Abstract
Purposes
This article describes an interesting phenomenon in which optimized freeze-dried (FD) biopharmaceutical formulations are generally more prone to degradation than their liquid counterparts during drop** and proposes an underlying cause for this surprising phenomenon.
Methods
Two monoclonal antibodies (mAbs) and a fusion protein (FP) were used as model biopharmaceuticals. The stability after drop** stress was determined by ultraviolet–visible (UV–Vis), size exclusion high-performance liquid chromatography (SE–HPLC), micro-flow imaging (MFI), and dynamic light scattering (DLS).
Results
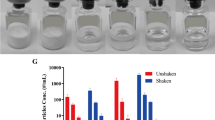
Contrary to what we would normally assume, the FD formulations of the three biopharmaceuticals studied here generally showed much higher amounts of protein sub-visible particles (SbVPs) than liquid formulations after applying the same drop** stress as determined by MFI and DLS. Traditional techniques, such as UV–Vis and SE–HPLC, could hardly detect such degradation.
Conclusions
We propose that the higher temperature caused by drop** for the FD powders than the liquid formulations was probably one of the root causes for the higher amount of particles formed for the FD powders. We also recommend that drop** stress should be included for early-stage screening and choosing liquid versus FD biopharmaceutical formulations.










Similar content being viewed by others
Abbreviations
- ASTM:
-
American Society for Testing and Materials
- CHO:
-
Chinese Hamster Ovary
- C p :
-
Specific heat capacity
- EC280 :
-
Extinction coefficient at 280 nm
- FD:
-
Freeze-dried
- FP:
-
Fusion protein
- IgG:
-
Immunoglobulin G
- IR:
-
Infrared
- mAb:
-
Monoclonal antibody
- MFI:
-
Micro-flow imaging
- NMPA:
-
National Medicinal Products Administration of China
- SbVP:
-
Sub-visible particle
- SE–HPLC:
-
Size exclusion high-performance liquid chromatography
- UV–Vis:
-
Ultraviolet–visible
References
Wang W, Singh S, Zeng DL, King K, Nema S. Antibody structure, instability, and formulation. J Pharm Sci. 2007;96:1–26.
Wang W, Ohtake S. Science and art of protein formulation development. Int J Pharm. 2019;568:118505.
Carpenter JF, Pikal MJ, Chang BS, Randolph TW. Rational design of stable lyophilized protein formulations: some practical advice. Pharm Res. 1997;14:969–75.
Tang X, Pikal MJ. Design of freeze-drying processes for pharmaceuticals: practical advice. Pharm Res. 2004;21:191–200.
Fang W-J, Qi W, Kinzell J, Prestrelski S, Carpenter JF. Effects of excipients on the chemical and physical stability of glucagon during freeze-drying and storage in dried formulations. Pharm Res. 2012;29:3278–91.
Telikepalli S, Kumru OS, Kim JH, Joshi SB, O’Berry KB, Blake-Haskins AW, et al. Characterization of the physical stability of a lyophilized IgG1 mAb after accelerated ship**-like stress. J Pharm Sci. 2015;104:495–507.
Randolph TW, Schiltz E, Sederstrom D, Steinmann D, Mozziconacci O, Schöneich C, et al. Do not drop: mechanical shock in vials causes cavitation, protein aggregation, and particle formation. J Pharm Sci. 2015;104:602–11.
Valotta Rodrigues R, Puryear M, Sederstrom D, Lengsfeld CS. Parameters influencing cavitation within vials subjected to drop shock. Sci Rep. 2019;9:19210.
Jiao N, Barnett GV, Christian TR, Narhi LO, Joh NH, Joubert MK, Cao S. Characterization of subvisible particles in biotherapeutic prefilled syringes: the role of polysorbate and protein on the formation of silicone oil and protein subvisible particles after drop shock. J Pharm Sci. 2020;109:640–5.
Wu H, Chisholm CF, Puryear M, Movafaghi S, Smith SD, Pokhilchuk Y, et al. Container surfaces control initiation of cavitation and resulting particle formation in protein formulations after application of mechanical shock. J Pharm Sci. 2020;109:1270–80.
Movafaghi S, Wu H, FrancinoUrdániz IM, Bull DS, Kelly MD, Randolph TW, Goodwin AP. The effect of container surface passivation on aggregation of intravenous immunoglobulin induced by mechanical shock. Biotechnol J. 2020;15:e2000096.
Duerkop M, Berger E, Dürauer A, Jungbauer A. Influence of cavitation and high shear stress on HSA aggregation behavior. Eng Life Sci. 2018;18:169–78.
Duerkop M, Berger E, Dürauer A, Jungbauer A. Impact of cavitation, high shear stress and air/liquid interfaces on protein aggregation. Biotechnol J. 2018;13:e1800062.
Kim NA, Kim DJ, Jeong SH. Do not flick or drop off-label use plastic syringes in handling therapeutic proteins before administration. Int J Pharm. 2020;587:119704.
Siska C, Harber P, Kerwin BA. Shocking data on parcel shipments of protein solutions. J Pharm Sci. 2020;109:690–5.
Fang W-J, Liu J-W, Zheng H-J, Shen B-B, Wang X, Kong Y, et al. Protein sub-visible particle and free radical formation of a freeze-dried monoclonal antibody formulation during drop**. J Pharm Sci. 2021;110:1625–34.
Fang W-J, Liu J-W, Barnard J, Wang H, Qian Y-C, Xu J. Effects of secondary package on freeze-dried biopharmaceutical formulation stability during drop**. J Pharm Sci. 2021;110:2916–24.
Qian C, Wang G, Wang X, Barnard J, Gao J-Q, Bao W, et al. Formation of protein sub-visible particles during powder grinding of a monoclonal antibody. Eur J Pharm Biopharm. 2020;149:1–11.
Fang W-J, Liu J-W, Gao H, Qian Y-C, Gao J-Q, Wang H. Secondary packages cannot protect liquid biopharmaceutical formulations from drop**-induced degradation. Pharm Res. 2021;38:1397–404.
Sharma DK, King D, Oma P, Merchant C. Micro-flow imaging: flow microscopy applied to sub-visible particulate analysis in protein formulations. AAPS J. 2010;12:455–64.
Didenko YT, McNamara WB, Suslick KS. Temperature of multibubble sonoluminescence in water. J Phys Chem A. 1999;103:10783–8.
Hyman SD, Lazio TJW, Kassim NE, Ray PS, Markwardt CB, Yusef-Zadeh F. Plasma formation and temperature measurement during single-bubble cavitation. Nature. 2005;434:52–5.
ACKNOWLEDGMENTS AND DISCLOSURES
We greatly appreciate Zhejiang Bioray Biopharmaceuticals for providing mAb-A, mAb-B, and FP-A for this study. The authors have no conflicts of interest to declare.
Funding
Funding was kindly provided by the Ministry of Science and Technology of China (Grant No. 2018ZX09J18107-002), the National Natural Science Foundation of China (Grant No. 81741144), and Experimental Technology Project of Zhejiang University (Grant No. SJS202003).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Fang, WJ., Pang, MJ., Liu, JW. et al. Freeze-Dried Biopharmaceutical Formulations are Surprisingly Less Stable than Liquid Formulations during Drop**. Pharm Res 39, 795–803 (2022). https://doi.org/10.1007/s11095-022-03235-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-022-03235-9