Abstract
Introduction
Postmenopausal women with osteoporosis (PMOP) are prone to fragility fractures. Osteoporosis is associated with alterations in the levels of specific circulating metabolites.
Objectives
To analyze the metabolic profile of individuals with PMOP and identify novel metabolites associated with bone mineral density (BMD).
Methods
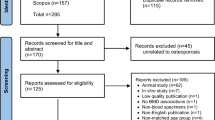
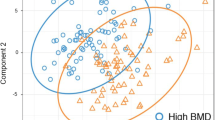
We performed an unsupervised metabolomics analysis of plasma samples from participants with PMOP and of normal controls (NC) with normal bone mass. BMD values for the lumber spine and the proximal femur were determined using dual-energy X-ray absorptiometry. Principal component analysis (PCA) and supervised partial least squares discriminant analysis (PLS-DA) were performed for metabolomic profile analyses. Metabolites with P < 0.05 in the t-test, VIP > 1 in the PLS-DA model, and SNR > 0.3 between the PMOP and NC groups were defined as differential abundant metabolites (DAMs). The SHapley additive explanations (SHAP) method was utilized to determine the importance of permutation of each DAM in the predictive model between the two groups. ROC analysis and correlation analysis of metabolite relative abundance and BMD/T-scores were conducted. KEGG pathway analysis was used for functional annotation of the candidate metabolites.
Results
Overall, 527 annotated molecular markers were extracted in the positive and negative total ion chromatogram (TIC) of each sample. The PMOP and NC groups could be differentiated using the PLS-DA model. Sixty-eight DAMs were identified, with most relative abundances decreasing in the PMOP samples. SHAP was used to identify 9 DAM metabolites as factors distinguishing PMOP from NC. The logistic regression model including Triethanolamine, Linoleic acid, and PC(18:1(9Z)/18:1(9Z)) metabolites demonstrated excellent discrimination performance (sensitivity = 97.0, specificity = 96.6, AUC = 0.993). The correlation analysis revealed that the abundances of Triethanolamine, PC(18:1(9Z)/18:1(9Z)), 16-Hydroxypalmitic acid, and Palmitic acid were significantly positively correlated with the BMD/T score (Pearson correlation coefficients > 0.5, P < 0.05). Most candidate metabolites were involved in lipid metabolism based on KEGG functional annotations.
Conclusion
The plasma metabolomic signature of PMOP patients differed from that of healthy controls. Marker metabolites may help provide information for the diagnosis, therapy, and prevention of PMOP. We highlight the application of feature selection approaches in the analysis of high-dimensional biological data.






Similar content being viewed by others
Data availability
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Ahmed, S. S., Santosh, W., Kumar, S., & Christlet, H. T. (2009). Metabolic profiling of Parkinson’s disease: Evidence of biomarker from gene expression analysis and rapid neural network detection. Journal Of Biomedical Science, 16, 63. https://doi.org/10.1186/1423-0127-16-63
Bloch, L., Friedrich, C. M., Alzheimer’s, I., & Disease Neuroimaging. (2021). Data analysis with Shapley values for automatic subject selection in Alzheimer’s disease data sets using interpretable machine learning. Alzheimer’S Research & Therapy, 13, 155. https://doi.org/10.1186/s13195-021-00879-4
Cooper, D. M., Thomas, C. D., Clement, J. G., Turinsky, A. L., Sensen, C. W., & Hallgrimsson, B. (2007). Age-dependent change in the 3D structure of cortical porosity at the human femoral midshaft. Bone, 40, 957–965. https://doi.org/10.1016/j.bone.2006.11.011
Deng, D., Pan, C., Wu, Z., Sun, Y., Liu, C., **ang, H., Yin, P., Shang, D., et al. (2021). An integrated metabolomic study of osteoporosis: Discovery and quantification of hyocholic acids as candidate markers. Frontiers in Pharmacology, 12, 725341. https://doi.org/10.3389/fphar.2021.725341
Dimai, H. P. (2017). Use of dual-energy X-ray absorptiometry (DXA) for diagnosis and fracture risk assessment; WHO-criteria, T- and Z-score, and reference databases. Bone, 104, 39–43. https://doi.org/10.1016/j.bone.2016.12.016
During, A. (2020). Osteoporosis: A role for lipids. Biochimie, 178, 49–55. https://doi.org/10.1016/j.biochi.2020.08.004
Eastell, R., & Szulc, P. (2017). Use of bone turnover markers in postmenopausal osteoporosis. Lancet Diabetes Endocrinol, 5, 908–923. https://doi.org/10.1016/S2213-8587(17)30184-5
Ferrucci, L., Baroni, M., Ranchelli, A., Lauretani, F., Maggio, M., Mecocci, P., Ruggiero, C., et al. (2014). Interaction between bone and muscle in older persons with mobility limitations. Current Pharmaceutical Design, 20, 3178–3197. https://doi.org/10.2174/13816128113196660690
Jacob, M., Lopata, A. L., Dasouki, M., & Abdel Rahman, A. M. (2019). Metabolomics toward personalized medicine. Mass Spectrometry Reviews, 38, 221–238. https://doi.org/10.1002/mas.21548
Johnson, C. H., Ivanisevic, J., & Siuzdak, G. (2016). Metabolomics: Beyond biomarkers and towards mechanisms. Nature Reviews Molecular Cell Biology, 17, 451–459. https://doi.org/10.1038/nrm.2016.25
Karasik, D., Rivadeneira, F., & Johnson, M. L. (2016). The genetics of bone mass and susceptibility to bone diseases. Nature Reviews Rheumatology, 12, 323–334. https://doi.org/10.1038/nrrheum.2016.48
Kim, H. J., Lee, Y., Chang, E. J., Kim, H. M., Hong, S. P., Lee, Z. H., Ryu, J., Kim, H. H., et al. (2007). Suppression of osteoclastogenesis by N,N-dimethyl-d-erythro-sphingosine: A sphingosine kinase inhibition-independent action. Molecular Pharmacology, 72, 418–428. https://doi.org/10.1124/mol.107.034173
Kocijan, R., Muschitz, C., Geiger, E., Skalicky, S., Baierl, A., Dormann, R., Plachel, F., Feichtinger, X., Heimel, P., Fahrleitner-Pammer, A., Grillari, J., Redl, H., Resch, H., Hackl, M., et al. (2016). Circulating microRNA signatures in patients with idiopathic and postmenopausal osteoporosis and fragility fractures. Journal of Clinical Endocrinology and Metabolism, 101, 4125–4134. https://doi.org/10.1210/jc.2016-2365
Lin, G., Wang, H., Dai, J., Li, X., Guan, M., Gao, S., Ding, Q., Wang, H., Fang, H., et al. (2017). Conjugated linoleic acid prevents age-induced bone loss in mice by regulating both osteoblastogenesis and adipogenesis. Biochemical and Biophysical Research Communications, 490, 813–820. https://doi.org/10.1016/j.bbrc.2017.06.122
Mathias, C., Muzzi, J. C. D., Antunes, B. B., Gradia, D. F., Castro, M. A. A., & Carvalho de Oliveira, J. (2021). Unraveling immune-related lncRNAs in breast cancer molecular subtypes. Frontiers in Oncology, 11, 692–170. https://doi.org/10.3389/fonc.2021.692170
Melton, L. J., III., Chrischilles, E. A., Cooper, C., Lane, A. W., & Riggs, B. L. (1992). Perspective. How many women have osteoporosis? Journal of Bone and Mineral Research, 7, 1005–1010. https://doi.org/10.1002/jbmr.5650070902
Miyamoto, T., Hirayama, A., Sato, Y., Koboyashi, T., Katsuyama, E., Kanagawa, H., Fujie, A., Morita, M., Watanabe, R., Tando, T., Miyamoto, K., Tsuji, T., Funayama, A., Soga, T., Tomita, M., Nakamura, M., Matsumoto, M., et al. (2018). Metabolomics-based profiles predictive of low bone mass in menopausal women. Bone Reports, 9, 11–18. https://doi.org/10.1016/j.bonr.2018.06.004
Ozbas, H., Tutgun Onrat, S., & Ozdamar, K. (2012). Genetic and environmental factors in human osteoporosis. Molecular Biology Reports, 39, 11289–11296. https://doi.org/10.1007/s11033-012-2038-5
Pan, P., Li, Y., **ao, Y., Han, B., Su, L., Su, M., Li, Y., Zhang, S., Jiang, D., Chen, X., Zhou, F., Ma, L., Bao, P., **e, L., et al. (2020). Prognostic assessment of COVID-19 in the Intensive care unit by machine learning methods: Model development and validation. Journal of Medical Internet Research, 22, e23128. https://doi.org/10.2196/23128
Panahi, N., Arjmand, B., Ostovar, A., Kouhestani, E., Heshmat, R., Soltani, A., Larijani, B., et al. (2021). Metabolomic biomarkers of low BMD: A systematic review. Osteoporosis International, 32, 2407–2431. https://doi.org/10.1007/s00198-021-06037-8
Parveen, B., Parveen, A., & Vohora, D. (2019). Biomarkers of osteoporosis: An update. Endocrine, Metabolic & Immune Disorders: Drug Targets, 19, 895–912. https://doi.org/10.2174/1871530319666190204165207
Pico, C., Serra, F., Rodriguez, A. M., Keijer, J., & Palou, A. (2019). Biomarkers of nutrition and health: New tools for new approaches. Nutrients, 11, 10. https://doi.org/10.3390/nu11051092
Shan, Z., Zhao, Y., Qiu, Z., Angxiu, S., Gu, Y., Luo, J., Bi, H., Luo, W., **ong, R., Ma, S., He, Z., Chen, L., et al. (2021). Conjugated linoleic acid prompts bone formation in ovariectomized osteoporotic rats and weakens osteoclast formation after treatment with ultraviolet B. Annals of Translational Medicine, 9, 503. https://doi.org/10.21037/atm-21-934
Si, L., Winzenberg, T. M., Jiang, Q., Chen, M., & Palmer, A. J. (2015). Projection of osteoporosis-related fractures and costs in China: 2010–2050. Osteoporosis International, 26, 1929–1937. https://doi.org/10.1007/s00198-015-3093-2
Si, Z., Zhou, S., Shen, Z., & Luan, F. (2020). High-throughput metabolomics discovers metabolic biomarkers and pathways to evaluating the efficacy and exploring potential mechanisms of osthole against osteoporosis based on UPLC/Q-TOF-MS coupled with multivariate data analysis. Frontiers in Pharmacology, 11, 741. https://doi.org/10.3389/fphar.2020.00741
Suzuki, S., Minamidate, T., Shiga, A., Ruike, Y., Ishiwata, K., Naito, K., Ishida, A., Deguchi, H., Fujimoto, M., Koide, H., Tatsuno, I., Ikeda, Ji., Yamazaki, Y., Sasano, H., Yokote, K., et al. (2020). Steroid metabolites for diagnosing and predicting clinicopathological features in cortisol-producing adrenocortical carcinoma. BMC Endocrine Disorders, 20, 173. https://doi.org/10.1186/s12902-020-00652-y
van Gastel, N., & Carmeliet, G. (2021). Metabolic regulation of skeletal cell fate and function in physiology and disease. Nature Metabolism, 3, 11–20. https://doi.org/10.1038/s42255-020-00321-3
Vardarajan, B., Kalia, V., Manly, J., Brickman, A., Reyes‐Dumeyer, D., Lantigua, R., Ionita‐Laza, I., Jones, D. P., Miller, G. W., Mayeux, R., et al. (2020). Differences in plasma metabolites related to Alzheimer’s disease, APOE epsilon4 status, and ethnicity. Alzheimers Dement (N Y), 6, e12025. https://doi.org/10.1002/trc2.12025
Wang, J., Yan, D., Zhao, A., Hou, X., Zheng, X., Chen, P., Bao, Y., Jia, W., Hu, C., Zhang, Z. L., Jia, W., et al. (2019). Discovery of potential biomarkers for osteoporosis using LC-MS/MS metabolomic methods. Osteoporosis International, 30, 1491–1499. https://doi.org/10.1007/s00198-019-04892-0
Xue, L., Wang, Y., Liu, L., Zhao, L., Han, T., Zhang, Q., Qin, L., et al. (2011). A HNMR-based metabonomics study of postmenopausal osteoporosis and intervention effects of Er-**an Decoction in ovariectomized rats. International Journal of Molecular Sciences, 12, 7635–7651. https://doi.org/10.3390/ijms12117635
Yan, Y., Wang, J., Huang, D., Lv, J., Li, H., An, J., Cui, X., Zhao, H., et al. (2022). Plasma lipidomics analysis reveals altered lipids signature in patients with osteonecrosis of the femoral head. Metabolomics, 18, 14. https://doi.org/10.1007/s11306-022-01872-0
Yang, Y., Yujiao, W., Fang, W., Linhui, Y., Ziqi, G., Zhichen, W., Zirui, W., Shengwang, W., et al. (2020). The roles of miRNA, lncRNA and circRNA in the development of osteoporosis. Biological Research, 53, 40. https://doi.org/10.1186/s40659-020-00309-z
You, Y. S., Lin, C. Y., Liang, H. J., Lee, S. H., Tsai, K. S., Chiou, J. M., Chen, Y. C., Tsao, C. K., Chen, J. H., et al. (2014). Association between the metabolome and low bone mineral density in Taiwanese women determined by (1)H NMR spectroscopy. Journal of Bone and Mineral Research, 29, 212–222. https://doi.org/10.1002/jbmr.2018
Zhang, M., Wang, Y., Zhang, Q., Wang, C., Zhang, D., Wan, J. B., Yan, C., et al. (2018). UPLC/Q-TOF-MS-based metabolomics study of the anti-osteoporosis effects of Achyranthes bidentata polysaccharides in ovariectomized rats. International Journal of Biological Macromolecules, 112, 433–441. https://doi.org/10.1016/j.ijbiomac.2018.01.204
Acknowledgements
The authors are greatful to Novogene Bioinformatics Technology Co., Ltd. for providing helps in metabolome data analysis and experimental technical support.
Funding
This study was supported by grants from National Natural Science Foundation of China (81702067); the Shaanxi Provincial Key Research and Development Program (2020GXLH-Y-027, 2021SF-030); the Fundamental Research Funds for the Central Universities (G2020KY0516).
Author information
Authors and Affiliations
Contributions
Jihan Wang and Dageng Huang designed the project, and co-wrote the manuscript. Jihan Wang recruited the participants, collected the clinical data. Yangyang Wang analyzed the metabolomics data, and did statistical analysis. Yuhong Zeng revised the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no conflict of interest.
Ethics approval and consent to participate
The current study was approved by Institutional Ethical Review Board, Northwestern Polytechnical University (Approval number: 202002023). Each participant signed informed consent.
Consent for publication
Not applicable
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
11306_2022_1937_MOESM1_ESM.tif
Fig. S1 ROC curve of combination test of M150T272, M279T81, and M769T49 in distinguishing between PMOP and NC. The ROC analysis was performed by MedCalc 19.0.4. Supplementary file1 (TIF 115 kb)
11306_2022_1937_MOESM2_ESM.tif
Fig S2. Correlation analysis between the relative abundance of DAMs and BMD/T-score in positive TIC mode. There were 37 DAMs in PMOP group compared to NC group in the positive TIC mode. Detailed information about ID and name of the 37 DAMs is shown in Table S1 (positive TIC). Supplementary file2 (TIF 2521 kb)
11306_2022_1937_MOESM3_ESM.tif
Fig S3. Correlation analysis between the relative abundance of DAMs and BMD/T-score in negative TIC mode. There were 31 DAMs in PMOP group compared to NC group in the negative TIC mode. Detailed information about ID and name of the negative DAMs is shown in Table S2 (negative TIC). Supplementary file3 (TIF 2210 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, J., Wang, Y., Zeng, Y. et al. Feature selection approaches identify potential plasma metabolites in postmenopausal osteoporosis patients. Metabolomics 18, 86 (2022). https://doi.org/10.1007/s11306-022-01937-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11306-022-01937-0