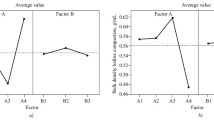
Features of using the generalized Harrington desirability function for the selection of a binder and lubricant for GML-1 (N-benzyl-N-methyl-1-phenylpyrrolo[1,2-a]pyrazine-3-carboxamide) tablets are considered. The factors affecting the characteristics of the finished dosage form are evaluated. Model formulations with different qualitative and quantitative compositions of disintegrant and lubricant excipients were developed. Their pharmaceutical and technological properties were studied. The factors having the greatest impact on indicators such as the mass loss on drying, strength, disintegration, and solubility of the tablets were identified. The type and amount of disintegrant and lubricant in the tablet that could produce a dosage form with the optimal pharmaceutical and technological characteristics were determined.

Similar content being viewed by others
References
S. V. Tishkov, E. V. Blynskaya, K. V. Alekseev, et al., Farm. Farmakol., 10(1), 69 – 81 (2022).
S. V. Tishkov, E. V. Blynskaya, K. V. Alekseev, et al., Biofarm. Zh., 14(1), 28 – 38 (2022).
S. A. Yarkov, G. V. Mokrov, T. A. Gudasheva, et al., Eksp. Klin. Farmakol., 79(1), 7 – 11 (2016).
M. R. Zhang, K. Kumata, J. Maeda, et al., J. Nucl. Med., 48(11), 1853 – 1861 (2007).
M. A. Yarkova, G. V. Mokrov, T. A. Gudasheva, et al., Khim.-farm. Zh., 50(8), 3 – 6 (2016); Pharm. Chem. J., 50(8), 501 – 504 (2016).
M. A. Yarkova, P. Yu. Povarnina, G. V. Mokrov, et al., Eksp. Klin. Farmakol., 80(4), 3 – 7 (2017).
A. A. Novitskii, P. O. Bochkov, R. V. Shevchenko, et al., Eksp. Klin. Farmakol., 81(6), 24 – 28 (2018).
D. V. Yudina, E. V. Blynskaya, K. V. Alekseev, et al., Farmatsiya, 67(3), 35 – 40 (2018).
M. A. Yarkova, E. V. Blynskaya, D. V. Yudina, et al., Khim.-farm. Zh., 53(4), 39 – 43 (2019); Pharm. Chem. J., 52(9), 790 – 795 (2018).
E. V. Blynskaya, K. V. Alekseev, S. V. Tishkov, et al., Mathematical Modeling of Tablet Technology [in Russian], Tipografiya Mittel’ Press, Moscow (2022).
L. M. Gaevaya, L. N. Grushevskaya, M. S. Sergeeva, et al., Eksp. Klin. Farmakol., 81(5s), 50 – 50a (2018).
L. M. Gaevaya, L. N. Grushevskaya, M. S. Sergeeva, et al., Khim.-farm. Zh., 52(9), 43 – 48 (2018); Pharm. Chem. J., 52(9), 790 – 795 (2018).
D. V. Yudina, E. V. Blynskaya, K. V. Alekseev, et al., Farmatsiya, 67(8), 27 – 36 (2018).
J. Wu, Z. Jiang, L. Wan, et al., Arabian J. Sci. Eng., 46(3), 2803 – 2814 (2021).
D. M. D’Addona, S. J. Raykar, D. Singh, and D. Kramar, Procedia CIRP, 99, 707 – 710 (2021).
M. Dadhich, O. S. Prajapati, and V. Sharma, Powder Technol., 378, 104 – 123 (2021).
D. E. Castillo, D. C. Montgomery, and D. R. McCarville, J. Quality Technol., 28(3), 337 – 345 (1996).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiko-Farmatsevticheskii Zhurnal, Vol. 58, No. 1, pp. 50 – 55, January, 2024.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gavrilov, D.I., Blynskaya, E.V., Tishkov, S.V. et al. Formulating GML-1 Tablets Using the Harrington Desirability Function. Pharm Chem J 58, 126–132 (2024). https://doi.org/10.1007/s11094-024-03126-w
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11094-024-03126-w