Abstract
In this study, silica-gel (SG)-supported deep eutectic solvent (DES) with a different loading dosage as the novel nanocomposite materials has been synthesized by sol–gel method. And the supported nanocomposite materials were used as the catalyst for the synthesis of 1,10-phenanthroline from the 8-aminoquinoline and acrolein. The properties and structure of catalysts were characterized by Fourier transform infrared, N2 adsorption–desorption, scanning electron microscopy, thermogravimetric analysis and elemental analysis techniques. It was found that the 12%DES/SG catalyst with the best catalytic activity possessed the suitable specific surface area 132 m2/g and 12% loading amount of DES, which combines the excellent properties of mesoporous materials and DES. The result of thermogravimetric analysis was exhibited that the nanocomposite materials could not be decomposed below 240 °C, which can be used as a stable catalytic material under reaction conditions. Furthermore, the effect of reaction parameters such as reaction temperature and catalyst amount, molar ratio of 8-aminoquinoline and acrolein and reaction time was also studied. Under the optimal process parameters, within the optimum parameters: 130 °C, acrolein: 8-AQ = 2:1, catalyst amount 20 wt%, and time 4 h, the conversion of 8-aminoquinoline and selectivity of 1,10-phenanthroline were 83.8% and 84.3%, respectively. Otherwise, the catalyst can be separated from the reaction system and reused. Therefore, the silica-gel-supported deep eutectic solvent nanocomposite materials can be considered as a potential catalytic material for the synthesis of 1,10-phenanthroline and other nitrogen-containing heterocyclic organic chemical intermediates.
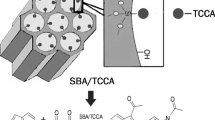
Graphical abstract
Replacing traditional sulfuric acid and hydrochloric acid with silica-gel-supported deep eutectic solvents as new nanocomposite catalysts provides a more efficient, greener and more economical strategy for the synthesis of 1,10-phenanthroline.










Similar content being viewed by others
References
A. Bencini, V. Lippolis, Chem. Rev. 254, 17–18 (2010)
L. Shu-Kun, Molecules. 4, 366 (1999)
H.A. Azab, Z.M. Anwar, M.A. Rizk, G.M. Khairy and M.H. El-Asfoury, J. Lumin. 157, (2015)
M.P. Algi, J. Fluor. Chem. 26, 1083 (2016)
Y.-X. Hu, G.-W. Zhao, Y. Dong, Y.-L. Lü, X. Li, D.-Y. Zhang, Dyes Pigments 137, 569 (2017)
R.S. Kumar, S. Arunachalam, Eur. J. Med. Chem. 44, 5 (2009)
H. Saggadi, D. Luart, N. Thiebault, I. Polaert, L. Estel, C. Len, RSC. Adv. 4, 41 (2014)
J. Marco-Contelles, E. Pérez-Mayoral, A. Samadi, M.D.C. Carreiras, E. Soriano, Chem. Rev. 109, 6 (2009)
C. Lüdtke, A. Haupt, M. Wozniak, N. Kulak, J. Fluor. Chem. 193, 98 (2017)
K. De, J. Legros, B. Crousse, S. Chandrasekaran, D. Bonnet-Delpon, Org. Biomol. Chem. 9, 2 (2011)
A. Takahashi, Y. Hirose, H. Kusama, N. Iwasawa, Chem. Commun. 5, 609 (2008)
E.L. Smith, A.P. Abbott, K.S. Ryder, Chem. Rev. 114, 21 (2014)
S. Korner, J. Albert, C. Held, Front Chem. 7, 661 (2019)
P. Kalhor, K. Ghandi, Molecules 24, 22 (2019)
T.J. Trivedi, J.H. Lee, H.J. Lee, Y.K. Jeong, J.W. Choi, Green. Chem. 18, 9 (2016)
M.H. Chakrabarti, F.S. Mjalli, I.M. AlNashef, M.A. Hashim, M.A. Hussain, L. Bahadori, C.T.J. Low, Renew. Sust. Energ. Rev. 30, 254 (2014)
M. Wu, Y. Bai, X. Chen, Q. Wang, G. Wang, Res. Chem. Intermed. 47, 3551 (2021)
F. Shi, Q. Zhang, D. Li, Y. Deng, Chem 11, 18 (2005)
P. Makos, E. Slupek, A. Malachowska, Materials 13, 8 (2020)
C. Batarseh, A. Levi-Zada, R. Abu-Reziq, J. Mater. Chem. A 7, 5 (2019)
X.-Y. Liu, X.-P. Li, R.-X. Zhao, H. Zhang, New. J. Chem. 45, 15901 (2021)
M.R. Awual, J. Mol. Liq. 284, 502 (2019)
M.R. Awual, M.M. Hasan, J. Mol. Liq. 294, 111679 (2019)
M.R. Awual, M.M. Hasan, H. Znad, Chem. Eng. J. 259, 611 (2015)
M.A. Islam, M.J. Angove, D.W. Morton, B.K. Pramanik, M.R. Awual, J. Environ. Chem. Eng. 8, 2 (2020)
M.R. Awual, Chem. Eng. J. 266, 368 (2015)
M.R. Awual, Chem. Eng. J. 307, 85 (2017)
M. Abdollahi-Alibeik, M. Pouriayevali, Catal. Commun. 22, 13 (2012)
S. Shylesh, P.P. Samuel, C. Srilakshmi, R. Parischa, A.P. Singh, J. Mol. Catal. A Chem. 274, 1 (2007)
G. Li, T. Zhu, Y. Lei, Korean. J Chem. Eng. 32, 10 (2015)
D. Chen, L. Cao, F. Huang, P. Imperia, Y.-B. Cheng, R.A. Caruso, J. Am. Chem. Soc. 132, 12 (2010)
K.T. Kubra, M.S. Salman, M.N. Hasan, A. Islam, S.H. Teo, M.M. Hasan, M.C. Sheikh, M.R. Awual, J. Mol. Liq. 338, 116667 (2021)
M.R. Awual, Compos. Part B Eng. 172, 387 (2019)
M. Safa, B. Mokhtarani, H.R. Mortaheb, K. Tabar Heidar, A. Sharifi, M. Mirzaei, Energy Fuels 31(9), 10196 (2017)
X.R. Zeng, L.L. Wang, J.L. Wang, Can. J. Chem. Eng. 99, 6 (2020)
M.M. Rahman, T.A. Sheikh, A.M. Asiri, M.R. Awual, New J. Chem. 43, 11 (2019)
A.S. de Oliveira, S.J.S. Vasconcelos, J.R. de Sousa, F.F. de Sousa, J.M. Filho, A.C. Oliveira, Chem. Eng. J. 168, 2 (2011)
L. Abahmane, A. Knauer, J.M. Köhler, G.A. Groß, Chem. Eng. J. 167, 2–3 (2011)
G. Chelucci, D. Addis, S. Baldino, Tetrahedron Lett. 48, 19 (2007)
Y. Lu, Y. Jahng, Chin. J. Chem. 37, 3 (2019)
X.-T. Sun, J. Zhu, Y.-T. **a, L. Wu, ChemCatChem 9, 13 (2017)
A. Li, Z. Yang, T. Yang, C.-W. Luo, Z.-S. Chao, C.-S. Zhou, Catal. Commun. 115, 21 (2018)
K. Abbas, H. Znad, M.R. Awual, Chem. Eng. J. 334, 432 (2018)
Acknowledgements
We are thankful for the financial support provided by Sichuan Province Regional Innovation Cooperation (2020YFQ0003), Key R & D project of Sichuan Province (2020YFG0159) and Western China Young Scholars Category A (2019): Preparation of the second-generation dimethyl carbonate catalyst and its application technology development in large-scale industrial plants. The authors also would like to thank Mengdie Li and Bin He from Shiyanjia Lab (www.shiyanjia.com) for the SEM, BET and TGA tests. The author also wishes to thank the anonymous reviewers and editor for their helpful suggestions and enlightening comments.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wu, M., Bai, Y., Wang, Q. et al. Silica-gel-supported deep eutectic solvent (DES) as an efficient novel catalytic system for synthesis of 1,10-phenanthroline. Res Chem Intermed 48, 2575–2591 (2022). https://doi.org/10.1007/s11164-022-04726-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-022-04726-5