed culture. Environ Sci Pollut Res 17, 1035–1044 (2010). https://doi.org/10.1007/s11356-009-0248-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-009-0248-8