Abstract
Aims
The objective of this study was to investigate the response of the iron (Fe) (hydr-) oxides-bounded organic carbon (Fe-OC) process, characterized by the interaction of Fe (hydr-) oxides with soil organic carbon (SOC) in coastal wetlands, to different flooding and soil salinity conditions.
Methods
The study concentrated on Scirpus mariqueter in the Yangtze River Estuary, analyzing variations in various forms of Fe (hydr-) oxides, Fe-OC, and Fe redox cycling bacteria in the rhizosphere and bulk soils under diverse flooding and soil salinity conditions.
Results
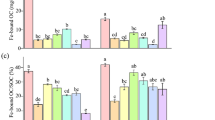
The study found that during the growing season, the levels of crystalline (FeDH) and amorphous (FeHH) Fe (hydr-) oxides were elevated compared to the non-growing season. In the rhizosphere, around 80% of SOC was associated with Fe (hydr-) oxides, significantly higher than the 50% in bulk soil. Elevated areas in the tidal flat exhibited increased FeDH, FeHH, and total Fe content in rhizosphere soils. Soils with higher salinity had more Fe (hydr-) oxides compared to low salinity soils. Furthermore, salinity reduced the prevalence of Gallionella and Geobacter, which are conducive to the preservation of FeHH, leading to a greater FeHH-OC ratio in the total SOC.
Conclusions
The bond between Fe (hydr-) oxides and SOC in soils becomes stronger during the growth season of S. mariqueter. The Fe-OC process’s reaction to environmental factors like salinity and flooding profoundly affects the dynamics of SOC in wetland ecosystems. These findings are essential for wetland management, emphasizing the need to preserve these ecosystems for their role in SOC sequestration.







Similar content being viewed by others
References
Adhikari D (2015) Selective stabilization of aliphatic organic carbon by iron oxide. Sci Rep 5(1):1–7. https://doi.org/10.1038/srep11214
Bernard L, Basile-Doelsch I, Derrien D, Fanin N, Fontaine S, Guenet B, Karimi B, Marsden C, Maron A (2022) Advancing the mechanistic understanding of the priming effect on soil organic matter mineralisation. Funct Ecol 36(6):1355–1377. https://doi.org/10.1111/1365-2435.14038
Bhattacharyya A, Campbell A, Tfaily M, Lin Y, Kukkadapu R, Silver W, Nico P, Pett-Ridge J (2018) Redox fluctuations control the coupled cycling of iron and carbon in tropical forest soils. Environ Sci Technol 52(24):14129–14139. https://doi.org/10.1021/acs.est.8b03408
Chapman K, Hayes A, Kelly B, Langley A (2019) Exploring the oxygen sensitivity of wetland soil carbon mineralization. Biol Lett 15:20180407. https://doi.org/10.1098/rsbl.2018.0407
Chen C, Hall J, Coward E, Thompson A (2020) Iron-mediated organic matter decomposition in humid soils can counteract protection. Nat Commun 11:2255. https://doi.org/10.1038/s41467-020-16071-5
Chen C, Kukkadapu R, Sparks L (2015) Influence of coprecipitated organic matter on Fe2+(aq)-catalyzed transformation of ferrihydrite: implications for carbon dynamics. Environ Sci Technol 49:10927–10936. https://doi.org/10.1021/acs.est.5b02448
Chen C, Meile C, Wilmoth J, Barcellos D, Thompson A (2018) Influence of pO2 on iron redox cycling and anaerobic organic carbon mineralization in a humid tropical forest soil. Environ Sci Technol 52:7709–7719. https://doi.org/10.1021/acs.est.8b01368
Coward K, Thompson T, Plante F (2017) Iron-mediated mineralogical control of organic matter accumulation in tropical soils. Geoderma 306:206–216. https://doi.org/10.1016/j.geoderma.2017.07.026
Dang M, Macdonald B, Warneke S, White I (2017) Available carbon and nitrate increase greenhouse gas emissions from soils affected by salinity. Soil Res 55(1):47–57. https://doi.org/10.1071/sr16010
Dolinar N, Regvar M, Abram D, Gaberščik A (2016) Water-level fluctuations as a driver of Phragmites australis primary productivity, litter decomposition, and fungal root colonisation in an intermittent wetland. Hydrobiologia 774(1):69–80. https://doi.org/10.1007/s10750-015-2492-x
Doran G, Eberbach P, Helliwell S (2006) The impact of rice plant roots on the reducing conditions in flooded rice soils. Chemosphere 63:1892–1902. https://doi.org/10.1016/j.chemosphere.2005.10.027
Finzi C, Abramoff Z, Spiller S, Brzostek R, Darby A, Kramer A, Phillips P (2015) Rhizosphere processes are quantitatively important components of terrestrial carbon and nutrient cycles. Glob Change Biol 21:2082–2094. https://doi.org/10.1111/gcb.12816
Futamata H, Sakai M, Ozawa H, Urashima Y, Sueguchi T, Matsuguchi T (1998) Chemotactic response to amino acids of fluorescent pseudomonads isolated from spinach roots grown in soils with different salinity levels. Soil Sci Plant Nutr 44:1–7. https://doi.org/10.1080/00380768.1998.10414421
Henry A, Doucette W, Norton J, Bugbee B (2007) Changes in crested wheatgrass root exudation caused by flood, drought, and nutrient tress. J Environ Qual 36:904–912. https://doi.org/10.2134/jeq2006.0425sc
Herbert R, Boon P, Burgin J, Neubauer C, Franklin B, Ardón M, Hopfensperger N, Lamers M, Gell P (2015) A global perspective on wetland salinization: ecological consequences of a growing threat to freshwater wetlands. Ecosphere 6(10):art206. https://doi.org/10.1890/ES14-00534.1
Hopkinson S, Cai J, Hu X (2012) Carbon sequestration in wetland dominated coastal systems—a global sink of rapidly diminishing magnitude. Curr Opin Environ Sustain 4:186–194. https://doi.org/10.1016/j.cosust.2012.03.005
Huang H (2021) linkET: Everything is Linkable. R package version 0.0.7
Huang X, Liu X, Liu J, Chen H (2021) Iron-bound organic carbon and their determinants in peatlands of China. Geoderma 391:114974. https://doi.org/10.1016/j.geoderma.2021.114974
Hyun H, Smith C, Kostka E (2007) Relative contributions of sulfate- and iron(III) reduction to organic matter mineralization and process controls in contrasting habitats of the Georgia saltmarsh. Appl Geochem 22:2637–2651. https://doi.org/10.1016/j.apgeochem.2007.06.005
Jiang J, Huang X, Li Z, Yan Z, Li Z, Ding H (2015) Soil organic carbon storage in tidal wetland and its relationships with soil physico-chemical factors: a case study of Dongtan of Chongming, Shanghai. J Ecol Rural Environ (in Chinese) 31:540–547
Jiang C, Li Z, Xue M, Yan Z, Liang X, Chen C (2022) Pioneer salt marsh species Scirpus mariqueter disperses quicker in summer with seed contribution from current and last year. Estuar Coast Shelf Sci 264:107682. https://doi.org/10.1016/j.ecss.2021.107682
Jiang X, Tian Y, Ji X, Lu C, Zhang J (2020) Influences of plant species and radial oxygen loss on nitrous oxide fluxes in constructed wetlands. Ecol Eng 142:105644. https://doi.org/10.1016/j.ecoleng.2019.105644
Kaiser K, Guggenberge G (2000) The role of DOM sorption to mineral surfaces in the preservation of organic matter in soils. Org Geochem 31:711–725. https://doi.org/10.1016/S0146-6380(00)00046-2
Keiluweit M, Bougoure J, Nico S, Pett-Ridge J, Weber K, Kleber M (2015) Mineral protection of soil carbon counteracted by root exudates. Nat Clim Change 5:588–595. https://doi.org/10.1038/nclimate2580
Khan N, Seshadri B, Bolan N, Saint P, Kirkham B, Chowdhury S, Yamaguchi N, Lee Y, Li G, Kunhikrishnan A, Qiu R, Zhu G, Syu H (2016) Root iron plaque on wetland plants as a dynamic pool of nutrients and contaminants. Adv Agron 138:1–96. https://doi.org/10.1016/bs.agron.2016.04.002
Kostka E, Luther W (1994) Partitioning and speciation of solid phase iron in saltmarsh sediments. Geochim Cosmochim Ac 58:1701–1710. https://doi.org/10.1016/0016-7037(94)90531-2
Lalonde K, Mucci A, Ouellet A, Gelinas Y (2012) Preservation of organic matter in sediments promoted by iron. Nature 483:198–200. https://doi.org/10.1038/nature10855
Li S, Chen S, Wang M, Lei X, Zheng H, Sun X, Wang L, Han Y (2020) Iron fractions responsible for the variation of cd bioavailability in paddy soil under variable pe + pH conditions. Chemosphere 251:126355. https://doi.org/10.1016/j.chemosphere.2020.126355
Lin M, Chen Y, Cheng L, Zheng Y, Wang W, Sardans J, Song L, Guggenbergerf G, Zou C, Ding L, Tariq A, Zeng J, Alrefaei F, Peñuelas J (2023) Response of Topsoil Fe-bound organic carbon pool and microbial community to Spartina alterniflora invasion in coastal wetlands. CATENA 232:107414. https://doi.org/10.1016/j.catena.2023.107414
Liu X, Lin C, Vadeboncoeur A, Yang Z, Chen S, **ong D, Xu C, Li Y, Yang Y (2019) Root litter inputs exert greater influence over soil C than does aboveground litter in a subtropical natural forest. Plant Soil 444:489–499. https://doi.org/10.1007/s11104-019-04294-5
Liu R, Ma T, Lin C, Chen J, Lei K, Liu X, Qiu W (2020) Transfer and transformation mechanisms of Fe bound-organic carbon in the aquitard of a lake-wetland system during reclamation. Environ Pollut 263:114441. https://doi.org/10.1016/j.envpol.2020.114441
Luo M, Zeng S, Tong C, Huang F, Yu Q, Guo B, Wang H (2014) Abundance and speciation of iron across a subtropical tidal marsh of the Min River Estuary in the East China Sea. Appl Geochem 45:1–13. https://doi.org/10.1016/j.apgeochem.2014.02.014
Lv J, Zhang S, Wang S, Luo L, Cao D, Christie P (2016) Molecular-scale investigation with ESI-FT-ICR-MS on fractionation of dissolved organic matter induced by adsorption on iron oxyhydroxides. Environ Sci Technol 50:2328–2336. https://doi.org/10.1021/acs.est.5b04996
McBeth M, Fleming J, Emerson D (2013) The transition from freshwater to marine iron-oxidizing bacterial lineages along a salinity gradient on the Sheepscot River, Maine, USA. Env Microbiol Rep 5:453–463. https://doi.org/10.1111/1758-2229.12033
Mcleod E, Chmura GL, Bouillon S, Salm R, Björk M, Duarte CM, Lovelock CE, Schlesinger WH, Silliman BR (2011) A blueprint for blue carbon: toward an improved understanding of the role of vegetated coastal habitats in sequestering CO2. Front Ecol Environ 9(10):552–560
Meng H, Yan Z, Li X (2022) Effects of exogenous organic acids and flooding on root exudates, rhizosphere bacterial community structure, and iron plaque formation in Kandelia obovata seedlings. Sci Total Environ 830:154695. https://doi.org/10.1016/j.scitotenv.2022.154695
Neubauer C, Franklin B, Berrier J (2013) Saltwater intrusion into tidal freshwater marshes alters the biogeochemical processing of organic carbon. Biogeosciences 10:8171–8183. https://doi.org/10.5194/bg-10-8171-2013
Neubauer C, Toledo-Durán E, Emerson D, Megonigal J (2007) Returning to their roots: iron-oxidizing bacteria enhance short-term plaque formation in the wetland-plant rhizosphere. Geomicrobiol J 24(1):65–73. https://doi.org/10.1080/01490450601134309
Ponnamperuma FN (1972) The chemistry of submerged soils. Adv Agron 24:29–96
Phillips P, Fahey J (2006) Tree species and mycorrhizal associations influence the magnitude of rhizosphere effects. Ecology 87:1302–1313. https://doi.org/10.1890/0012-9658(2006)87[1302:tsamai]2.0.co;2
Preston M, Nault R, Trofymow A, Smyth C, CIDET Working Group (2009) Chemical changes during 6 years of decomposition of 11 litters in some Canadian forest sites. Part 1. Elemental composition, tannins, phenolics, and proximate fractions. Ecosys- tems 12(7):1053–1077. https://doi.org/10.1007/s10021-009-9267-z
Rashid M (1985) Geochemistry of Marine Humic compounds. Springer New York, NY, pp 1-300
Roden E, Wetzel G (1996) Organic carbon oxidation and suppression of methane production by microbial Fe(III) oxide reduction in vegetated and unvegetated freshwater wetland sediments. Limnol Oceanogr 41:1733–1748. https://doi.org/10.4319/lo.1996.41.8.1733
Schuerch M, Spencer T, Temmerman S, Kirwan ML, Wolff C, Lincke D, McOwen J, Pickering D, Reef R, Vafeidis T, Hinke J, Nicholls J, Brown S (2018) Future response of global coastal wetlands to sea-level rise. Nature 561:231–234. https://doi.org/10.1038/s41586-018-0476-5
Setia R, Gottschalk P, Smith P, Marschner P, Baldock J, Setia D, Smith J (2013) Soil salinity decreases global soil organic carbon stocks. Sci Total Environ 465:267–272. https://doi.org/10.1016/j.scitotenv.2012.08.028
Shyla B, Bhaskar V, Nagendrappa G (2012) Iron(III) oxidized nucleophilic coupling of catechol with o-tolidine/p-toluidine followed by 1,10-phenanthroline as new and sensitivity improved spectrophotometric methods for iron present in chemicals, pharmaceutical, edible green leaves, nuts and lake water samples. Spectrochim Acta A 86:152–158. https://doi.org/10.1016/j.saa.2011.09.068
Thompson A, Chadwick A, Rancourt G, Chorover J (2006) Iron-oxide crystallinity increases during soil redox oscillations. Geochim Cosmochim Ac 70:1710–1727. https://doi.org/10.1016/j.gca.2005.12.005
Tong T, Li R, Wu S, **e S (2019) The distribution of sediment bacterial community in mangroves across China was governed by geographic location and eutrophication. Mar Pollut Bull 140:198–203. https://doi.org/10.1016/j.marpolbul.2019.01.046
Wagai R, Mayer M (2007) Sorptive stabilization of organic matter in soils by hydrous iron oxides. Geochim Cosmochim Ac 71:25–35. https://doi.org/10.1016/j.gca.2006.08.047
Weaver L, Tarney J (1984) Empirical approach to estimating the composition of the continental crust. Nature 310:575–577. https://doi.org/10.1038/310575a0
Weiss V, Emerson D, Megonigal P (2004) Geochemical control of microbial Fe(III) reduction potential in wetlands: comparison of the rhizosphere to non-rhizosphere soil. Fems Microbiol Ecol 48(1):89–100. https://doi.org/10.1016/j.femsec.2003.12.014
Wickham H (2016) ggplot2: elegant graphics for data analysis. Springer-Verlag, New York
Wickham H, François R, Henry L, Müller K (2022) dplyr: A Grammar of Data Manipulation_. R package version 1.0.10. https://CRAN.R-project.org/package=dplyr
**ao S, Luo M, Liu Y, Bai J, Yang Y, Zhai Z, Huang J (2021) Rhizosphere effect and its associated soil-microbe interactions drive iron fraction dynamics in tidal wetland soils. Sci Total Environ 756:144056. https://doi.org/10.1016/j.scitotenv.2020.144056
Xue L, Li X, Yan Z, Zhang Q, Ding W, Huang X, Tian B, Ge Z, Yin Q (2018) Native and non-native halophytes resiliency against sea-level rise and saltwater intrusion. Hydrobiologia 806:47–65. https://doi.org/10.1007/s10750-017-3333-x
Yang L, Shi W, Bouma J, Ysebaert T, Luo X (2012) Wave attenuation at a salt marsh margin: a case study of an exposed coast on the Yangtze Estuary. Estuar Coast 35:169–182. https://doi.org/10.1007/s12237-011-9424-4
Yu G (2018) Root exudates and microbial communities drive mineral dissolution and the formation of nano-size minerals in soils: implications for soil carbon storage. In: Giri B, Prasad RAV (ed) Root biology Cham: Springer, pp 143–166
Yu C, **e S, Song Z, **a S, Åström E (2021) Biogeochemical cycling of iron (hydr-) oxides and its impact on organic carbon turnover in coastal wetlands: a global synthesis and perspective. Earth-Sci Rev 218:103658
Yuan L, Zhang L, **ao D, Huang H (2011) The application of cutting plus waterlogging to control Spartina alterniflora on saltmarshes in the Yangtze Estuary, China. Estuar Coast Shelf S 92:103–110. https://doi.org/10.1016/j.ecss.2010.12.019
Zhao Q, Adhikari D, Huang R, Patel A, Wang X, Tang Y, Obrist D, Roden E, Yang Y (2017) Coupled dynamics of iron and iron-bound organic carbon in forest soils during anaerobic reduction. Chem Geol 464:118–126. https://doi.org/10.1016/j.chemgeo.2016.12.014
Acknowledgements
The work described in this paper was supported by National Key R&D Program of China [grant number 2023YFE0113100], the Key Projects of National Natural Science Foundation of China [grant number 42141016], and National Natural Science Foundation of China [grant number 41877413].
Author information
Authors and Affiliations
Contributions
Yuxin Bi: Investigation, Writing - original draft, Formal analysis. Tianyou Li: Investigation, Formal analysis. Wenzhen Zhao: Investigation, Resources. Liming Xue: Investigation, Resources. Ying Lei: Investigation, Visualization. Qiqiong Zhang: Investigation, Visualization. **aoqing Gao: Investigation, Resources. **uzhen Li: Supervision. Zhongzheng Yan: Conceptualization, Supervision, Writing - review & editing.
Corresponding author
Ethics declarations
This research does not involving any Human Participants and/or Animals. None of the authors have any potential conflicts of interest. All the authors were informed and consent this submission.
Competing of interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Responsible Editor: Luca Bragazza.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Bi, Y., Li, T., Zhao, W. et al. Variations in Iron-bound carbon in Scirpus mariqueter rhizosphere and bulk soils across different salinities and tidal elevations. Plant Soil (2024). https://doi.org/10.1007/s11104-024-06787-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11104-024-06787-4