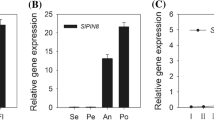
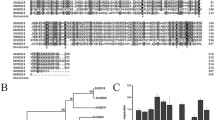
Abstract
Mitogen-activated protein kinases (MAPKs) play important roles in various aspects including plant development and stress response. The biological role of MAPKs in tomato development remains unclear. Here, multiple stable transgenetic lines were developed using either gene overexpression or RNA interference (RNAi) technology to investigate the role of SlMAPK7 in tomato reproductive development. Either upregulation or downregulation of SlMAPK7 did not drastically disturb the vegetative growth of tomato. However, in RNAi-based gene silencing plants, approximately 90 % of the pollen grains were identified to be abnormal and lack viability, which were malformed or collapsed. Cytological observations showed that anthers could develop normal microspore mother cells, but the uninucleate microspores failed to form normal mature pollen grains. Transmission electron microscopy examination demonstrated that accelerated degradation of tapetum occurred in the anther and finally the intine layer of pollen appears abnormal. In addition, although inhibition of SlMAPK7 gene did not alter fruit morphology, seed number in RNAi fruits was markedly reduced. Thus, the results demonstrated that SlMAPK7 is involved in tomato pollen development, possibly via regulation the degradation of tapetum.











Similar content being viewed by others
Abbreviations
- CTAB:
-
Cetyl trimethyl ammonium bromide
- DAPI:
-
4′,6-Diamidino-2-phenylindole
- GFP:
-
Green fluorescent protein
- GUS:
-
β-Glucuronidase
- HYG:
-
Hygromycin resistant gene
- MAPK:
-
Mitogen-activated protein kinase
- MAPKK:
-
MAPK kinase
- MAPKKK:
-
MAPKK kinase
- MMC:
-
Microspore mother cell
- QRT-PCR:
-
Quantitative reverse transcription polymerase chain reaction
- SEM:
-
Scanning electron microscopy
- RNAi:
-
RNA interference
- TEM:
-
Transmission electron microscopy examination
References
Agrawal GK, Iwahashi H, Rakwal R (2003) Rice MAPKs. Biochem Bioph Res Com 302:171–180
Alexander MP (1969) Differential staining of aborted and nonaborted pollen. Stain Technol 144:117–122
Beck M, Komis G, Müller J, Menzel D, Samaj J (2010) Arabidopsis homologs of nucleus- and phragmoplast-localized kinase 2 and 3 and mitogen-activated protein kinase 4 are essential for microtubule organization. Plant Cell 22:755–771
Beck M, Komis G, Ziemann A, Menzel D, Šamaj J (2011) Mitogen-activated protein kinase 4 is involved in the regulation ofmitotic and cytokinetic microtubule transitions in Arabidopsis thaliana. New Phytol 189:1069–1083
Cao J, Yu X, Ye W, Lu G, **ang X (2006) Functional analysis of a novel male fertility CYP86MF gene in Chinese cabbage (Brassica campestris L. ssp. chinensis Makino). Plant Cell Rep 24:715–723
Cargnello M, Roux P (2011) Activation and function of the MAPKs and their substrates, the MAPK-activated protein kinases. Microbiol Mol Biol Rev 75:50–83
Coronado MJ, Gonzalez-Melendi P, Seguı JM, Ramırez C, Barany I, Testillano PS, Risueno MC (2002) MAPKs entry into the nucleus at specific interchromatin domains in plant differentiation and proliferation processes. J Struct Biol 140(1):200–213
Coronado MJ, Testillano PS, Wilson C, Vicente O, Heberle-Bors E, Risueño MC (2007) In situ molecular identification of the Ntf4 MAPK expression sites in maturing and germinating pollen. Biol Cell 99(4):209–221
Gerszberg A, Hnatuszko-Konka K, Kowalczyk T, Kononowicz AK (2015) Tomato (Solanum lycopersicum L.) in the service of biotechnology. Plant Cell Tiss Org Cult 120:881–902
Guan YF, Meng XZ, Khanna R, LaMontagne E, Liu Y, Zhang SQ (2014) Phosphorylation of a WRKY transcription factor by MAPKs is required for pollen development and function in Arabidopsis. PLoS Genet 10(5):e1004384
Hamel LP, Nicole MC, Sritubtim S, Morency MJ, Ellis M, Ehlting J, Beaudoin N, Barbazuk B, Klessig D, Lee J et al (2006) Ancient signals: comparative genomics of plant MAPK and MAPKK gene families. Trends Plant Sci 11:192–198
Heberle-Bors E, Voronin V, Touraev A, Sanchez Testillano P, Risueño MC, Wilson C (2001) MAP kinase signaling during pollen development. Sex Plant Rep. 14(1):15–19
Hord CLH, Sun YJ, Pillitteri LJ, Torii KU, Wang H, Zhang S, Ma H (2008) Regulation of Arabidopsis early anther development by the mitogen-activated protein kinases, MPK3 and MPK6, and the ERECTA and related receptor-like kinases. Mol Plant 1:645–658
Huang L, Cao J, Ye W, Liu T, Jiang L, Ye Y (2008) Transcriptional differences between the male-sterile mutant bcms and wild-type Brassica campestris ssp. chinensis reveal genes related to pollen development. Plant Biol 10:342–355
Johnson GL, Lapadat R (2002) Mitogen-activated protein kinase pathways mediated by ERK, JNK, and p38 protein kinases. Science 298:1911–1912
Kawanabe T, Ariizumi T, Kawai-Yamada M, Uchimiya H, Toriyama K (2006) Abolition of the tapetum suicide program ruins microsporogenesis. Plant Cell Physiol 47:784–787
Kong F, Wang J, Cheng L, Liu S, Wu J, Peng Z, Lu G (2012) Genome-wide analysis of the mitogen-activated protein kinase gene family in Solanum lycopersicum. Gene 499:108–120
Kosetsu K, Matsunaga S, Nakagami H, Colcombet J, Sasabe M, Soyano T, Takahashi Y, Hirt H, Machida Y (2010) The MAP kinase MPK4 is required for cytokinesis in Arabidopsis thaliana. Plant Cell 22:3778–3790
Krens SF, Spaink HP, Snaar-Jagalska BE (2006) Functions of the MAPK family in vertebrate-development. FEBS Lett 580:4984–4990
Lee SK, Kim BG, Kwon TR, Jeong MJ, Park SR, Lee JW, Byun MO, Kwon HB, Matthews BF, Hong CB et al (2011) Overexpression of the mitogen-activated protein kinase gene OsMAPK33 enhances sensitivity to salt stress in rice (Oryza sativa L.). J Biosci 36:139–151
Li S, Franklin-Tong VE (2011) Modulating and monitoring MAPK activity during programmed cell death in pollen. Methods Mol Biol 779:165–183
Li J, Yu M, Geng LL, Zhao J (2010) The fasciclin-like arabinogalactan protein gene, FLA3, is involved in microspore development of Arabidopsis. Plant J 64:482–497
Lin SE, Dong H, Zhang F, Qiu L, Wang FZ, Cao JS, Huang L (2013) BcMF8, a putative arabinogalactan protein-encoding gene, contributes to pollen wall development, aperture formation and pollen tube growth in Brassica campestris. Ann Bot-Landon, pp 1–12
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−∆∆Ct method. Methods 25:402–408
Lou Y, Xu XF, Zhu J, Gu JN, Blackmore S, Yang ZN (2014) The tapetal AHL family protein TEK determines nexine formation in the pollen wall. Nat Commun 5
Ma H (2005) Molecular genetic analyses of microsporogenesis and microgametogenesis in flowering plants. Annu Rev Plant Biol 56:393–434
MAPK Group (2002) Mitogen-activated protein kinase cascades in plants: a new nomenclature. Trends Plant Sci 7:301–308
Mascarenhas JP (1989) The male gametophyte of flowering plants. Plant Cell 1:57–664
McCormick S (1993) Male gametophyte development. Plant Cell 5(10):1265–1275
McCormick S (2004) Control of male gametophyte development. Plant Cell 16(suppl):S142–S153
McDonald BA, Martinez JP (1990) Restriction fragment length polymorphisms in Septoria tritici occur at a high frequency. Curr Genet 17:133–138
Nicole MC, Hamel LP, Morency MJ, Beaudoin N, Ellis BE, Séguin A (2006) MAP-** genomic organization and organ-specific expression profiles of poplar MAP kinases and MAP kinase kinases. BMC Genom 7:223
Ovecka M, Lichtscheidl IK (2009) MAPK signaling. Signaling in Plants, pp 51–68
Peng Z, Cheng L, He YJ, Wang J, Guan XY, Liu SY, Lu G (2013) Cytological study on microsporogenesis of Solanum lycopersicum var. Micro-Tom under high temperature stress. Acta Ecologica Sinica 33:2084–2092
Petersen M, Brodersen P, Naested H, Andreasson E, Lindhart U, Johansen B, Nielsen HB, Lacy M, Austin MJ, Parker JE et al (2000) Arabidopsis MAP Kinase 4 negatively regulates systemic acquired resistance. Cell 103:1111–1120
Pitzschke A, Schikora A, Hirt H (2009) MAPK cascade signaling networks in plant defence. Curr Opin Plant Biol 12:421–426
Regan SM, Moffatt BA (1990) Cytochemical analysis of pollen development in wild-type Arabidopsis and a male sterile mutant. Plant Cell 2:877–889
Rogers HJ, Bate N, Combe J, Sullivan J, Sweetman J, Swan C, Lonsdale DM, Twell D (2001) Functional analysis of cis-regulatory elements within the promoter of the tobacco late pollen gene g10. Plant Mol Biol 45:577–585
Rohila JS, Yang Y (2007) Rice mitogen-activated protein kinase gene family and its role in biotic and abiotic stress response. J Integr Plant Biol 49:751–759
Romeis T (2001) Protein kinases in the plant defence response. Curr Opin Plant Biol 4:407–414
Sanders PM, Bui AQ, Weterings K, McIntire K, Hsu YC, Lee PY, Truong MT, Beals T, Goldberg R (1999) Anther developmental defects in Arabidopsis thaliana male-sterile mutants. Sex Plant Reprod 11:297–322
Sawhney VK, Bhadula SK (1988) Microsporogenesis in the normal and male sterile stamenless-2 mutant of tomato (Lycopersicon esculentum). Can J Bot 66:2013–2021
Sorensen A, Krober S, Unte US, Huijser P, Dekker K, Saedler H (2003) The Arabidopsis ABORTED MICROSPORES (AMS) gene encodes a MYC class transcription factor. Plant J 33:413–423
Sun HJ, Uchii SU, Watanabe S, Ezura H (2006) A highly efficient transformation protocol for Micro-Tom, a model cultivar for tomato functional genomics. Plant Cell Physiol 47:426–431
Tena G, Asai T, Chiu WL, Sheen J (2001) Plant mitogen-activated protein kinase signaling cascades. Curr Opin Plant Biol 4:392–400
Wan LL, **a XY, Hong DF, Li J, Yang GS (2010) Abnormal vacuolization of the tapetum during the tetrad stage is associated with male sterility in the recessive genicmale sterile Brassica napus L. Line 9012A. Plant Biol 53:121–133
Wang Y, Zhang WZ, Song LF, Zou JJ, Su Z, Wu WH (2008) Transcriptome analyses show changes in gene expression to accompany pollen germination and tube growth in Arabidopsis. Plant Physiol 148:1201–1211
Wilson ZA, Zhang DB (2009) From Arabidopsis to rice: pathways in pollen development. J Exp Bot 60:1479–1492
Wilson C, Voronin V, Touraev A, Vicente O, Heberle-Bors E (1997) A developmentally regulated MAP kinase activated by hydration in tobacco pollen. Plant Cell 9:2093–2100
Wilson ZA, Morroll SM, Dawson J, Swarup R, Tighe PJ (2001) The Arabidopsis MALE STERILITY 1(MS1) gene is a transcriptional regulator of male gametogenesis, with homology to the PHD-finger family of transcription factors. Plant J 28:27–39
Wu J, Baldwin IT (2010) New insights into plant responses to the attack from insect herbivores. Ann Rev Genet 44:1–2
Wu HM, Cheun AY (2000) Programmed cell death in plant reproduction. Plant Mol Biol 44(3):267–281
Zeng Q, Chen JG, Ellis BE (2011) AtMPK4 is required for male-specific meiotic cytokinesis in Arabidopsis. Plant J 67:895–906
Zhang D, Liu D, Lv X, Wang Y, Xun Z, Liu Z, Li F, Lu H (2014) The cysteine protease CEP1, a key executor involved in tapetal programmed cell death, regulates pollen development in arabidopsis. Plant Cell 26:2939–2961
Zhu J, Chen H, Li H, Gao J, Jiang H, Wang C, Guan Y, Yang Z (2008) Defective in tapetal development and function 1is essential for anther development and tapetal function for microspore maturation in Arabidopsis. Plant J 55:266–277
Acknowledgments
This work was supported by the Grants from the Nation Natural Science Foundation of China [Grant Numbers 31071337, 31271633], the Natural Science Foundation of Zhejiang Province (R3110209) and Zhejiang Province key science and technology innovation team (2013TD05).
Author contributions
G.L. and L.C. conceived and designed research. X.G., L.C., L.Q., Y.W., and T.Z. conducted experiments. L.C., Y.Z. and C.P. analyzed data. L.C., G.L. wrote the manuscript. All authors read and approved the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Lifei Chen and **aoyan Guan have contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Chen, L., Guan, X., Qin, L. et al. Downregulation of the mitogen-activated protein kinase SlMAPK7 gene results in pollen abortion in tomato. Plant Cell Tiss Organ Cult 126, 79–92 (2016). https://doi.org/10.1007/s11240-016-0979-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-016-0979-4