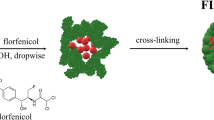
Abstract
As is the case with other veterinary antibiotics, florfenicol (FFC) faces certain limitations, such as low solubility in water, or the fact that it is reported to interfere with the immune response after some immunoprofilactic actions in livestock. Aiming to improve its efficacy and overall performance, FFC was loaded into a polymeric nanobased delivery system by succesfully using the emulsion-evaporation technique. The poly(lactic-co-glycolic acid) (PLGA) nanoparticles loaded with FFC were characterized in terms of size (101 ± 0.52 nm), zeta potential (26.80 ± 1.30 mV) and poly-dispersity index (0.061 ± 0.019). The achieved loading was 2.24 μg FFC/mg of NPs, with an entrapment efficiency of 7.9%. The antimicrobial effect, the anti-biofilm formation and the cytotoxicity properties of the NPs were evaluated. The results indicated a MIC decreased by ~97.13% for S. aureus, 99.33% for E.coli and 64.1% for P. aeruginosa when compared to free FFC. The minimum inhibitory concentration (MIC) obtained indicated the potential for using a significantly lower dose of florfenicol. The delivery system produced biofilm inhibition while showing no cytotoxic effects when tested on porcine primary fibroblasts and horse mesenchymal stem cells. These findings suggest that florfenicol can be improved and formulations optimized for use in veterinary medicine through its incorporation into a nanobased delivery system designed to release in a controlled manner over time.







Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Al-kafaween MA, Mohd Hilmi AB, Jaffar N, Al-Jamal HAN, Zahri MK (2019) Determination of optimum incubation time for formation of Pseudomonas aeruginosa and streptococcus pyogenes biofilms in microtiter plate. Bull Natl Res Centre 43(1). https://doi.org/10.1186/s42269-019-0131-9
Arcangioli MA, Leroy-Sétrin S, Martel JL, Chaslus-Dancla E (1999) A new chloramphenicol and florfenicol resistance gene flanked by two integron structures in salmonella typhimurium DT104. FEMS Microbiol Lett 174(2):327–332. https://doi.org/10.1016/S0378-1097(99)00161-5
Bertrand N, Leroux JC (2012) The journey of a drug-carrier in the body: an anatomo-physiological perspective. J Control Release 161(2):152–163. https://doi.org/10.1016/j.jconrel.2011.09.098
Bolton LF, Kelley LC, Lee MD, Fedorka-Cray PJ, Maurer JJ (1999) Detection of multidrug-resistant salmonella enterica serotype typhimurium DT104 based on a gene which confers cross-resistance to florfenicol and chloramphenicol. J Clin Microbiol 37(5):1348–1351. https://doi.org/10.1128/jcm.37.5.1348-1351.1999
Cerbu C, Kah M, White JC, Astete CE, Sabliov CM (2021) Fate of biodegradable engineered nanoparticles used in veterinary medicine as delivery systems from a one health perspective. Molecules 26(3). https://doi.org/10.3390/molecules26030523
Chevalier MT, Gonzalez J, Alvarez V (2015) Biodegradable polymeric microparticles as drug delivery devices. IFMBE Proc 49:187–190. https://doi.org/10.1007/978-3-319-13117-7_49
Costa GA, Rossatto FCP, Medeiros AW, Correa APF, Brandelli A, Frazzon APG, Da Motta A de S (2018) Evaluation antibacterial and antibiofilm activity of the antimicrobial peptide P34 against Staphylococcus aureus and Enterococcus faecalis. An Acad Bras Cienc 90(1):73–84. https://doi.org/10.1590/0001-3765201820160131
Danaei M, Dehghankhold M, Ataei S, Hasanzadeh Davarani F, Javanmard R, Dokhani A, Khorasani S, Mozafari MR (2018) Impact of particle size and polydispersity index on the clinical applications of lipidic nanocarrier systems. Pharmaceutics 10(2):1–17. https://doi.org/10.3390/pharmaceutics10020057
Devadasu VR, Bhardwaj V, Kumar MNVR (2013) Can controversial nanotechnology promise drug delivery? Chem Rev 113(3):1686–1735. https://doi.org/10.1021/cr300047q
Dowling PM (2002) Adverse drug reactions in horses. Clin Tech Equine Pract 1(2):58–67. https://doi.org/10.1053/ctep.2002.34868
Dupuis V, Cerbu C, Witkowski L, Potarniche AV, Timar MC, Żychska M, Sabliov CM (2022) Nanodelivery of essential oils as efficient tools against antimicrobial resistance: a review of the type and physical-chemical properties of the delivery systems and applications. In Drug Delivery, vol 29, issue 1. Taylor and Francis Ltd., pp. 1007–1024. https://doi.org/10.1080/10717544.2022.2056663
Economou V, Gousia P (2015) Agriculture and food animals as a source of antimicrobial-resistant bacteria. In Infection and drug resistance, vol 8. Dove Medical Press Ltd., pp. 49–61. https://doi.org/10.2147/IDR.S55778
Fernández-Alarcón C, Singer RS, Johnson TJ (2011) Comparative genomics of multidrug resistance-encoding incA/C plasmids from commensal and pathogenic escherichia coli from multiple animal sources. PLoS ONE 6(8). https://doi.org/10.1371/journal.pone.0023415
Fráguas-Eggenschwiler M, Eggenschwiler R, Söllner JH, Cortnumme L, Vondran FWR, Cantz T, Ott M, Niemann H (2021) Direct conversion of porcine primary fibroblasts into hepatocyte-like cells. Sci Rep 11(1). https://doi.org/10.1038/s41598-021-88727-1
Hu D, Cao S, Zhang G, **ao Y, Liu S, Shang Y (2017) Florfenicol-induced mitochondrial dysfunction suppresses cell proliferation and autophagy in fibroblasts. Sci Rep 7(1):1–13. https://doi.org/10.1038/s41598-017-13860-9
Hutchings M, Truman A, Wilkinson B (2019) Antibiotics: past, present and future. Curr Opin Microbiol 51(Figure 1):72–80. https://doi.org/10.1016/j.mib.2019.10.008
Karp F, Busatto C, Turino L, Luna J, Estenoz D (2018) PLGA nano- and microparticles for the controlled release of fl orfenicol : Experimental and theoretical study. 47248:1–10. https://doi.org/10.1002/app.47248
Kim EH, Aoki T (1996) Sequence analysis of the florfenicol resistance gene encoded in the transferable R-plasmid of a fish pathogen, Pasteurella piscicida. Microbiol Immunol 40(9):665–669. https://doi.org/10.1111/j.1348-0421.1996.tb01125.x
Li P, Zhu T, Zhou D, Lu W, Liu H (2020) Analysis of resistance to Florfenicol and the related mechanism of dissemination in different animal-derived Bacteria. 10(2):1–11. https://doi.org/10.3389/fcimb.2020.00369
Makadia HK, Siegel SJ (2011) Poly lactic-co-glycolic acid (PLGA) as biodegradable controlled drug delivery carrier. Polymers 3(3):1377–1397. https://doi.org/10.3390/polym3031377
Marshall SA, Jones RN, Wanger A, Washington JA, Doern GV, Leber AL, Haugen TH (1996) Proposed MIC quality control guidelines for National Committee for clinical laboratory standards susceptibility tests using seven veterinary antimicrobial agents: Ceftiofur, Enrofloxacin, Florfenicol, penicillin G-Novobiocin, Pirlimycin, Premafloxacin, and Spectinomycin. J Clin Microbiol 34(8). https://journals.asm.org/journal/jcm
Miethke M, Pieroni M, Weber T, Brönstrup M, Hammann P, Halby L, Arimondo PB, Glaser P, Aigle B, Bode HB, Moreira R, Li Y, Luzhetskyy A, Medema MH, Pernodet JL, Stadler M, Tormo JR, Genilloud O, Truman AW, … Müller R (2021) Towards the sustainable discovery and development of new antibiotics. Nat Rev Chem 5(10):726–749. Nature Research. https://doi.org/10.1038/s41570-021-00313-1
Páll E, Niculae M, Brudașcă GF, Ravilov RK, Șandru CD, Cerbu C, Olah D, Zăblău S, Potârniche AV, Spinu M, Duca G, Rusu M, Rzewuska M, Vasiu A (2021) Assessment and antibiotic resistance profiling in vibrio species isolated from wild birds captured in Danube delta biosphere reserve, Romania. Antibiotics 10(3). https://doi.org/10.3390/antibiotics10030333
Papich MG (2016) Florfenicol. Saunders Handbook of Veterinary Drugs, 327–329. https://doi.org/10.1016/b978-0-323-24485-5.00264-3
Park BK, Lim JH, Kim MS, Hwang YH, Yun HI (2008a) Pharmacokinetics of florfenicol and its metabolite, florfenicol amine, in dogs. Res Vet Sci 84(1):85–89. https://doi.org/10.1016/j.rvsc.2007.04.001
Park BK, Lim JH, Kim MS, Hwang YH, Yun HI (2008b) Pharmacokinetics of florfenicol and its metabolite, florfenicol amine, in dogs. Res Vet Sci 84(1):85–89. https://doi.org/10.1016/j.rvsc.2007.04.001
Patra JK, Das G, Fraceto LF, Campos EVR, Rodriguez-Torres MDP, Acosta-Torres LS, Diaz-Torres LA, Grillo R, Swamy MK, Sharma S, Habtemariam S, Shin HS (2018) Nano based drug delivery systems: recent developments and future prospects. J Nanobiotechnol 16(1):1–33. https://doi.org/10.1186/s12951-018-0392-8
Paudel S, Cerbu C, Astete CE, Louie SM, Sabliov C, Rodrigues DF (2019) Enrofloxacin-impregnated PLGA Nanocarriers for efficient therapeutics and diminished generation of reactive oxygen species [research-article]. ACS Appl Nano Mater 2(8):5035–5043. https://doi.org/10.1021/acsanm.9b00970
Pinto R, Torres-Vergara P, Luengo J, Gómez-Gaete C, Troncoso J, Schneider M, Von Plessing C (2014) Development and characterization of PLGA nanoparticles loaded with florfenicol. Lat Am J Pharm 33:1139
Prestinaci F, Pezzotti P, Pantosti A (2015) Antimicrobial resistance: a global multifaceted phenomenon. In Pathogens and global health, vol 109, issue 7. Maney Publishing, pp. 309–318. https://doi.org/10.1179/2047773215Y.0000000030
Raval N, Maheshwari R, Kalyane D, Youngren-Ortiz SR, Chougule MB, Tekade RK (2018) Importance of physicochemical characterization of nanoparticles in pharmaceutical product development. In Basic fundamentals of drug delivery. Elsevier Inc. https://doi.org/10.1016/B978-0-12-817909-3.00010-8
Shankar P (2016) Book review: tackling drug-resistant infections globally. Arch Pharm Pract 7(3):110. https://doi.org/10.4103/2045-080x.186181
Shuang G, Yu S, Weixiao G, Dacheng W, Zhichao Z, **g L, Xuming D (2011) Immunosuppressive activity of florfenicol on the immune responses in mice. 356–366. https://doi.org/10.3109/08820139.2010.551434
Sousa De Almeida M, Susnik E, Drasler B, Taladriz-Blanco P, Petri-Fink A, Rothen-Rutishauser B (2021) Understanding nanoparticle endocytosis to improve targeting strategies in nanomedicine. In Chemical society reviews, vol 50, issue 9. Royal Society of Chemistry, pp 5397–5434. https://doi.org/10.1039/d0cs01127d
Varma KJ, Adams PE, Powers TE, Powers JD, Lamendolat JF, Lamendola JD, Pharmacokinetics JF (1986) Pharmacokinetics of florfenicol in veal calves. J Vet Pharmacol Ther 9
Vinken M, Blaauboer BJ (2017) In vitro testing of basal cytotoxicity: establishment of an adverse outcome pathway from chemical insult to cell death. Toxicol in Vitro 39:104–110. https://doi.org/10.1016/j.tiv.2016.12.004
Wei CF, Shien JH, Chang SK, Chou CC (2016) Florfenicol as a modulator enhancing antimicrobial activity: example using combination with thiamphenicol against Pasteurella multocida. Front Microbiol 7(MAR). https://doi.org/10.3389/fmicb.2016.00389
Xu R (2008) Progress in nanoparticles characterization: sizing and zeta potential measurement. Particuology 6(2):112–115. https://doi.org/10.1016/j.partic.2007.12.002
Yuan X, Liu J, Li R, Zhou J, Wei J, Jiao S, Wang ZA, Du Y (2020) Chitosan oligosaccharides coupling inhibits bacterial biofilm-related antibiotic resistance against Florfenicol. Molecules 25(24). https://doi.org/10.3390/MOLECULES25246043
Acknowledgments
This work was supported by a grant of the Ministry of Research, Innovation and Digitization, CNCS -UEFISCDI, project number PN-III-P1-1.1-PD-2021-0033, within PNCD III, as well as by the USDA National Institute of Food and Agriculture, AFRI projects # 2017-07878 and # 2018-07406, and the USDA-NIFA Hatch Project #1008750.
Funding
This work was supported by a grant of the Ministry of Research, Innovation and Digitization, CNCS -UEFISCDI, project number PN-III-P1-1.1-PD-2021-0033, within PNCD III, as well as USDA National Institute of Food and Agriculture, AFRI projects # 2017-07878 and # 2018-07406, and the USDA-NIFA Hatch Project #1008750.
Author information
Authors and Affiliations
Contributions
Conceptualization: Constantin Cerbu and Cristina M. Sabliov; Methodology: Emilia Trif, Constantin Cerbu, Carlos E. Astete, Sumit Libi and Emoke Pall; Formal analysis and investigation: Emilia Trif, Constantin Cerbu, Carlos E. Astete, Sumit Libi, Emoke Pall, Septimiu Tripon, Diana Olah, Adrian Valentin Potarniche, Writing - original draft preparation: Emilia Trif; Writing - review and editing: Constantin Cerbu, Cristina M. Sabliov, Carlos E. Astete, Sumit Libi, Lucjan Witkowski and Marina Spinu; Funding acquisition: Constantin Cerbu and Cristina M. Sabliov; Resources: Constantin Cerbu, Cristina M Sabliov, and Marina Spinu; Supervision: Gheorghe Florinel Brudasca and Cristina M. Sabliov.
Corresponding author
Ethics declarations
Competing interests
All authors declare they have no financial interests. All authors read and approved the final manuscript.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Trif, E., Cerbu, C., Astete, C.E. et al. Delivery of florfenicol in veterinary medicine through a PLGA-based nanodelivery system: improving its performance and overcoming some of its limitations. Vet Res Commun 48, 259–269 (2024). https://doi.org/10.1007/s11259-023-10205-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11259-023-10205-y