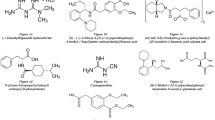
A fast and reliable reversed-phase high-performance liquid chromatography (HPLC) method has been developed for the simultaneous determination of vildagliptin, sitagliptin and saxagliptin in tablets. A stationary phase of Gemini C18 column (250 × 4.6 mm i.d., 5 μm) was used with a mobile phase mixture of methanol: o-phosphoric acid in gradient elution mode. The detection wavelength, flow rate and injection volume were 212 nm, 1 mL/min and 20 μL, respectively. Metformin was also screened in the same method since it is used in combination with vildagliptin and sitagliptin in tablets. The linearity ranges for vildagliptin, sitagliptin and saxagliptin determination were 5.00 – 250.0, 1.00 – 350.0 and 0.50 – 350.0 μg/mL, respectively. The limits of detection (LOD) and quantitation (LOQ) were found to be 0.86 and 2.86 μg/mL for vildagliptin, 0.27 and 0.89 μg/mL for sitagliptin, and 0.25 and 0.84 μg/mL for saxagliptin, respectively. The developed method has been applied to drugs in tablets and proved its reliability for the quality control. The application to the spiked urine samples was also successful and could be further validated for use in biological fluid analysis.




Similar content being viewed by others
References
K. G. Seshadri, M. H. B. Kirubha, Indian J. Pharm. Sci., 71(6), 608 – 614 (2009).
F. Remm, W. M. Franz, C. Brenner., Eur. Heart J. Cardiovasc. Pharmacother., 2(3), 185 – 193 (2016).
A. Cahn, I. Raz, Expert Opin. Emerg. Drugs, 18 (2), 245 – 258 (2013).
K. Hanumanthrao, A. L. Rao, K. B. C. Sekhar, Int. J. Pharm. Chem. Biol. Sci., 4, 361 – 366 (2014).
M. A. Mohammad, E. F. Elkady, M. A. Fouad, Eur. J. Chem., 3(2), 152 – 155 (2012).
A. B. Pharne, B. Santhakumari, A. S. Ghemud, et al., Int. J. Pharm. Chem. Biol. Sci., 4, 119 – 123 (2012).
P. Ramacandra, M. V. Deecaraman, R. S. Chandan, World J. Pharm. Res., 3, 2125 – 2132 (2014).
P. R. Satpathy, V. M. Goud, B. Bhagya, et al., World J. Pharm. Res., 3, 2303 – 2310 (2014).
M. V. Dalawai, P. D. Sanasi, H. K. Sharma, Int. J. Pharm. Sci. and Res., 7(4), 1493 – 1502 (2016).
E. F. Elkady, M. A. Fouad, et al., J. Liq. Chromatogr. Rel., 37, 1895 – 1908 (2014).
C. S. Gowra, Y. P. Reddy, et al., Chromatography, 76(17 – 18), 1153 – 1162 (2013).
P. Ramalingam, V. Bhaskar, Y. P. Reddy, et al., Indian J. Pharm. Sci., 76, 407 – 415 (2014).
B. Sravan, K. Ramakrishna, et al., Chirality, 25(12), 883 – 889 (2013).
R. Rezaee, M. Qomi, F. Piroozi, J. Serbian Chem. Soc., 80(10), 1311 – 1320 (2015).
S. M. Riad, G. Y. Mahmoud, et al., J. AOAC Int., 96(2), 301 – 309 (2013).
Y. Tang, X. Li, N. Wen, et al., China Drug Rev. Annu. Rep., 29(8), 1370 – 1373 (2009).
M. F. Abdel-Ghany, O. Abdel-Aziz, M. F. Ayad, et al., J. Chromatogr. Sci., 53(4), 554 – 564 (2015).
A. K. Bondu, R. Chipirishetti, B. Pillutla, et al., World J. Pharm. Res., 3(4), 1549 – 1557 (2014).
I. Eman, D. A. Hamdy, S. S. Mourad, et al., J. Chromatogr. Sci., 54(1), 79 – 87 (2016).
J. W. Gao, Y. M. Yuan, Y. S. Lu, et al., Biomed. Chromatogr., 26(12), 1482 – 1487 (2012).
A. M. Hanan, K. R. Nesrin, S. D. Sherine, et al., Cairo Univers. Bull. Fac. Pharm., 55(2), 311 – 317 (2017).
N. Narendra, G. Jeyabalan, Asian J. Pharm. Res. Health Care, 4(3), 70 – 74 (2012).
Z. Alkather, M. Hailat, R. Al-Shdefat, et al., Curr. Pharmaceut. Anal., 17(10), 1263 – 1271 (2021).
A. M. Mohamad, C. Andac, S. Caglar Andac., J. Chromatogr Sci., 58(10), 907 – 914 (2020).
ICH Guideline, Q2(R1) Validation of Analytical Procedure: Text and Methodology, London, UK (2005).
Y. L. He, Clin. Pharm., 51(3), 147 – 162 (2012).
E. M. Migoya, C. H. Stevens, A. J. Bergman, et al., Can. J. Clin. Pharm., 16, 165 – 170 (2009).
R. Sekar, K. Singh, A.W. R. Arokiaraj, et al., Int. Rev. Cell Mol. Biol., 326, 279 – 341 (2016).
D. W. Boulton, L. Li, E. U. Frevert, et al., Clin. Pharm., 50(4), 253 – 265 (2011).
ICH Guideline, Reviewer Guidance, Validation of Chromatographic Methods, Center for Drug Evaluation and Research, U. S. Food and Drug Administration, FDA, M. D. Rockville (1994).
Acknowledgements
Authors would like to thank Seda Kahramaner, Nusret Kara and Elif Terlemez for their valuable support. The ethical approval number: B.08.06. YOK. 2. I. U. E. 50. 0. 05. 00/02, Date: 5th of March, 2014, Committee Name: Local Ethics Committee of Istanbul University, Institute of Cardiology.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Andac, S.C., Ozdemir, E. & Andac, C.A. Determination of Three Gliptins in Tablets by Reversed Phase Liquid Chromatography with an Application of Spiked Urine Samples. Pharm Chem J 57, 460–466 (2023). https://doi.org/10.1007/s11094-023-02905-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11094-023-02905-1