Abstract
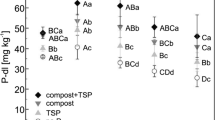
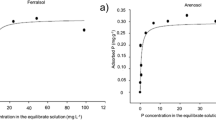
This study was designed to characterize the release of available P from different ratios of mono-ammonium phosphate:struvite (MAP:struvite) in soils as affected by the season in which it was applied, representing field conditions. A 63-day soil incubation evaluated release of soil test P (STP) as Olsen P from MAP:struvite ratios (0:0, 100:0, 75:25, 50:50, 37.5:62.5, 25:75, 12.5:87.5, 0:100) as temperature decreased (26 to 15 °C) and increased (12.4 to 22.6 °C) to mimic fall and spring temperatures, respectively. All treatments decreased in STP concentration from the beginning to end of both fall and spring incubations. The amount of decrease in STP during the incubation period was greater for the fall incubation compared to the spring incubation. After 63 days of the fall incubation, treatments 100:0 and 0:100 decreased the most from 22.9 to 12.1 mg STP kg−1 and 21.2 to 7.7 mg STP kg−1, respectively. After 63 days in the spring incubation, 100:0 and 0:100 decreased from 20.2 to 11.8 mg STP kg−1 and 18.1 to 14.0 mg STP kg−1. Overall results suggest that soil STP decreased in the range of 12 to 26 °C for soils fertilized with MAP:struvite mixtures, regardless of season.




Similar content being viewed by others
Data Availability
Data will be made availability upon request.
References
Achat, D. L., Sperandio, M., Daumer, M.-L., Santellani, A.-C., Prud’Homme, L., Akhtar, M., & Morel, C. (2014). Plant-availability of phosphorus recycled from pig manures and dairy effluents as assessed by isotopic labeling techniques. Geoderma, 232–234, 24–33.
Ahmed, S., Klassen, T. N., Keyes, S., Daly, M., Jones, D. L., Mavrogordato, M., Sinclair, I., & Roose, T. (2016). Imaging the interaction of roots and phosphate fertiliser granules using 4D X-ray tomography. Plant and Soil, 401, 125–134.
Bargaz, A., K. Lyamloili, M. Chtouki, Y. Zeroual, & D. Dhiba. (2018). Soil microbial resources for improving fertilizers efficiency in an integrated plant nutrient management system. Frontiers in Microbiology, 9, 1606. https://doi.org/10.3389/fmicb.2018.01606
Barrow, N. J. (1979). Three effects of temperature on the reactions between inorganic phosphate and soil. Journal of Soil Science, 30, 271–279.
Bowers, K. E., & Westerman, P. W. (2005). Performance of cone-shaped fluidized bed struvite crystallizers in removing phosphorus from wastewater. T. ASAE, 48(3), 1227–1234.
Bundy, L.G., H. Tunney, & Halvorson A.D. (2005). Agronomic aspects of phosphorus management. In: Phosphorus: Agriculture and the environment. American Society of Agronomy Inc., Crop Science Society of America Inc., Soil Science Society of America Inc.
Cabeza, R., Steingrobe, B., Römer, W., & Claassen, N. (2011). Effectiveness of recycled P products as P fertilizers, as evaluated in pot experiments. Nutrient Cycling in Agroecosystems, 91, 173–184.
Degryse, F., Baird, R., da Silva, R. C., & McLaughlin, M. J. (2017). Dissolution rate and agronomic effectiveness of struvite fertilizers–Effect of soil pH, granulation and base excess. Plant and Soil, 410, 139–152.
Fan, X. H., & Li, Y. C. (2010). Nitrogen release from slow-release fertilizers as affected by soil type and temperature. Soil Science Society of America Journal, 74, 1635–1641.
Gavlak, R., D. Horneck, & Miller R.O. (2005). Soil, plant and water reference methods for the western region (3rd ed.). WREP 125.
Grant, C. A., Flaten, D. N., Tomasiewicz, D. J., & Sheppard, S. C. (2001). The importance of early season phosphorus nutrition. Canadian Journal of Plant Science, 81, 211–224.
Hilt, K., Harrison, J., Bowers, K., Stevens, R., Bary, A., & Harrison, K. (2016). Agronomic response of crops fertilized with struvite derived from dairy manure. Water, Air, and Soil Pollution, 227, 388. https://doi.org/10.1007/s11270-016-3093-7
Horta, C. (2017). Bioavailability of phosphorus from composts and struvite in acid soils. Revista Brasileira De Engenharia Agricola e Ambiental, 21(7), 459–464.
Koenig, R.T., Horneck D. , Platt T. , Petersen P. , Stevens R. , Fransen S., & Brown B. (2009). Nutrient management guide for dryland and irrigated alfalfa in the inland Northwest. Pacific Northwest Extension PNW0611, Washington State University
Lunt, O. R., Kofranek, A. M., & Clark, S. B. (1964). Availability of minerals from magnesium ammonium phosphates. Agricultural and Food Chemistry, 12(6), 497–504.
Miller, R.O., Gavlak R., & Horneck D. (2013). Soil, plant and water reference methods for the western region (4th ed.). WREP 125.
Norberg, S., E. Mackey, S. Fransen, J. Harrison, D. Llewellyn, and L. Whitefield. (2018). Develo** practical phosphorus and potassium tissue test recommendations and utilizing struvite in modern alfalfa systems. National Alfalfa and Forage Alliance Final Project Report 2018 Grant. https://www.alfalfa.org/pdf/researchArticels/18Norberg-USAFRI-Final.pdf
Olsen, S.R., Cole C.V., Watanbe F.S., & Dean L.A. (1954). Estimation of available phosphorus in soils by extraction with sodium bicarbonate. U.S. Department of Agriculture Circular No. 939. USDA.
Rothbaum, H. P., & Rhode, A. G. (1976). Long-term leaching of nutrients from magnesium ammonium phosphate at various temperatures. N.Z. Journal of Experimental Agriculture, 4, 405–413.
Sheppard, S. C., & Racz, G. J. (1984). Effects of soil temperature on phosphorus extractability. I. Extraction and plant uptake of soil and fertilizer phosphorus. Canadian Journal of Soil Science, 64, 241–254.
Singh, B. B., & Jones, J. P. (1977). Phosphorus sorption isotherm for evaluating phosphorus requirements of lettuce at five temperature regimes. Plant and Soil, 46, 31–44.
Sun, D., Bi, Q., Li, K., Dai, P., Yu, Y., Zhou, W., Lv, T., Liu, X., Zhu, J., Zhang, Q., **, C., Lu, L., & Lin, X. (2017). Significance of temperature and water availability for soil phosphorus transformation and microbial community composition as affected by fertilizer sources. Biology and Fertility of Soils, 54(2), 229–241.
Talboys, P. J., Heppell, J., Roose, T., Healey, J. R., Jones, D. L., & Withers, P. J. A. (2016). Struvite: A slow-release fertilizer for sustainable phosphorus management? Plant and Soil, 401, 109–123.
Acknowledgements
I would like to add an acknowledgement saying: "The authors would like to acknowledge KUO Testing Labs, Inc., Othello, WA, USA for their technical support.
Funding
Financial support was provided through a USDA NRCS conservation innovation grant 69-3A75-17–51.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mackey, E., Harrison, J.H., Tao, H. et al. Effect of Application Season on Release of Available Phosphorus from Soil Receiving Mono-ammonium Phosphate or Struvite. Water Air Soil Pollut 232, 467 (2021). https://doi.org/10.1007/s11270-021-05396-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-021-05396-6