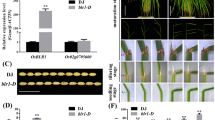
Abstract
Key message
We proposed a working model of BR to promote leaf size through cell expansion. In the BR signaling pathway, GhBES1 affects cotton leaf size by binding to and activating the expression of the E-box element in the GhEXO2 promoter region.
Abstract
Brassinosteroid (BR) is an essential phytohormone that controls plant growth. However, the mechanisms of BR regulation of leaf size remain to be determined. Here, we found that the BR deficient cotton mutant pagoda1 (pag1) had a smaller leaf size than wild-type CRI24. The expression of EXORDIUM (GhEXO2) gene, was significantly downregulated in pag1. Silencing of BRI1-EMS-SUPPRESSOR 1 (GhBES1), inhibited leaf cell expansion and reduced leaf size. Overexpression of GhBES1.4 promoted leaf cell expansion and enlarged leaf size. Expression analysis showed GhEXO2 expression positively correlated with GhBES1 expression. In plants, altered expression of GhEXO2 promoted leaf cell expansion affecting leaf size. Furthermore, GhBES1.4 specifically binds to the E-box elements in the GhEXO2 promoter, inducing its expression. RNA-seq data revealed many down-regulated genes related to cell expansion in GhEXO2 silenced plants. In summary, we discovered a novel mechanism of BR regulation of leaf size through GhBES1 directly activating the expression of GhEXO2.








Similar content being viewed by others
Data availability
All datasets generated for this study are included in the article/Supplementary Files.
References
Ali F, Qanmber G, Li F, Wang Z (2021) Updated role of ABA in seed maturation, dormancy, and germination. J Adv Res 35:199–214
Andres RJ, Bowman DT, Jones DC, Kuraparthy V (2016) Major leaf shapes of cotton: genetics and agronomic effects in crop production. J Cotton Sci 20(4):330–340
Andres RJ, Coneva V, Frank MH, Tuttle JR, Samayoa LF, Han S-W, Kaur B, Zhu L, Fang H, Bowman DT (2017) Modifications to a LATE MERISTEM IDENTITY1 gene are responsible for the major leaf shapes of Upland cotton (Gossypium hirsutum L.). Proc National Acad Sci 114(1):E57–E66
Aya K, Hobo T, Sato-Izawa K, Ueguchi-Tanaka M, Kitano H, Matsuoka M (2014) A novel AP2-type transcription factor, SMALL ORGAN SIZE1, controls organ size downstream of an auxin signaling pathway. Plant Cell Physiol 55(5):897–912
Azpiroz R, Wu Y, LoCascio JC, Feldmann KA (1998) An Arabidopsis brassinosteroid-dependent mutant is blocked in cell elongation. Plant Cell 10(2):219–230
Bai M-Y, Zhang L-Y, Gampala SS, Zhu S-W, Song W-Y, Chong K, Wang Z-Y (2007) Functions of OsBZR1 and 14-3-3 proteins in brassinosteroid signaling in rice. Proc Natl Acad Sci 104(34):13839–13844
Bajwa KS, Shahid AA, Rao AQ, Bashir A, Aftab A, Husnain T (2015) Stable transformation and expression of GhEXPA8 fiber expansin gene to improve fiber length and micronaire value in cotton. Front Plant Sci 6:838
Cheng Y, Lu L, Yang Z, Wu Z, Qin W, Yu D, Ren Z, Li Y, Wang L, Li F, Yang Z (2016) GhCaM7-like, a calcium sensor gene, influences cotton fiber elongation and biomass production. Plant Physiol Biochem 109:128–136
Chen L-G, Gao Z, Zhao Z, Liu X, Li Y, Zhang Y, Liu X, Sun Y, Tang W (2019) BZR1 family transcription factors function redundantly and indispensably in BR signaling but exhibit BRI1-independent function in regulating anther development in Arabidopsis. Mol Plant 12(10):1408–1415
Chen L, Yang H, Fang Y, Guo W, Chen H, Zhang X, Dai W, Chen S, Hao Q, Yuan S (2021) Overexpression of GmMYB14 improves high-density yield and drought tolerance of soybean through regulating plant architecture mediated by the brassinosteroid pathway. Plant Biotechnol J 19(4):702–716
Choe S, Dilkes BP, Fujioka S, Takatsuto S, Sakurai A, Feldmann KA (1998) The DWF4 gene of Arabidopsis encodes a cytochrome P450 that mediates multiple 22α-hydroxylation steps in brassinosteroid biosynthesis. Plant Cell 10(2):231–243
Chory J, Nagpal P, Peto CA (1991) Phenotypic and genetic analysis of det2, a new mutant that affects light-regulated seedling development in Arabidopsis. Plant Cell 3(5):445–459
Chung Y, Choe S (2013) The regulation of brassinosteroid biosynthesis in Arabidopsis. Crit Rev Plant Sci 32(6):396–410
Clough SJ, Bent AFJTPJ (1998) Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J 16(6):735–743
Clouse SD, Langford M, McMorris TC (1996) A brassinosteroid-insensitive mutant in Arabidopsis thaliana exhibits multiple defects in growth and development. Plant Physiol 111(3):671–678
Clouse SD, Sasse JM (1998) Brassinosteroids: essential regulators of plant growth and development. Ann Rev Plant Biol 49(1):427–451
Coll-Garcia D, Mazuch J, Altmann T, Müssig C (2004) EXORDIUM regulates brassinosteroid-responsive genes. FEBS Lett 563(1–3):82–86
Dinesh-Kumar S, Anandalakshmi R, Marathe R, Schiff M, Liu Y (2003) Virus-induced gene silencing. In: Plant Functional Genomics. Springer, pp 287–293
Dockter C, Gruszka D, Braumann I, Druka A, Druka I, Franckowiak J, Gough SP, Janeczko A, Kurowska M, Lundqvist J (2014) Induced variations in brassinosteroid genes define barley height and sturdiness, and expand the green revolution genetic toolkit. Plant Physiol 166(4):1912–1927
Dolan L, Poethig RS (1991) Genetic analysis of leaf development in cotton.
Farrar K, Evans IM, Top** JF, Souter MA, Nielsen JE, Lindsey K (2003) EXORDIUM–a gene expressed in proliferating cells and with a role in meristem function, identified by promoter trap** in Arabidopsis. Plant J 33(1):61–73
Goh H-H, Sloan J, Dorca-Fornell C, Fleming A (2012) Inducible repression of multiple expansin genes leads to growth suppression during leaf development. Plant Physiol 159(4):1759–1770
Gonzalez N, Vanhaeren H, Inzé D (2012) Leaf size control: complex coordination of cell division and expansion. Trends Plant Sci 17(6):332–340
Guo H, Li L, Aluru M, Aluru S, Yin Y (2013) Mechanisms and networks for brassinosteroid regulated gene expression. Curr Opin Plant Biol 16(5):545–553
He J-X, Gendron JM, Sun Y, Gampala SS, Gendron N, Sun CQ, Wang Z-Y (2005) BZR1 is a transcriptional repressor with dual roles in brassinosteroid homeostasis and growth responses. Science 307(5715):1634–1638
He P, Zhang Y, Li H, Fu X, Shang H, Zou C, Friml J, **ao G (2021) GhARF16-1 modulates leaf development by transcriptionally regulating the GhKNOX2-1 gene in cotton. Plant Biotechnol J 19(3):548
Jensen JK, Kim H, Cocuron JC, Orler R, Ralph J, Wilkerson CG (2011) The DUF579 domain containing proteins IRX15 and IRX15-L affect xylan synthesis in Arabidopsis. Plant J 66(3):387–400
Jiang J, Zhang C, Wang X (2015) A recently evolved isoform of the transcription factor BES1 promotes brassinosteroid signaling and development in Arabidopsis thaliana. Plant Cell 27(2):361–374. https://doi.org/10.1105/tpc.114.133678
Kim T-W, Guan S, Sun Y, Deng Z, Tang W, Shang J-X, Sun Y, Burlingame AL, Wang Z-Y (2009) Brassinosteroid signal transduction from cell-surface receptor kinases to nuclear transcription factors. Nat Cell Biol 11(10):1254–1260
Kour J, Kohli SK, Khanna K, Bakshi P, Sharma P, Singh AD, Ibrahim M, Devi K, Sharma N, Ohri P (2021) Brassinosteroid signaling, crosstalk and physiological functions in plants under heavy metal stress. Front Plant Sci 12:29
Kukurba KR, Montgomery SBJCSHP (2015) RNA sequencing and analysis. Cold Spring Harb Protoc 11:084970
Kumar S, Tamura K, Nei MJB (1994) MEGA: molecular evolutionary genetics analysis software for microcomputers. Bioinformatics 10(2):189–191
Lee YK, Kim G-T, Kim I-J, Park J, Kwak S-S, Choi G, Chung W-I (2006) LONGIFOLIA1 and LONGIFOLIA2, two homologous genes, regulate longitudinal cell elongation in Arabidopsis. Development 133(21):4305–4314
Li J (2010) Regulation of the nuclear activities of brassinosteroid signaling. Curr Opin Plant Biol 13(5):540–547
Li J, Chory JJC (1997) A putative leucine-rich repeat receptor kinase involved in brassinosteroid signal transduction. Cell 90(5):929–938
Li J, Nagpal P, Vitart V, McMorris TC, Chory JJS (1996) A role for brassinosteroids in light-dependent development of arabidopsis. Science 272(5260):398–401
Li L, Yu X, Thompson A, Guo M, Yoshida S, Asami T, Chory J, Yin Y (2009) Arabidopsis MYB30 is a direct target of BES1 and cooperates with BES1 to regulate brassinosteroid-induced gene expression. Plant J 58(2):275–286
Li Q-F, Lu J, Yu J-W, Zhang C-Q, He J-X (1861) Liu Q-Q (2018) The brassinosteroid-regulated transcription factors BZR1/BES1 function as a coordinator in multisignal-regulated plant growth. Biochim Biophys Acta (BBA) 6:561–571
Liu D, Shi S, Hao Z, **ong W, Luo M (2019) OsbZIP81, A Homologue of Arabidopsis VIP1, may positively regulate JA levels by directly targetting the genes in JA signaling and metabolism pathway in rice. Int J Mol Sci 20(9):2360
Liu L, **e Z, Lu L, Qanmber G, Chen G, Li S, Guo M, Sun Z, Liu Z, Yang Z (2021a) Identification of BR biosynthesis genes in cotton reveals that GhCPD-3 restores BR biosynthesis and mediates plant growth and development. Planta 254(4):1–17
Liu W, Lv Y, Li X, Feng Z, Wang L (2021b) Comparative transcriptome analysis uncovers cell wall reorganization and repressed cell division during cotton fiber initiation. BMC Dev Biol 21(1):1–13
Liu Z, Qanmber G, Lu L, Qin W, Liu J, Li J, Ma S, Yang Z, Yang Z (2018) Genome-wide analysis of BES1 genes in Gossypium revealed their evolutionary conserved roles in brassinosteroid signaling. Sci China Life Sci 61(12):1566–1582
Liu ZH, Chen Y, Wang NN, Chen YH, Wei N, Lu R, Li Y, Li XB (2020) A basic helix–loop–helix protein (GhFP1) promotes fibre elongation of cotton (Gossypium hirsutum) by modulating brassinosteroid biosynthesis and signalling. New Phytol 225(6):2439–2452
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2ΔΔCT. Method 25(4):402–408
Malik W, Shah MSA, Abid MA, Qanmber G, Noor E, Qayyum A, Liang C, Guo S, Zhang R (2018) Genetic basis of variation for fiber quality and quality related biochemical traits in Bt and non-Bt colored cotton. Intl J Agric Biol 20:2117–2124
Mao J, Li JJIJOMS (2020) Regulation of Three Key Kinases of Brassinosteroid Signaling Pathway. Int J Mol Sci 21(12):4340
Nikolov LA, Runions A, Gupta MD, Tsiantis M (2019) Leaf development and evolution. Curr Top Dev Biol 131:109–139
Noguchi T, Fujioka S, Choe S, Takatsuto S, Yoshida S, Yuan H, Feldmann KA, Fejpp T (1999) Brassinosteroid-insensitive dwarf mutants of Arabidopsis accumulate brassinosteroids. Plant Physiol 121(3):743–752
Nolan T, Chen J, Yin YJBJ (2017) Cross-talk of brassinosteroid signaling in controlling growth and stress responses. Biochem J 474(16):2641–2661
Pertea M, Pertea GM, Antonescu CM, Chang T-C, Mendell JT, Salzberg SL (2015) StringTie enables improved reconstruction of a transcriptome from RNA-seq reads. Nat Biotechnol 33(3):290–295
Phillips R, Sarang M, Gibson N (1993) Semiquantitative measurement of gene-expression by rt-PCR-a cautionary tale. Int J Oncol 3(6):1097–1102
Praveena J, Dash S, Behera L, Rout GRJCJOAS Technology (2020) Brassinosteroids: a multifunctional phytohormone of plant development and stress responses. Curr J Appl Sci Technol 39:174–196
Qian W, Wu C, Fu Y, Hu G, He Z, Liu W (2017) Novel rice mutants overexpressing the brassinosteroid catabolic gene CYP734A4. Plant Mol Biol 93(1–2):197–208
Qiao S, Sun S, Wang L, Wu Z, Li C, Li X, Wang T, Leng L, Tian W, Lu T (2017) The RLA1/SMOS1 transcription factor functions with OsBZR1 to regulate brassinosteroid signaling and rice architecture. Plant Cell 29(2):292–309
Robinson MD, McCarthy DJ, Smyth GKJB (2010) edgeR: a Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 26(1):139–140
Saitou N, Nei MJMB (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4(4):406–425
Schröder F, Lisso J, Lange P, Müssig C (2009) The extracellular EXO protein mediates cell expansion in Arabidopsis leaves. BMC Plant Biol 9(1):1–12
Schröder F, Lisso J, Müssig CJPS (2012) Expression pattern and putative function of EXL1 and homologous genes in Arabidopsis. Plant Signal Behav 7(1):22–27
Shang B, Xu C, Zhang X, Cao H, **n W, Hu Y (2016) Very-long-chain fatty acids restrict regeneration capacity by confining pericycle competence for callus formation in Arabidopsis. Proc Natl Acad Sci 113(18):5101–5106
Sousa AO, Camillo LR, Assis ETC, Lima NS, Silva GO, Kirch RP, Silva DC, Ferraz A, Pasquali G, Costa MG (2020) EgPHI-1, a PHOSPHATE-INDUCED-1 gene from Eucalyptus globulus, is involved in shoot growth, xylem fiber length and secondary cell wall properties. Planta 252(3):1–17
Sun Y, Fan XY, Cao DM, Tang W, He K, Zhu JY, He JX, Bai MY, Zhu S, Oh E (2010) Integration of brassinosteroid signal transduction with the transcription network for plant growth regulation in arabidopsis. Dev Cell 19(5):765–777
Szekeres M, Németh K, Koncz-Kálmán Z, Mathur J, Kauschmann A, Altmann T, Rédei GP, Nagy F, Schell J, Koncz C (1996) Brassinosteroids rescue the deficiency of CYP90, a cytochrome P450, controlling cell elongation and de-etiolation in Arabidopsis. Cell 85(2):171–182
Thompson JD, Gibson TJ, Higgins DGJC (2003) Multiple sequence alignment using ClustalW and ClustalX. Curr Protocols Bioinform 1:23–2322
Tian F, Yang D-C, Meng Y-Q, ** J, Gao G (2020) PlantRegMap: charting functional regulatory maps in plants. Nucleic Acids Res 48(D1):D1104–D1113
Todd J, Post-Beittenmiller D, Jaworski JG (1999) KCS1 encodes a fatty acid elongase 3-ketoacyl-CoA synthase affecting wax biosynthesis in Arabidopsis thaliana. Plant J 17(2):119–130
Vert G, Nemhauser JL, Geldner N, Hong F, Chory J (2005) Molecular Mechanisms of Steroid Hormone Signaling in Plants. Ann Rev Cell Dev Biol 21:177–201
Wan S, Wu J, Zhang Z, Sun X, Lv Y, Gao C, Ning Y, Ma J, Guo Y, Zhang Q (2009) Activation tagging, an efficient tool for functional analysis of the rice genome. Plant Mol Biol 69(1–2):69–80
Wang L, Yang Z, Zhang B, Yu D, Liu J, Gong Q, Qanmber G, Li Y, Lu L, Lin Y (2018) Genome-wide characterization and phylogenetic analysis of GSK gene family in three species of cotton: evidence for a role of some GSKs in fiber development and responses to stress. BMC Plant Biol 18(1):1–21
Wang X, Zhang J, Yuan M, Ehrhardt DW, Wang Z, Mao TJTPC (2012) Arabidopsis microtubule destabilizing protein40 is involved in brassinosteroid regulation of hypocotyl elongation. Plant Cell 24(10):4012–4025
Wanjie SW, Welti R, Moreau RA, Chapman KD (2005) Identification and quantification of glycerolipids in cotton fibers: reconciliation with metabolic pathway predictions from DNA databases. Lipids 40(8):773–785
Waterhouse AM, Procter JB, Martin DM, Clamp M, Barton GJJB (2009) Jalview Version 2—a multiple sequence alignment editor and analysis workbench. Bioinformatics 25(9):1189–1191
Wu P, Song X, Wang Z, Duan W, Hu R, Wang W, Li Y, Hou X (2016) Genome-wide analysis of the BES1 transcription factor family in Chinese cabbage (Brassica rapa ssp pekinensis). Plant Growth Regul 80(3):291–301
**e L, Yang C, Wang XJJ (2011) Brassinosteroids can regulate cellulose biosynthesis by controlling the expression of CESA genes in Arabidopsis. J Exp Bot 62(13):4495–4506
Yang C-J, Zhang C, Lu Y-N, ** J-Q, Wang X-LJM (2011) The mechanisms of brassinosteroids’ action: from signal transduction to plant development. Mol Plant 4(4):588–600
Yang ZR, Zhang CJ, Yang XJ, Liu K, Wu ZX, Zhang XY, Zheng W, Xun QQ, Liu CL, Lu LL, Yang ZE, Qian YY, Xu ZZ, Li CF, Li J, Li FG (2014) PAG1, a cotton brassinosteroid catabolism gene, modulates fiber elongation. New Phytol 203(2):437–448. https://doi.org/10.1111/nph.12824
Ye H, Li L, Guo H, Yin Y (2012) MYBL2 is a substrate of GSK3-like kinase BIN2 and acts as a corepressor of BES1 in brassinosteroid signaling pathway in Arabidopsis. Proc Natl Acad Sci 109(49):20142–20147
Yin Y, Wang Z-Y, Mora-Garcia S, Li J, Yoshida S, Asami T, Chory JJC (2002) BES1 accumulates in the nucleus in response to brassinosteroids to regulate gene expression and promote stem elongation. Cell 109(2):181–191
Yu D, Qanmber G, Lu L, Wang L, Li J, Yang Z, Liu Z, Li Y, Chen Q, Mendu V (2018a) Genome-wide analysis of cotton GH3 subfamily II reveals functional divergence in fiber development, hormone response and plant architecture. BMC Plant Biol 18(1):1–18
Yu G, Wang L-G, Han Y, He Q-Y (2012) clusterProfiler: an R package for comparing biological themes among gene clusters. OMICS 16(5):284–287
Yu H, Feng W, Sun F, Zhang Y, Qu J, Liu B, Lu F, Yang L, Fu F, Li W (2018b) Cloning and characterization of BES1/BZR1 transcription factor genes in maize. Plant Growth Regul 86(2):235–249
Zhang L-Y, Bai M-Y, Wu J, Zhu J-Y, Wang H, Zhang Z, Wang W, Sun Y, Zhao J, Sun X (2009) Antagonistic HLH/bHLH transcription factors mediate brassinosteroid regulation of cell elongation and plant development in rice and Arabidopsis. Plant Cell 21(12):3767–3780
Acknowledgements
We thank **n Li and Peng Huo (Zhengzhou Research Center, Institute of Cotton Research of CAAS, Zhengzhou) for their technical assistance.
Funding
This work was supported by grants from the National Natural Science Foundation of China (31971987), Creative Research Groups of China (31621005), Natural Science Foundation of Henan (212300410093) and State Key Laboratory of Cotton Biology Open Fund (CB2020A02).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. SL: Experimentation, writing. KX: Methodology, formal analysis, Writing & editing. GQ: Writing & editing. GC: Conceptualization, writing – review & editing. LL: Software, data curation. MG: Formal analysis. YH: Review & editing. LL: Methodology, data curation. ZL: Supervision. ZY: Supervision.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
11103_2022_1313_MOESM1_ESM.tif
Graphical representation of average cell area, number of cells per leaf area, and total number of cortical cells. (A) The average area of cells in the selected area in the leaf of pag1 and WT (CRI24) plants. (B) The average area of cells in the selected area in the leaf of overexpression GhBES1.4, GhBES1-RNAi, and WT (CRI24) plants. (C) The average area of cells in the selected area in the leaf of VIGS-GhEXO2 and vector control plants. (D) The number of cells per leaf area in the selected area in the leaf of pag1 and WT (CRI24) plants (20,000 μm 2). (E) The number of cells per leaf area in the selected area in the leaf of overexpression GhBES1.4, GhBES1-RNAi, and WT (CRI24) plants (20,000 μm 2). (F) The number of cells per leaf area in the selected area in the leaf of VIGS-GhEXO2 and vector control plants (20,000 μm 2). (G) The total number of cortical cells in the leaf of pag1 and WT (CRI24) plants. (H) The total number of cortical cells in the leaf of overexpression GhBES1.4, GhBES1-RNAi, and WT (CRI24) plants. (I) The total number of cortical cells in the leaf of VIGS-GhEXO2 and vector control plants. Student’s t-test: ** P<0.05,***P<0.001. Supplementary file1 (TIF 17631 kb)
11103_2022_1313_MOESM2_ESM.tif
(A) Heatmap of expression of DEGs related to hormone-related pathway in pag1 cotyledon transcriptome data. (B) Heatmap of DEG expression associated with cell expansion-related pathways in GhBES1.4 overexpression cotyledon transcriptome data. Supplementary file2 (TIF 3320 kb)
11103_2022_1313_MOESM3_ESM.tif
(A) Multiple sequence alignment of GhEXO2 and Arabidopsis EXO proteins showed the presence of the conserved Phi_1 domain and a signal peptide region in all observed EXO proteins. (B) Phylogenetic analysis of GhEXO genes and Arabidopsis EXO genes. Supplementary file3 (TIF 20380 kb)
11103_2022_1313_MOESM4_ESM.tif
Identification of GhBES1 transgenic plants. (A) qRT-PCR validation of GhBES1.2, GhBES1.4, and GhBES1.5 transcript levels in RNAi plants. (B) PCR validation of GhBES1-pBI121 vector in RNAi plants. Primer sequences are provided in Table S2. (C) qRT-PCR validation of GhBES1.4 transcript levels in overexpressed plants. (D) PCR validation of GhBES1-pCAMBIA2300 vector in GhBES1.4 overexpression plants. Student’s t-test: *P<0.05, **P<0.01,***P<0.001. Supplementary file4 (TIF 9890 kb)
11103_2022_1313_MOESM6_ESM.tif
(A) Microscopic imaging analysis for cell elongation in the stem of GhEXO2 silenced and control plants. (B) Graphical representation of the number of cells in the selected area in the stem of GhEXO2 silenced and control plants. Each experiment was performed with three biological repeats and the error bars indicate the standard deviation among these replicates. Student’s t-test: ***P<0.001. Supplementary file6 (TIF 7008 kb)
11103_2022_1313_MOESM7_ESM.tif
Relative expression patterns of cell elongation genes. qRT-PCR analysis of selected genes (from RNA-seq data) related to cell elongation (GhKCS, GhEXPA4, GhEXPA8s, GhIRX15-L, and GhLNG1) and BR biosynthesis (GhCPD and GhDWF4) was conducted. Each experiment was performed in three biological repeats and the error bar indicated the standard deviation among these replicates. Supplementary file7 (TIF 10603 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, S., **ng, K., Qanmber, G. et al. GhBES1 mediates brassinosteroid regulation of leaf size by activating expression of GhEXO2 in cotton (Gossypium hirsutum). Plant Mol Biol 111, 89–106 (2023). https://doi.org/10.1007/s11103-022-01313-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11103-022-01313-5