Abstract
Introduction
Horses with asthma or osteoarthritis frequently receive ω-3 fatty acid supplements. Docosahexaenoic (DHA; 22:6) and eicosapentaenoic (EPA; 20:5) acids are essential ω-3 fatty acid precursors of anti-inflammatory mediators and components of structural glycerophospholipids (GPL) that act as reservoirs of these fatty acids. Analysis of the incorporation of dietary DHA + EPA into GPL pools in different body compartments has not been undertaken in horses.
Objectives
We undertook a detailed study of dietary supplementation with DHA + EPA in horses and monitored incorporation into DHA- and EPA-containing glycerophosphocholines (GPC) 38:5, 38:6, 40:5, and 40:6 in plasma, synovial fluid (SF), and surfactant.
Methods
Horses (n = 20) were randomly assigned to the supplement or control group and evaluated on days 0, 30, 60, and 90. GPC in plasma, SF, and surfactant were measured by high-resolution mass spectrometry with less than 3 ppm mass error. Validation of DHA and EPA incorporation into these GPC was conducted utilizing MS2 of the [M + Cl]− adducts of GPC.
Results
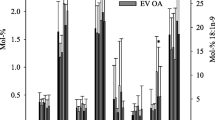
Dietary supplementation resulted in augmented levels of GPC 38:5, 38:6, 40:5, and 40:6 in all compartments. Maximum incorporation into GPCs was delayed until 60 days. Significant increases in the levels of GPC 38:5, 40:5, and 40:6, containing docosapentaenoic acid (DPA; 22:5), also was noted.
Conclusions
DHA and EPA supplementation results in augmented storage pools of ω-3 essential fatty acids in SF and surfactant GPC. This has the potential to improve the ability of anti-inflammatory mechanisms to resolve inflammatory pathways in these critical compartments involved in arthritis and asthma.



Similar content being viewed by others
Data availability
Detailed data and materials are available upon request. A supplemental data table is available in Excel format.
Code availability
Not applicable.
References
Abdulnour, R. E., Sham, H. P., Douda, D. N., Colas, R. A., Dalli, J., Bai, Y., Ai, X., Serhan, C. N., & Levy, B. D. (2016). Aspirin-triggered resolvin D1 is produced during self-resolving gram-negative bacterial pneumonia and regulates host immune responses for the resolution of lung inflammation. Mucosal Immunology, 9, 1278–1287.
Abdulrazaq, M., Innes, J. K., & Calder, P. C. (2017). Effect of omega-3 polyunsaturated fatty acids on arthritic pain: A systematic review. Nutrition, 39–40, 57–66.
Adler, N., Schoeniger, A., & Fuhrmann, H. (2018). Polyunsaturated fatty acids influence inflammatory markers in a cellular model for canine osteoarthritis. Journal of Animal Physiology and Animal Nutrition (Berlin), 102, e623–e632.
Bligh, E. G., & Dyer, W. J. (1959). A rapid method of total lipid extraction and purification. Canadian Journal of Biochemistry and Physiology, 37, 911–917.
Bullone, M., Vargas, A., Elce, Y., Martin, J. G., & Lavoie, J. P. (2017). Fluticasone/salmeterol reduces remodelling and neutrophilic inflammation in severe equine asthma. Science and Reports, 7, 8843.
Calder, P. C. (2014). Very long chain omega-3 (n-3) fatty acids and human health. European Journal of Lipid Science and Technology, 116, 1280–1300.
Calder, P. C. (2015). Marine omega-3 fatty acids and inflammatory processes: Effects, mechanisms and clinical relevance. Biochimica et Biophysica Acta (BBA)-Molecular and Cell Biology of Lipids, 1851, 469–484.
Calder, P. C. (2017a). Omega-3 fatty acids and inflammatory processes: from molecules to man. Biochemical Society Transactions, 45, 1105–1115.
Calder, P. C. (2017b). Very long-chain n-3 fatty acids and human health: fact, fiction and the future. Proceedings of the Nutrition Society, 77, 1–21.
Calder, P. C., & Deckelbaum, R. J. (2014). Dietary fatty acids in health and disease. Current Opinion in Clinical Nutrition and Metabolic Care, 17, 111–115.
Caron, J. P., Gandy, J. C., Brown, J. L., & Sordillo, L. M. (2019). Omega-3 fatty acids and docosahexaenoic acid oxymetabolites modulate the inflammatory response of equine recombinant interleukin1beta-stimulated equine synoviocytes. Prostaglandins and Other Lipid Mediators, 142, 1–8.
Christmann, U., Hite, R. D., Tan, R. H., Thatcher, C. D., Witonsky, S. G., Werre, S. R., & Buechner-Maxwell, V. A. (2010). Surfactant alterations in horses with recurrent airway obstruction at various clinical stages. American Journal of Veterinary Research, 71, 468–475.
Christmann, U., Hite, R. D., Witonsky, S. G., Buechner-Maxwell, V., & Wood, P. L. (2019). Evaluation of lipid markers in surfactant obtained from asthmatic horses exposed to hay. American Journal of Veterinary Research, 80, 300–305.
Christmann, U., Livesey, L. C., Taintor, J. S., Waldridge, B. M., Schumacher, J., Grier, B. L., & Hite, R. D. (2006). Lung surfactant function and composition in neonatal foals and adult horses. Journal of Veterinary Internal Medicine, 20, 1402–1407.
Cokelaere, S. M., Plomp, S. G. M., de Boef, E., de Leeuw, M., Bool, S., van de Lest, C. H. A., van Weeren, P. R., & Korthagen, N. M. (2018). Sustained intra-articular release of celecoxib in an equine repeated LPS synovitis model. European Journal of Pharmaceutics and Biopharmaceutics, 128, 327–336.
Corbee, R. J., Barnier, M. M., van de Lest, C. H., & Hazewinkel, H. A. (2013). The effect of dietary long-chain omega-3 fatty acid supplementation on owner’s perception of behaviour and locomotion in cats with naturally occurring osteoarthritis. Journal of Animal Physiology and Animal Nutrition (Berlin), 97, 846–853.
Couetil, L. L., Cardwell, J. M., Gerber, V., Lavoie, J. P., Leguillette, R., & Richard, E. A. (2016). Inflammatory airway disease of horses-revised consensus statement. Journal of Veterinary Internal Medicine, 30, 503–515.
Dartt, D. A., Hodges, R. R., & Serhan, C. N. (2019). Immunoresolvent resolvin D1 maintains the health of the ocular surface. Advances in Experimental Medicine and Biology, 1161, 13–25.
de Grauw, J. C., van Loon, J. P., van de Lest, C. H., Brunott, A., & van Weeren, P. R. (2014). In vivo effects of phenylbutazone on inflammation and cartilage-derived biomarkers in equine joints with acute synovitis. The Veterinary Journal, 201, 51–56.
Elzinga, S. E., Betancourt, A., Stewart, J. C., Altman, M. H., Barker, V. D., Muholland, M., Bailey, S., Brennan, K. M., & Adams, A. A. (2019). Effects of docosahexaenoic acid-rich microalgae supplementation on metabolic and inflammatory parameters in horses with equine metabolic syndrome. Journal of Equine Veterinary Science, 83, 102811.
Gabbs, M., Leng, S., Devassy, J. G., Monirujjaman, M., & Aukema, H. M. (2015). Advances in our understanding of oxylipins derived from dietary PUFAs. Advances in Nutrition, 6, 513–540.
Giera, M., Ioan-Facsinay, A., Toes, R., Gao, F., Dalli, J., Deelder, A. M., Serhan, C. N., & Mayboroda, O. A. (2012). Lipid and lipid mediator profiling of human synovial fluid in rheumatoid arthritis patients by means of LC-MS/MS. Biochimica et Biophysica Acta, 1821, 1415–1424.
Gioxari, A., Kaliora, A. C., Marantidou, F., & Panagiotakos, D. P. (2018). Intake of omega-3 polyunsaturated fatty acids in patients with rheumatoid arthritis: A systematic review and meta-analysis. Nutrition, 45, 114–124.
Henneke, D. R., Potter, G. D., Kreider, J. L., & Yeates, B. F. (1983). Relationship between condition score, physical measurements and body fat percentage in mares. Equine Veterinary Journal, 15, 371–372.
Hess, T., Braun, S., & Herkelman, K. (2019). The effects of various levels of docosahexaenoic acid oninflammatory markers in conditioned horses during lactatethreshold tests. Journal of Equine Veterinary Science, 72, 64–71.
Hess, T., Ross-Jones, T., & De Zootecnia, R. B. (2014). Omega-3 fatty acid supplementation in horses Dietary n-3 and n-6 long-chain polyunsaturated fatty acids (PUFA). Revista Brasileira de Zootecnia, 43, 677–683.
Hess, T. M., Rexford, J. K., Hansen, D. K., Harris, M., Schauermann, N., Ross, T., Engle, T. E., Allen, K. G. D., & Mulligan, C. M. (2012). Effects of two different dietary sources of long chain omega-3, highly unsaturated fatty acids on incorporation into the plasma, red blood cell, and skeletal muscle in horses. Journal of Animal Science, 90, 3023–3031.
Huang, L. S., Hung, N. D., Sok, D. E., & Kim, M. R. (2010). Lysophosphatidylcholine containing docosahexaenoic acid at the sn-1 position is anti-inflammatory. Lipids, 45, 225–236.
Hung, N. D., Kim, M. R., & Sok, D. E. (2011a). 2-Polyunsaturated acyl lysophosphatidylethanolamine attenuates inflammatory response in zymosan A-induced peritonitis in mice. Lipids, 46, 893–906.
Hung, N. D., Kim, M. R., & Sok, D. E. (2011b). Mechanisms for anti-inflammatory effects of 1-[15(S)-hydroxyeicosapentaenoyl] lysophosphatidylcholine, administered intraperitoneally, in zymosan A-induced peritonitis. British Journal of Pharmacology, 162, 1119–1135.
Hung, N. D., Kim, M. R., & Sok, D. E. (2011c). Oral administration of 2-docosahexaenoyl lysophosphatidylcholine displayed anti-inflammatory effects on zymosan A-induced peritonitis. Inflammation, 34, 147–160.
Khol-Parisini, A., van den Hoven, R., Leinker, S., Hulan, H. W., & Zentek, J. (2007). Effects of feeding sunflower oil or seal blubber oil to horses with recurrent airway obstruction. Canadian Journal of Veterinary Research, 71, 59–65.
King, S. S., Abughazaleh, A. A., Webel, S. K., & Jones, K. L. (2008). Circulating fatty acid profiles in response to three levels of dietary omega-3 fatty acid supplementation in horses. Journal of Animal Science, 86, 1114–1123.
Kosinska, M. K., Liebisch, G., Lochnit, G., Wilhelm, J., Klein, H., Kaesser, U., Lasczkowski, G., Rickert, M., Schmitz, G., & Steinmeyer, J. (2013). A lipidomic study of phospholipid classes and species in human synovial fluid. Arthritis and Rheumatism, 65, 2323–2333.
Kremer, J. M., Lawrence, D. A., Petrillo, G. F., Litts, L. L., Mullaly, P. M., Rynes, R. I., Stocker, R. P., Parhami, N., Greenstein, N. S., Fuchs, B. R., & Mathur, A. (1995). Effects of high-dose fish oil on rheumatoid arthritis after stop** nonsteroidal antiinflammatory drugs. Clinical and immune correlates. Arthritis and Rheumatism, 38, 1107–1114.
Lagarde, M., Bernoud, N., Thies, F., Brossard, N., Lemaitre-Delaunay, D., Croset, M., & Lecerf, J. (2001). Lysophosphatidylcholine as a carrier of docosahexaenoic acid to target tissues. World Review of Nutrition and Dietetics, 88, 173–177.
Leguillette, R. (2003). Recurrent airway obstruction–heaves. The Veterinary Clinics of North America. Equine Practice, 19, 63–86.
Leveille, P., Chouinard-Watkins, R., Windust, A., Lawrence, P., Cunnane, S. C., Brenna, J. T., & Plourde, M. (2017). Metabolism of uniformly labeled (13)C-eicosapentaenoic acid and (13)C-arachidonic acid in young and old men. American Journal of Clinical Nutrition, 106, 467–474.
Levy, B. D., & Serhan, C. N. (2014). Resolution of acute inflammation in the lung. Annual Review of Physiology, 76, 467–492.
Manhart, D. R., Scott, B. D., Gibbs, P. G., Coverdale, J. A., Eller, E. M., Honnas, C. M., & Hood, D. M. (2009). Markers of inflammation in arthritic horses fed omega-3 fatty acids. The Professional Animal Scientist., 25, 155–160.
Nogradi, N., Couetil, L. L., Messick, J., Stochelski, M. A., & Burgess, J. R. (2015). Omega-3 fatty acid supplementation provides an additional benefit to a low-dust diet in the management of horses with chronic lower airway inflammatory disease. Journal of Veterinary Internal Medicine, 29, 299–306.
Ostermann, A. I., & Schebb, N. H. (2017). Effects of omega-3 fatty acid supplementation on the pattern of oxylipins: A short review about the modulation of hydroxy-, dihydroxy-, and epoxy-fatty acids. Food and Function, 8, 2355–2367.
Ostermann, A. I., West, A. L., Schoenfeld, K., Browning, L. M., Walker, C. G., Jebb, S. A., Calder, P. C., & Schebb, N. H. (2019). Plasma oxylipins respond in a linear dose-response manner with increased intake of EPA and DHA: Results from a randomized controlled trial in healthy humans. American Journal of Clinical Nutrition, 109, 1251–1263.
Ross-Jones, T. N., Hess, T., Rexford, J. K., Ahrens, N., Engle, T. E., & Hansen, D. K. (2014). Effects of omega-3 long chain fatty acid supplementation on equine synovial fluid fatty acid composition and prostaglandin E 2. Journal of Equine Veterinary Science, 34, 779–783.
Ross-Jones, T. N., McIlwraith, C. W., Kisiday, J. D., Hess, T. M., Hansen, D. K., & Black, J. (2016). Influence of an n-3long-chain polyunsaturated fatty acid-enriched diet on experimentally induced synovitis in horses. Journal of Animal Physiology and Animal Nutrition, 100, 565–577.
Rouser, G., Siakotos, A. N., & Fleischer, S. (1966). Quantitative analysis of phospholipids by thin-layer chromatography and phosphorus analysis of spots. Lipids, 1, 85–86.
Senftleber, N. K., Nielsen, S. M., Andersen, J. R., Bliddal, H., Tarp, S., Lauritzen, L., Furst, D. E., Suarez-Almazor, M. E., Lyddiatt, A., & Christensen, R. (2017). Marine oil supplements for arthritis pain: A systematic review and meta-analysis of randomized trials. Nutrients, 9, 42.
Serhan, C. N., Chiang, N., & Dalli, J. (2018). New pro-resolving n-3 mediators bridge resolution of infectious inflammation to tissue regeneration. Molecular Aspects of Medicine, 64, 1–17.
Serhan, C. N., & Levy, B. D. (2018). Resolvins in inflammation: Emergence of the pro-resolving superfamily of mediators. The Journal of Clinical Investigation, 128, 2657–2669.
Shahidi, F., & Ambigaipalan, P. (2018). Omega-3 polyunsaturated fatty acids and their health benefits. Annual Review of Food Science and Technology, 9, 345–381.
Shearer, G. C., & Walker, R. E. (2018). An overview of the biologic effects of omega-6 oxylipins in humans. Prostaglandins Leukotrienes and Essential Fatty Acids, 137, 26–38.
Souza, P. R., & Norling, L. V. (2016). Implications for eicosapentaenoic acid- and docosahexaenoic acid-derived resolvins as therapeutics for arthritis. European Journal of Pharmacology, 785, 165–173.
Walker, C. G., West, A. L., Browning, L. M., Madden, J., Gambell, J. M., Jebb, S. A., & Calder, P. C. (2015). The pattern of fatty acids displaced by EPA and DHA following 12 months supplementation varies between blood cell and plasma fractions. Nutrients, 7, 6281–6293.
Wood, P. L. (2017). Non-targeted lipidomics utilizing constant infusion high resolution ESI mass spectrometry. Neuromethods: Lipidomics. (p. 125). Springer Protocols.
Wood, P. L. (2019). Targeted lipidomics and metabolomics evaluations of cortical neuronal stress in schizophrenia. Schizophrenia Research, 212, 107–112.
Wood, P. L., & Woltjer, R. L. (2020). Electrospray ionization high resolution mass spectrometry of the chloride adducts of steroids, mono- and oligo-saccharides, xyloglycans, ceramides, gangliosides, and phenols. In P. L. Wood (Ed.), Neuromethods: Metabolomics. Springer Protocols In press.
Wood, P. L., Donohue, M. N., Cebak, J. E., Beckmann, T. G., Treece, M., Johnson, J. W., & Miller, L. M. J. (2018). Tear film amphiphilic and anti-inflammatory lipids in bovine pink eye. Metabolites. https://doi.org/10.3390/metabo8040081.
Wood, P. L., Scoggin, K., Ball, B. A., Lawrence, L. M., Troedsson, M. H., Brennan, K. M., Goedde, L., & Squires, E. L. (2017). Lipidomics evaluation of the effects of feeding a yeast-based supplement containing algal docosahexaenoic acid (DHA) on DHA-containing glycerophospholipids in stallion spermatozoa. Journal of Veterinary Medicine and Research, 4, 1071–1078.
Zierenberg, O., & Grundy, S. M. (1982). Intestinal absorption of polyenephosphatidylcholine in man. Journal of Lipid Research, 23, 1136–1142.
Acknowledgements
The authors would like to thank Dr Stacy Anderson and Alyssa Reed for their help with sample collection, Joey Morgan, Julie Wells, Ashley Caldwell, Saira Ortega Morales, Raeshelle Strobel, and Michelle Norden Sund for their assistance with laboratory analysis, and the Lincoln Memorial University–College of Veterinary Medicine student chapter of the American Association of Equine Practitioners for assisting with horse feeding and monitoring. The supplement used in this study was provided at no cost by Nutramax. The daily balancer pellet fed to horses throughout the study was provided at no cost by Purina.
Funding
This research was funded by Lincoln Memorial University.
Author information
Authors and Affiliations
Contributions
Study design (PLW, UC); Study conduct (PLW, PG, CLH, CMP, ALE, JRP, JJS, JSC, ATD, DJC, UC, EM), Data reduction (PLW, CLH, CMP, ALE, JRP, JJS, JSC, ATD, DJC), Manuscript (PLW, UC), Manuscript review (all contributors).
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Research involving human and animal participants
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Christmann, U., Hancock, C.L., Poole, C.M. et al. Dynamics of DHA and EPA supplementation: incorporation into equine plasma, synovial fluid, and surfactant glycerophosphocholines. Metabolomics 17, 41 (2021). https://doi.org/10.1007/s11306-021-01792-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11306-021-01792-5