Abstract
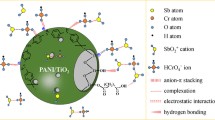
A hybrid composite, denoted as arginine-functionalized polyaniline@jujube shells (Arg-PANI@JS), was designed by oxidative polymerization of monomer-aniline on the surface of jujube shells matrix in the occurrence of arginine. This adsorbent was subsequently characterized and used in hexavalent chromium Cr(VI) adsorption assays. An experimental batch adsorption setup was utilized to assess the effectiveness of the newly developed adsorbent in detoxifying Cr(VI) ions from the solution. The experimental results were successfully anticipated by a pseudo-second-order model (PSO) and the Freundlich isotherm with a maximum uptake capacity of 1142.86 mg.g−1. The thermodynamic investigation showed that the process was both spontaneous and endothermic. The pivotal driving force behind elucidating the binding mechanism of Cr(VI) species was determined to be electrostatic interactions. Furthermore, our assessment demonstrated that the Arg-PANI@JS composite can be readily regenerated using a NaOH solution and effectively reutilized for removing Cr(VI) from aqueous solutions. Consequently, these findings underscore the promising practical utilization of the Arg-PANI@JS composite in wastewater treatment.







Similar content being viewed by others
Data Availability
The data supporting the findings of this study are available within the manuscript or available from the corresponding author upon reasonable request.
References
Ai, L., Jiang, J., & Zhang, R. (2010). Uniform polyaniline microspheres: A novel adsorbent for dye removal from aqueous solution. Synthetic Metals, 160(7–8), 762–767. https://doi.org/10.1016/j.synthmet.2010.01.017
Akti, F., & Okur, M. (2018). The removal of acid violet 90 from aqueous solutions using PANI and PANI/Clinoptilolite composites: Isotherm and kinetics. Journal of Polymers and the Environment, 26(11), 4233–4242. https://doi.org/10.1007/s10924-018-1297-1
Ali, A., Ajaz Hussain, M., Abbas, A., Tahir Haseeb, M., Azhar, I., Muhammad, G., et al. (2023). Succinylated Salvia spinosa hydrogel: Modification, characterization, cadmium-uptake from spiked high-hardness groundwater and statistical analysis of sorption data. Journal of Molecular Liquids, 376, 121438. https://doi.org/10.1016/j.molliq.2023.121438
Ansari, M. O., Kumar, R., Ansari, S. A., Ansari, S. P., Barakat, M. A., Alshahrie, A., & Cho, M. H. (2017). Anion selective pTSA doped polyaniline@graphene oxide-multiwalled carbon nanotube composite for Cr(VI) and Congo red adsorption. Journal of Colloid and Interface Science, 496, 407–415. https://doi.org/10.1016/j.jcis.2017.02.034
Ba Mohammed, B., Hsini, A., Abdellaoui, Y., Abou Oualid, H., Laabd, M., El Ouardi, M., et al. (2020). Fe-ZSM-5 zeolite for efficient removal of basic Fuchsin dye from aqueous solutions: Synthesis, characterization and adsorption process optimization using BBD-RSM modeling. Journal of Environmental Chemical Engineering, 8(5), 104419. https://doi.org/10.1016/j.jece.2020.104419
Ballav, N., Choi, H. J., Mishra, S. B., & Maity, A. (2014). Synthesis, characterization of Fe3O4@glycine doped polypyrrole magnetic nanocomposites and their potential performance to remove toxic Cr(VI). Journal of Industrial and Engineering Chemistry, 20(6), 4085–4093. https://doi.org/10.1016/j.jiec.2014.01.007
Bhaumik, M., Maity, A., Srinivasu, V. V., & Onyango, M. S. (2011). Enhanced removal of Cr(VI) from aqueous solution using polypyrrole/Fe3O4 magnetic nanocomposite. Journal of Hazardous Materials, 190(1–3), 381–390. https://doi.org/10.1016/j.jhazmat.2011.03.062
Bouargane, B., Oubelhas, I., Moreno, S. P., Biyoune, M. G., Bakiz, B., Bolivar, J. P., & Atbir, A. (2023). Process of preparing chloride-free KNS compound fertilizers from phosphogypsum waste using a quaternary phase diagram. Process Safety and Environmental Protection, 177, 995–1005. https://doi.org/10.1016/j.psep.2023.07.037
Bulletin official N° 6199”. (2013). ISSN 0851 – 1217.
Chafai, H., Laabd, M., Elbariji, S., Bazzaoui, M., & Albourine, A. (2017). Study of congo red adsorption on the polyaniline and polypyrrole. Journal of Dispersion Science and Technology, 38(6), 832–836. https://doi.org/10.1080/01932691.2016.1207185
Chigondo, M., Paumo, H. K., Bhaumik, M., Pillay, K., & Maity, A. (2019). Magnetic arginine-functionalized polypyrrole with improved and selective chromium(VI) ions removal from water. Journal of Molecular Liquids, 275, 778–791. https://doi.org/10.1016/j.molliq.2018.11.032
El Messaoudi, N., El Khomri, M., Dbik, A., Bentahar, S., Lacherai, A., & Bakiz, B. (2016). Biosorption of Congo red in a fixed-bed column from aqueous solution using jujube shell: Experimental and mathematical modeling. Journal of Environmental Chemical Engineering, 4(4), 3848–3855. https://doi.org/10.1016/j.jece.2016.08.027
Fiol, N., & Villaescusa, I. (2009). Determination of sorbent point zero charge: Usefulness in sorption studies. Environmental Chemistry Letters, 7(1), 79–84. https://doi.org/10.1007/s10311-008-0139-0
Gong, K., Hu, Q., **ao, Y., Cheng, X., Liu, H., Wang, N., et al. (2018). Triple layered core–shell ZVI@carbon@polyaniline composite enhanced electron utilization in Cr( vi ) reduction. Journal of Materials Chemistry A, 6(24), 11119–11128. https://pubs.rsc.org/en/content/articlelanding/2018/ta/c8ta03066a
Han, X., Liu, Y., **ong, L., Huang, H., Zhang, Q., Li, L., et al. (2019). Facile Assembly of polyaniline/graphene oxide composite hydrogels as adsorbent for Cr(VI) removal. Polymer Composites, 40(S2). https://doi.org/10.1002/pc.25161
Hsini, A., Essekri, A., Aarab, N., Laabd, M., Ait Addi, A., Lakhmiri, R., & Albourine, A. (2020a). Elaboration of novel polyaniline@Almond shell biocomposite for effective removal of hexavalent chromium ions and Orange G dye from aqueous solutions. Environmental Science and Pollution Research, 27(13), 15245–15258. https://doi.org/10.1007/s11356-020-08039-1
Hsini, A., Naciri, Y., Laabd, M., El Ouardi, M., Ajmal, Z., Lakhmiri, R., et al. (2020b). Synthesis and characterization of arginine-doped polyaniline/walnut shell hybrid composite with superior clean-up ability for chromium (VI) from aqueous media: Equilibrium, reusability and process optimization. Journal of Molecular Liquids, 316, 113832. https://doi.org/10.1016/j.molliq.2020.113832
Hussain, M. A., Abbas, A., Habib, M. G., Ali, A., Farid-ul-Haq, M., Hussain, M., et al. (2021). Adsorptive removal of Ni(II) and Co(II) from aqueous solution using succinate-bonded polysaccharide isolated from Artemisia vulgaris seed mucilage. Desalination and Water Treatment, 231, 182–195. https://doi.org/10.5004/dwt.2021.27468
Imgharn, A., Aarab, N., Hsini, A., Naciri, Y., Elhoudi, M., Haki, M. A., et al. (2022). Application of calcium alginate-PANI@sawdust wood hydrogel bio-beads for the removal of orange G dye from aqueous solution. Environmental Science and Pollution Research, 29(40), 60259–60268. https://doi.org/10.1007/s11356-022-20162-9
Imgharn, A., Laabd, M., Naciri, Y., Hsini, A., Mahir, F.-Z., Zouggari, H., & Albourine, A. (2023). Insights into the performance and mechanism of PANI@Hydroxapatite-Montmorillonite for hexavalent chromium Cr (VI) detoxification. Surfaces and Interfaces, 36, 102568. https://doi.org/10.1016/j.surfin.2022.102568
Imgharn, A., Sun, T., Nicolle, J., Naciri, Y., Hsini, A., Albourine, A., & Ania, A. (2024). A simple approach to prepare a C3N4/MoO3 heterojunction with improved photocatalytic performance for the degradation of methylparaben. Catalysts. https://doi.org/10.3390/catal14030170
Iqbal, A., Yusaf, A., Usman, M., Hussain Bokhari, T., & Mansha, A. (2023). Insight into the degradation of different classes of dyes by advanced oxidation processes; a detailed review. International Journal of Environmental Analytical Chemistry, 1–35. https://doi.org/10.1080/03067319.2022.2125312
Jiang, W., Cai, Q., Xu, W., Yang, M., Cai, Y., Dionysiou, D. D., & O’Shea, K. E. (2014). Cr(VI) Adsorption and reduction by humic acid coated on magnetite. Environmental Science & Technology, 48(14), 8078–8085. https://doi.org/10.1021/es405804m
Jobby, R., Jha, P., Yadav, A. K., & Desai, N. (2018). Biosorption and biotransformation of hexavalent chromium [Cr(VI)]: A comprehensive review. Chemosphere, 207, 255–266. https://doi.org/10.1016/j.chemosphere.2018.05.050
Khandegar, V., & Saroha, A. K. (2013). Electrocoagulation for the treatment of textile industry effluent – A review. Journal of Environmental Management, 128, 949–963. https://doi.org/10.1016/j.jenvman.2013.06.043
Kumar, P. A., Chakraborty, S., & Ray, M. (2008). Removal and recovery of chromium from wastewater using short chain polyaniline synthesized on jute fiber. Chemical Engineering Journal, 141(1–3), 130–140. https://doi.org/10.1016/j.cej.2007.11.004
Kumar, R., Barakat, M. A., & Alseroury, F. A. (2017). Oxidized g-C3N4/polyaniline nanofiber composite for the selective removal of hexavalent chromium. Scientific Reports, 7(1), 12850. https://doi.org/10.1038/s41598-017-12850-1
Kumari, B., Tiwary, R. K., & Yadav, M. (2022). Non linear regression analysis and RSM modeling for removal of Cr (VI) from aqueous solution using PANI@WH composites. Materials Chemistry and Physics, 290, 126457. https://doi.org/10.1016/j.matchemphys.2022.126457
Laabd, M., Hallaoui, A., Aarb, N., Essekri, A., Eljazouli, H., Lakhmiri, R., & Albourine, A. (2019). Removal of polycarboxylic benzoic acids using polyaniline-polypyrrole copolymer: Experimental and DFT studies. Fibers and Polymers, 20(5), 896–905. https://doi.org/10.1007/s12221-019-8212-9
Laabd, M., Imgharn, A., Hsini, A., Naciri, Y., Mobarak, M., Szunerits, S., et al. (2022). Efficient detoxification of Cr(VI)-containing effluents by sequential adsorption and reduction using a novel cysteine-doped PANi@faujasite composite: Experimental study supported by advanced statistical physics prediction. Journal of Hazardous Materials, 422, 126857. https://doi.org/10.1016/j.jhazmat.2021.126857
Laktif, T., Imgharn, A., Hsini, A., Elhoudi, M., Aarab, N., Laabd, M., et al. (2022). Sunflower seed shells@polyaniline: a novel composite for the removal of pharmaceutical pollutants from wastewater. International Journal of Environmental Analytical Chemistry, 1–18. https://doi.org/10.1080/03067319.2022.2060086
Lu, C., Yang, J., Khan, A., Yang, J., Li, Q., & Wang, G. (2022). A highly efficient technique to simultaneously remove acidic and basic dyes using magnetic ion-exchange microbeads. Journal of Environmental Management, 304, 114173. https://doi.org/10.1016/j.jenvman.2021.114173
Mahir, F.-Z., Zouggari, H., Imgharn, A., Hsini, A., Aarab, N., Laabd, M., et al. (2023). Facile elaboration of arginine-functionalized PANI@graphitic carbon nitride for efficient removal of hexavalent chromium. Diamond and Related Materials, 136, 109963. https://doi.org/10.1016/j.diamond.2023.109963
Naciri, Y., Chennah, A., Jaramillo-Páez, C., Navío, J. A., Bakiz, B., Taoufyq, A., et al. (2019). Preparation, characterization and photocatalytic degradation of Rhodamine B dye over a novel Zn3(PO4)2/BiPO4 catalyst. Journal of Environmental Chemical Engineering, 7(3), 103075. https://doi.org/10.1016/j.jece.2019.103075
Pavan, F. A., Lima, E. C., Dias, S. L. P., & Mazzocato, A. C. (2008). Methylene blue biosorption from aqueous solutions by yellow passion fruit waste. Journal of Hazardous Materials, 150(3), 703–712. https://doi.org/10.1016/j.jhazmat.2007.05.023
Pehlivan, E., Tran, H. T., Ouédraogo, W. K. I., Schmidt, C., Zachmann, D., & Bahadir, M. (2013). Sugarcane bagasse treated with hydrous ferric oxide as a potential adsorbent for the removal of As(V) from aqueous solutions. Food Chemistry, 138(1), 133–138. https://doi.org/10.1016/j.foodchem.2012.09.110
Rathnayake, S. I., Martens, W. N., **, Y., Frost, R. L., & Ayoko, G. A. (2017). Remediation of Cr (VI) by inorganic-organic clay. Journal of Colloid and Interface Science, 490, 163–173. https://doi.org/10.1016/j.jcis.2016.11.070
Sunil, K., Sherugar, P., Rao, S., Lavanya, C., Balakrishna, G. R., Arthanareeswaran, G., & Padaki, M. (2021). Prolific approach for the removal of dyes by an effective interaction with polymer matrix using ultrafiltration membrane. Journal of Environmental Chemical Engineering, 9(6), 106328. https://doi.org/10.1016/j.jece.2021.106328
Tu, B., Wen, R., Wang, K., Cheng, Y., Deng, Y., Cao, W., et al. (2020). Efficient removal of aqueous hexavalent chromium by activated carbon derived from Bermuda grass. Journal of Colloid and Interface Science, 560, 649–658. https://doi.org/10.1016/j.jcis.2019.10.103
Uğurlu, M. (2009). Adsorption of a textile dye onto activated sepiolite. Microporous and Mesoporous Materials, 119(1–3), 276–283. https://doi.org/10.1016/j.micromeso.2008.10.024
Wang, Y., Peng, C., Padilla-Ortega, E., Robledo-Cabrera, A., & López-Valdivieso, A. (2020). Cr(VI) adsorption on activated carbon: Mechanisms, modeling and limitations in water treatment. Journal of Environmental Chemical Engineering, 8(4), 104031. https://doi.org/10.1016/j.jece.2020.104031
Wang, F., Zhang, Y., Fang, Q., Li, Z., Lai, Y., & Yang, H. (2021). Prepared PANI@nano hollow carbon sphere adsorbents with lappaceum shell like structure for high efficiency removal of hexavalent chromium. Chemosphere, 263, 128109. https://doi.org/10.1016/j.chemosphere.2020.128109
Xu, Y., Chen, J., Chen, R., Yu, P., Guo, S., & Wang, X. (2019). Adsorption and reduction of chromium(VI) from aqueous solution using polypyrrole/calcium rectorite composite adsorbent. Water Research, 160, 148–157. https://doi.org/10.1016/j.watres.2019.05.055
Yu, X., Tong, S., Ge, M., Wu, L., Zuo, J., Cao, C., & Song, W. (2013). Adsorption of heavy metal ions from aqueous solution by carboxylated cellulose nanocrystals. Journal of Environmental Sciences, 25(5), 933–943. https://doi.org/10.1016/S1001-0742(12)60145-4
Zhan, Y., Wan, X., He, S., & He, Y. (2018). Sulfonated poly(arylene ether nitrile)/polypyrrole core/shell nanofibrous mat: An efficient absorbent for the removal of hexavalent chromium from aqueous solution. Journal of Chemical Technology & Biotechnology, 93(5), 1432–1442. https://doi.org/10.1002/jctb.5511
Zhang, B., Luan, L., Gao, R., Li, F., Li, Y., & Wu, T. (2017). Rapid and effective removal of Cr(VI) from aqueous solution using exfoliated LDH nanosheets. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 520, 399–408. https://doi.org/10.1016/j.colsurfa.2017.01.074
Zhou, T., Li, C., **, H., Lian, Y., & Han, W. (2017). Effective Adsorption/Reduction of Cr(VI) Oxyanion by Halloysite@Polyaniline Hybrid Nanotubes. ACS Applied Materials & Interfaces, 9(7), 6030–6043. https://doi.org/10.1021/acsami.6b14079
Funding
We declare that there was no external funding received for the research presented in this manuscript.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of Interest
The authors have declared no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Bacha, K.A.E., Imgharn, A., Hsini, A. et al. Arginine-Polyaniline Embedded Jujube Shells Composite for Outstanding Cr(VI) Detoxification from aqueous solution. Water Air Soil Pollut 235, 380 (2024). https://doi.org/10.1007/s11270-024-07194-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-024-07194-2