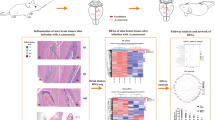
Abstract
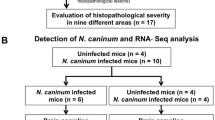
Toxoplasma gondii (T. gondii) is an obligate intracellular parasite of warm-blooded vertebrates. At present, High-throughput RNA sequencing analysis have made it possible to determine the role of effective genes in host immune response. The aim of the present study is to global transcriptome analysis of the brain of mice infected with T. gondii Tehran strain for the first time and also to evaluate the expression of effective genes in the chronic form of infection. RNA was extracted from the samples and the library was prepared and sequenced using the IlluminaNovaSeq 6000 system. After analyzing gene expression changes, the results were confirmed by real-time method. We found 125 genes that were significantly differentially expressed between infected and non-infected samples (p < 0.0005). Gene ontology analysis revealed that the expression of many genes is critical for pathways such as T cell receptor signaling pathway, Natural Killer cell mediated cytotoxicity, Lysosome and Apoptosis of the host. As infection with Tehran strain leads to chronic infection in mice, therefore, we investigated the genes effective in creating the chronic form of Toxoplasma infection. The comparative analysis of genes showed increases in the expression of genes ctla4, ccl4, cd3e, c3, lcn2, gbp5, usp18, cyba, tap1 and samhd1 in the in the infected sample, which highlights their role in causing chronic infection. RNA-seq provides a valuable tool for analyzing host transcriptomes, better understanding the parasite-host interaction, and develo** future drug and vaccine targets.













Similar content being viewed by others
Data availability
RNA-seq data obtained at NCBI were deposited at SRA under project accession number PRJNA910664.
References
Aliberti J, Valenzuela JG, Carruthers VB et al (2003) Molecular mimicry of a CCR5 binding-domain in the microbial activation of dendritic cells. Nat Immunol 4:485–490
Arimoto K, Löchte S, Stoner SA et al (2017) STAT2 is an essential adaptor in USP18-mediated suppression of type I interferon signaling. Nat Struct Mol Biol 24:279–289
Bottari NB, Schetinger MRC, Pillat MM et al (2019) Resveratrol as a therapy to restore neurogliogenesis of neural progenitor cells infected by Toxoplasma Gondii. Mol Neurobiol 56:2328–2338
Chandrasekaran S, Kochanowsky JA, Merritt EF et al (2022) IFN-γ stimulated murine and human neurons mount anti-parasitic defenses against the intracellular parasite Toxoplasma Gondii. Nat Commun 13:1–13
Chen L-F, Han X-L, Li F-X et al (2018) Comparative studies of Toxoplasma Gondii transcriptomes: insights into stage conversion based on gene expression profiling and alternative splicing. Parasit Vectors 11:1–11
Choi HG, Gao F-F, Zhou W et al (2020) The role of PI3K/AKT pathway and NADPH oxidase 4 in host ROS manipulation by Toxoplasma Gondii. Korean J Parasitol 58:237
Christiansen C, Maus D, Hoppenz E et al (2022) In vitro maturation of Toxoplasma Gondii bradyzoites in human myotubes and their metabolomic characterization. Nat Commun 13:1–15
Cockram PE, Kist M, Prakash S et al (2021) Ubiquitination in the regulation of inflammatory cell death and cancer. Cell Death Differ 28:591–605
Cowan MN, Sethi I, Harris TH (2022) Microglia in CNS Infections: insights from Toxoplasma Gondii and other pathogens. Trends Parasitol
Daher D, Shaghlil A, Sobh E et al (2021) Comprehensive overview of Toxoplasma Gondii-induced and associated Diseases. Pathogens 10:1351
de Lima Bessa G, de Almeida Vitor RW, dos Santos Martins-Duarte E (2021) Toxoplasma Gondii in South America: a differentiated pattern of spread, population structure and clinical manifestations. Parasitol Res 120:3065–3076
Dian S, Ganiem AR, Ekawardhani S (2022) Cerebral toxoplasmosis in HIV-infected patients: a review. Pathog Glob Health 1–10
Elzeky SM, Nabih N, Abdel-Magied AA, et al (2022) Seroprevalence and genetic characterization of toxoplasma gondii among children with neurodevelopmental disorders in Egypt. J Trop Med 2022
Fisch D, Clough B, Khan R et al (2021) Toxoplasma-proximal and distal control by GBPs in human macrophages. Pathog Dis 79:ftab058
Foroutan M, Dalvand S, Daryani A et al (2018) Rolling up the pieces of a puzzle: a systematic review and meta-analysis of the prevalence of toxoplasmosis in Iran. Alexandria J Med 54:189–196
Garfoot AL, Cervantes PW, Knoll LJ (2019) Transcriptional analysis shows a robust host response to Toxoplasma Gondii during early and late chronic Infection in both male and female mice. Infect Immun 87:e00024–e00019
He J-J, Ma J, Elsheikha HM et al (2016) Transcriptomic analysis of mouse liver reveals a potential hepato-enteric pathogenic mechanism in acute Toxoplasma gondii Infection. Parasit Vectors 9:1–13
Hu Z, Weng X, Xu C et al (2018) Metagenomic next-generation sequencing as a diagnostic tool for toxoplasmic encephalitis. Ann Clin Microbiol Antimicrob 17:1–6
Jia B, Lu H, Liu Q et al (2013) Genome-wide comparative analysis revealed significant transcriptome changes in mice after Toxoplasma Gondii Infection. Parasit Vectors 6:1–12
Khan IA, Moretto M (2022) Immune responses to Toxoplasma Gondii. Curr Opin Immunol 77:102226
Khan A, Shaik JS, Behnke M et al (2014) NextGen sequencing reveals short double crossovers contribute disproportionately to genetic diversity in Toxoplasma Gondii. BMC Genomics 15:1–15
Lu G, Zhou J, Zhao YH et al (2019) Transcriptome sequencing investigated the tumor-related factors changes after T. Gondii Infection. Front Microbiol 10:181
Mahmoudzadeh S, Nozad Charoudeh H, Marques CS et al (2021) The role of IL-12 in stimulating NK cells against Toxoplasma gondii Infection: a mini-review. Parasitol Res 120:2303–2309
Matta SK, Patten K, Wang Q et al (2018) NADPH oxidase and guanylate binding protein 5 restrict survival of avirulent type III strains of Toxoplasma Gondii in naive macrophages. MBio 9:e01393–e01318
McGettigan PA (2013) Transcriptomics in the RNA-seq era. Curr Opin Chem Biol 17:4–11
Miranda FJB, Rocha BC, Pereira MCA et al (2021) Toxoplasma Gondii-Induced neutrophil extracellular traps amplify the innate and adaptive response. MBio 12:e01307–e01321
Mousavi-Hasanzadeh M, Sarmadian H, Ghasemikhah R et al (2020) Evaluation of toxoplasma gondii Infection in western Iran: seroepidemiology and risk factors analysis. Trop Med Health 48:1–7
Pittman KJ, Aliota MT, Knoll LJ (2014) Dual transcriptional profiling of mice and Toxoplasma Gondii during acute and chronic Infection. BMC Genomics 15:1–19
Sasai M, Yamamoto M (2019) Innate, adaptive, and cell-autonomous immunity against Toxoplasma Gondii Infection. Exp Mol Med 51:1–10
Shallberg LA, Hunter CA (2021) Long live the king: Toxoplasma Gondii nucleomodulin inhibits necroptotic cell death. Cell Host Microbe 29:1165–1166
Sikorski PM, Commodaro AG, Grigg ME (2021) A protective and pathogenic role for complement during acute Toxoplasma gondii Infection. Front Cell Infect Microbiol 11:634610
Splitt SD, Souza SP, Valentine KM et al (2018) PD-L1, TIM-3, and CTLA-4 blockade fails to promote resistance to secondary Infection with virulent strains of Toxoplasma Gondii. Infect Immun 86:e00459–e00418
Suwancharoen C, Phuangsri C, Prakhammin K, Japa O (2022) Molecular detection and dense granule antigen 6 genoty** of feline Toxoplasma Gondii in Phayao, Thailand. Vet World 15
Tanaka S, Nishimura M, Ihara F et al (2013) Transcriptome analysis of mouse brain infected with Toxoplasma Gondii. Infect Immun 81:3609–3619
Umeda K, Tanaka S, Ihara F et al (2017) Transcriptional profiling of toll-like receptor 2-deficient primary murine brain cells during Toxoplasma Gondii Infection. PLoS ONE 12:e0187703
Yan Z, Yuan H, Wang J et al (2022) Four Chemotherapeutic compounds that Limit Blood-Brain-Barrier Invasion by Toxoplasma Gondii. Molecules 27:5572
Yu L, Shwab K, Hill RD et al (2019) Dynamics of gene expression in mice infected with different genotypes of Toxoplasma gondii
Acknowledgements
The authors would like to thank Kerman University of Medical Sciences (Kerman, Iran) for supporting this study.
Funding
This work was supported by Kerman University of Medical Sciences (No.1399.548).
Author information
Authors and Affiliations
Contributions
MA Conceptualization, Investigation, Methodology, Software, Writing – original draft, review & editing. ZB Project administration, Supervision, Writing - review & editing, Funding acquisition. AA Conceptualization, Project administration, Supervision, Writing - review & editing. MHB Methodology, review & editing. NZA Methodology, Writing - review & editing. A DWriting - review & editing. E A Methodology, Software. MM Methodology, Software. MA Methodology, Software. FZ review & editing. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Ethical approval
This study was approved by the Ethics Committee in accordance with the instructions of the Ethics Commission of Kerman University of Medical Sciences (Project ethic number: IR.KMU.REC.1399.548).
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Asadi, M., Babaei, Z., Afgar, A. et al. Brain –cyst-driven genes expression in Toxoplasma Gondii Tehran strain: a parasitic-immunogenicity assessment by dint of RNA-Seq. Vet Res Commun (2024). https://doi.org/10.1007/s11259-023-10241-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11259-023-10241-8