Abstract
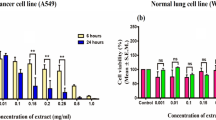
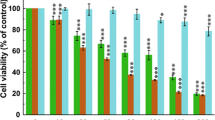
Biomolecules from Streptomyces spp. are emerging sources of natural drugs and have been focused on over the decade. The discovery of bioactive chemotherapeutic molecules from soil Streptomyces spp. has opened the medium for the search for natural drugs. In the current study, 8-HOQ was extracted and purified from soil Streptomyces spp. and was evaluated on A549 and BEAS cell lines. The apoptotic and caspase mediated pathways were evaluated using cell proliferation, dual fluorescent staining, migration, invasion and mRNA as well as protein quantification of apoptotic markers. In vitro cytotoxicity test revealed that 8-HOQ possesses potent cytotoxicity activities with IC50 values of 26 µM, 5 µM, 7.2 µM at 24 h, 48 h, and 72 h respectively against A549 lung cancer cell lines. The result also demonstrated that 8-HOQ from Streptomyces spp significantly inhibited the A549 lung cancer cell lines and activated the intrinsic pathways of apoptosis. The caspase-3 and caspase-8 activities were potentially elevated in 8-HOQ treated A549 cell lines and confirmed that 8-HOQ mediated A549 cancer cell death through the intrinsic pathway. The results explored caspase-mediated apoptosis as a mechanism underlying the inhibition of cancer cell viability in a dose-dependent manner. The expression of P53, BCL2 and STAT3 were inhibited in A549 cell lines and confirmed the metastasis inhibitory potential of 8-HOQ by blocking migration and invasion in A549 cell lines. These results indicated that 8-HOQ from Streptomyces spp. potentially inhibited growth and migration of A549 lung cancer cell lines.







Similar content being viewed by others
References
Allemailem KS (2021) Antimicrobial potential of naturally occurring bioactive secondary metabolites. J Pharm Bioallied Sci 13(2):155–162
Almuhayawi MS, Mohamed MSM, Abdel-Mawgoud M, Selim S, Al Jaouni SK, AbdElgawad H (2021) Bioactive potential of several actinobacteria isolated from microbiologically barely explored desert habitat, Saudi Arabia. Biology 10:235. https://doi.org/10.3390/biology10030235
Ayswaria R, Vasu V, Krishna R (2020) Diverse endophytic Streptomyces species with dynamic metabolites and their meritorious applications: a critical review. Crit Rev Microbiol 46(6):750–758
Bai L, Zhou H, Xu R, Zhao Y, Chinnaswamy K, McEachern D, Chen J, Yang CY, Liu Z, Wang M, Liu L, Jiang H, Wen B, Kumar P, Meagher JL, Sun D, Stuckey JA, Wang S (2019) A potent and selective small-molecule degrader of STAT3 achieves complete tumor regression in vivo. Cancer Cell 36(5):498-511.e17
Baud MGJ, Bauer MR, Verduci L, Dingler FA, Patel KJ, Horil Roy D, Joerger AC, Fersht AR (2018) Aminobenzothiazole derivatives stabilize the thermos labile p53 cancer mutant Y220C and show anticancer activity in p53–Y220C cell lines. Eur J Med Chem 152:101–114
Carmeliet P, Baes N (2008) Metabolism and therapeutic angiogenesis. Engl J Med 358:2511–2512
Dang YF, Yang SH, Jiang XN, Gong FL, Yang XX, Cheng YN, Guo XL (2021) Combination treatment strategies with a focus on rosiglitazone and adriamycin for insulin resistant liver cancer. J Drug Target 29(3):336–348
Duraipandiyan V, Sasi AH, Islam VIH, Valanarasu M, Ignacimuthu S (2010) Antimicrobial properties of actinomycetes from the soil of Himalaya. J Mycol Méd 20(1):15–20
Graf E, Schneider K, Nicholson G, Ströbele M, Jones AL, Goodfellow M, Beil W, Fiedler HP (2007) Elloxazinones A and B, new aminophenoxazinones from Streptomyces griseus Acta 2871. J Antibiot 60:277–284
Hwang SY, Lee SH, Song KJ, Kim YP, Kawahara K (1998) Purification and characterization of antistaphylococcal sub- stance from Pseudomonas sp.KUH-001. J Microbiol Biotechnol 8:111–118
Islam VI, Saravanan S, Ignacimuthu S (2014) Microbicidal and anti-inflammatory effects of Actinomadura spadix (EHA-2) active metabolites from Himalayan soils. India World J Microbiol Biotechnol 30(1):9–18
Joseph DB, Paulraj MG, Ignacimuthu S et al (2016) Antimicrobial activity of soil actinomycetes isolated from Western Ghats in Tamil Nadu. India J Bacteriol Mycol 3(2):224–232
Kenawy E (2001) Biologically active polymers. IV. Synthesis and antimicrobial activity of polymers containing 8-HOQ moiety. J Appl Polymer Sci 82:1364–1374
Khalil HE, Ibrahim H.-I.M, Ahmed EA, Emeka PM, Alhaider IA (2022) Orientin, a Bio-Flavonoid from Trigonella hamosa L., Regulates COX-2/PGE-2 in A549 Cell Lines via miR-26b and miR-146a. Pharmaceuticals 15:154
Kumar P, Chauhan A, Kumar M, Kuanr BK, Kundu A, Solanki R, Kapur MK (2021) In vitro and in silico anticancer potential analysis of Streptomyces spp. extract against human lung cancer cell line. 3 Biotech 11(6):254
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 25:402–408
Mack SB, Zeeck A (1987) Secondary metabolites by chemical screening I. Calcium 3-hydroxyquinoline-2-carboxylate from a Streptomyces. J Antibiot 40:953–960
Narayana KJ, Prabhakar P, Vijayalakshmi M, Venkateswarlu Y, Krishna PS (2008) Study on bioactive compounds from Streptomyces sp. ANU 6277. Pol J Microbiol 57(1):35–9
Newman DJ, Cragg GM (2016) Natural products as sources of new drugs from 1981 to 2014. J Nat Prod 79:629–661. https://doi.org/10.1021/acs.jnatprod.5b01055
Njenga WP, Mwaura FB, Wagacha JM, Gathuru EM (2017) Methods of isolating actinomycetes from the soils of Menengai Crater in Kenya. Arch Clin Microbiol 8:3. https://doi.org/10.4172/1989-8436.100045
Olano C, Méndez C, Salas JA (2009) Antitumor compounds from actinomycetes: from gene clusters to new derivatives by combinatorial biosynthesis. Nat Prod Rep 26:628–660
Orchel A, Dzierzewicz Z, Parfiniewicz B, Weglarz L, Wilczok T (2005) Butyrate induced differentiation of colon cancer cells is PKC and JNK dependent. Dig Dis Sci 50:490–498
Rather SA, Kumar S, Rah B, Arif M, Ali A, Qazi P (2014) A potent cytotoxic metabolite from terrestrial actinomycete, Streptomyces collinus. Med Chem Res 23:382–387
Sanner MF (1999) Python: a programming language for software integration and development. J Mol Graph Model 1:57–61
Senthil KV, Kumaresan S, Tamizh MM, Hairul-Islam MI, Thirugnanasambantham K (2019) Anticancer potential of NF-κB targeting apoptotic molecule “flavipin” isolated from endophytic Chaetomium globosum. Phytomedicine 61:152830
Sepúlveda P, Encabo A, Carbonell-Uberos F et al (2007) BCL-2 expression is mainly regulated by JAK/STAT3 pathway in human CD34+ hematopoietic cells. Cell Death Differ 14:378–380. https://doi.org/10.1038/sj.cdd.4402007
Shaaban K, Shepherd M, Ahmed T et al (2012) Pyramidamycins A-D and 3-hydroxyquinoline-2-carboxamide; cytotoxic benzamides from Streptomyces sp. DGC1. J Antibiot 65:615–622. https://doi.org/10.1038/ja.2012.81
Shah AM, Wani A, Qazi PH, Rehman S, Mushtaq S, Ali SA, Hussain A, Shah A, Qazi AK, Makhdoomi US (2016) Isolation and characterization of alborixin from Streptomyces scabrisporus: a potent cytotoxic agent against human colon (HCT-116) cancer cells. Chem Biol Interact 256:198–208
Song X, Dilly AK, Kim SY, Choudry HA, Lee YJ (2014) Rapamycin-enhanced mitomycin C-induced apoptotic death is mediated through the S6K1-Bad-Bak pathway in peritoneal carcinomatosis. Cell Death Dis 5(6):e1281
Undabarrena A, Beltrametti F, Claverías FP, González M, Moore ERB, Seeger M, Cámara B (2016) Exploring the diversity and antimicrobial potential of marine actinobacteria from the Comau Fjord in Northern Patagonia. Chile Front Microbiol 7:1–16. https://doi.org/10.3389/fmicb.2016.01135
Valipour B, Mohammadi SM, Abedelahi A, Maragheh BFA, Naderali E, Dehnad A, Charoudeh HN (2018) Culture filtrate ether extracted metabolites from Streptomyces levis ABRIINW111 increased apoptosis and reduced proliferation in acute lymphoblastic leukemia. Biomed Pharm 108:216–223.
Wei Z, Xu C, Wang J, Lu F, Bie X, Lu Z (2017) Identification and characterization of Streptomyces flavogriseus NJ-4 as a novel producer of actinomycin D and holomycin. PeerJ 5:e3601
Yang L et al (2014) Interaction between 8-hydroxyquinoline ruthenium (II) complexes and basic fibroblast growth factors (bFGF): inhibiting angiogenesis and tumor growth through ERK and AKT signaling pathways. Metallomics 6(3):518–531
You BY, Wang YH, Kuo ML (1999) Evaluation of geno- toxicity and carcinogenicity on fungicide, copper 8-hydroxyquino- linate, by short-term tests. Zhiwu Baohu Xuehui Huikan (chin) 41:119–130
Zhang XG, Liu ZY, Liu JW, Zeng YL, Guo GJ, Sun QY (2017) Antitumor activity of a Rhodococcus sp. Lut0910 isolated from polluted soil. Tumour Biol 39(6):1010428317711661
Author information
Authors and Affiliations
Contributions
JB: Experimental design, troubleshoot, perform molecule extraction, cell culture experiments, data curation, initial draft manuscript preparation, editing, final corrections. MP: Data curation, final draft manuscript preparation, editing, final corrections. CM: Data curation, final draft manuscript preparation, editing, final corrections. HI: Experimental design, troubleshoot, perform cell culture experiments, data curation, draft manuscript preparation, editing. KT: Perform fluorescent experiments, molecular docking, initial draft preparation and corrections. AS: mRNA and protein experiments, final draft preparation and corrections. SF: Final draft corrections, statistics, data curation and final editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.







Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Balthazar, J.D., Soosaimanickam, M.P., Emmanuel, C. et al. 8-Hydroxyquinoline a natural chelating agent from Streptomyces spp. inhibits A549 lung cancer cell lines via BCL2/STAT3 regulating pathways. World J Microbiol Biotechnol 38, 182 (2022). https://doi.org/10.1007/s11274-022-03368-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11274-022-03368-4