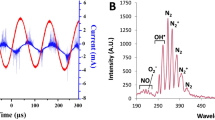
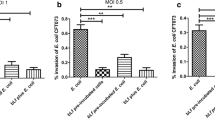
Abstract
Urinary tract infections (UTI) are often caused by resistant uropathogenic bacteria and can lead to sepsis or chronic renal failure. Cold plasma activated liquid (PAL) has known antimicrobial properties with applications in wound disinfection. The aim of our study is to evaluate PAL as a potential treatment of UTI in an animal model. Based on in vitro tests using a uropathogenic E. coli strain, PAL generated in the atmospheric air Glow Discharge plasma had the strongest antimicrobial effect in comparison to other types of PAL and was further tested in vivo. Transurethral PAL application had no effect on bacterial load in the 24 h mouse model of UTI. Upon investigating the treatment failure, we found that urine completely prevented any antimicrobial effects of PAL and PAL treatment of neutrophils resulted in their reduced viability and loss of mitochondrial membrane potential. These results do not support the hypothesis that the in vitro antimicrobial effects of PAL can be translated to the in vivo model of UTI. This could be explained by the attenuating effect of urine on the antimicrobial activity of PAL and its toxicity on immune cells. The detailed mechanism of the effects of urine on PAL requires further investigation.





Similar content being viewed by others
Data Availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Dubuc A et al (2018) Use of cold-atmospheric plasma in oncology: a concise systematic review. Ther Adv Med Oncol 10:1758835918786475
Gilmore BF et al (2018) Cold plasmas for biofilm control: opportunities and challenges. Trends Biotechnol 36(6):627–638
Scholtz V et al (2015) Nonthermal plasma—a tool for decontamination and disinfection. Biotechnol Adv 33(6 Pt 2):1108–1119
Assadian O et al (2019) Effects and safety of atmospheric low-temperature plasma on bacterial reduction in chronic wounds and wound size reduction: a systematic review and meta-analysis. Int Wound J 16(1):103–111
Arndt S et al (2013) Cold atmospheric plasma (CAP) changes gene expression of key molecules of the wound healing machinery and improves wound healing in vitro and in vivo. PLoS One 8(11):79325
Kupke LS et al (2021) Cold atmospheric plasma promotes the immunoreactivity of granulocytes in vitro. Biomolecules 11(6):902
Bekeschus S et al (2016) Neutrophil extracellular trap formation is elicited in response to cold physical plasma. J Leukoc Biol 100(4):791–799
Bekeschus S et al (2018) Physical plasma and leukocytes—immune or reactive? Biol Chem 400(1):63–75
Bekeschus S et al (2018) Cold physical plasma-treated buffered saline solution as effective agent against pancreatic cancer cells. Anticancer Agents Med Chem 18(6):824–831
Bauer G et al (2019) Cold atmospheric plasma and plasma-activated medium trigger RONS-based tumor cell apoptosis. Sci Rep 9(1):14210
Freund E et al (2019) Physical plasma-treated saline promotes an immunogenic phenotype in CT26 colon cancer cells in vitro and in vivo. Sci Rep 9(1):634
Zhang J et al (2019) Discharge plasma-activated saline protects against abdominal sepsis by promoting bacterial clearance. Shock 52(1):92–101
Boehm D et al (2016) Cytotoxic and mutagenic potential of solutions exposed to cold atmospheric plasma. Sci Rep 6:21464
Chauvin J et al (2019) Elucidation of in vitro cellular steps induced by antitumor treatment with plasma-activated medium. Sci Rep 9(1):4866
Griseti E, Merbahi N, Golzio M (2020) Anti-cancer potential of two plasma-activated liquids: implication of long-lived reactive oxygen and nitrogen species. Cancers (Basel) 12(3):721
Sardella E et al (2021) Plasma treated water solutions in cancer treatments: the contrasting role of RNS. Antioxidants Basel 10(4):605
Sersenova D et al (2021) Selective apoptotic effect of plasma activated liquids on human cancer cell lines. Molecules 26(14):4254
Sklias K, Santos Sousa J, Girard PM (2021) Role of short- and long-lived reactive species on the selectivity and anti-cancer action of plasma treatment in vitro. Cancers 13(4):615
Van Boxem W et al (2017) Anti-cancer capacity of plasma-treated PBS: effect of chemical composition on cancer cell cytotoxicity. Sci Rep 7(1):16478
Kaushik NK et al (2018) Biological and medical applications of plasma-activated media, water and solutions. Biol Chem 400(1):39–62
Laurita R et al (2015) Chemical analysis of reactive species and antimicrobial activity of water treated by nanosecond pulsed DBD air plasma. Clin Plasma Med 3(2):53–61
Shen J et al (2016) Bactericidal effects against S. aureus and physicochemical properties of plasma activated water stored at different temperatures. Sci Rep 6:28–505
Traylor MJ et al (2011) Long-term antibacterial efficacy of air plasma-activated water. J Phys D 44(47):472001
Yost AD, Joshi SG (2015) Atmospheric nonthermal plasma-treated PBS inactivates E. coli by oxidative DNA damage. PLoS ONE 10(10):e0139903
Sihra N et al (2018) Nonantibiotic prevention and management of recurrent urinary tract infection. Nat Rev Urol 15(12):750–776
Hung CS, Dodson KW, Hultgren SJ (2009) A murine model of urinary tract infection. Nat Protoc 4(8):1230–1243
Mobley HL et al (1990) Pyelonephritogenic Escherichia coli and killing of cultured human renal proximal tubular epithelial cells: role of hemolysin in some strains. Infect Immun 58(5):1281–1289
Machala Z et al (2004) DC glow discharges in atmospheric pressure air. J Adv Oxid Technol 7(2):133–137
Machala Z et al (2019) Chemical and antibacterial effects of plasma activated water: correlation with gaseous and aqueous reactive oxygen and nitrogen species, plasma sources and air flow conditions. J Phys D 52:3402
Machala Z, Hensel K, Jedlovský I, Leštinská L, Foltin V, Martišovitš V, Morvová M (2007) Emission spectroscopy of atmospheric pressure plasmas for bio-medical and environmental applications. J Mol Spectrosc 243(2):194–201
Machala Z et al (2013) Formation of ROS and RNS in water electro-sprayed through transient spark discharge in air and their bactericidal effects. Plasma Processes Polym 10(7):649–659
Chen TP, Liang J, Su TL (2018) Plasma-activated water: antibacterial activity and artifacts? Environ Sci Pollut Res Int 25(27):26699–26706
Li Y et al (2017) In vitro studies of the antimicrobial effect of non-thermal plasma-activated water as a novel mouthwash. Eur J Oral Sci 125(6):463–470
Ma R et al (2015) Non-thermal plasma-activated water inactivation of food-borne pathogen on fresh produce. J Hazard Mater 300:643–651
Zhou R et al (2018) Cold atmospheric plasma activated water as a prospective disinfectant: the crucial role of peroxynitrite. Green Chem 20(23):5276–5284
Pan J et al (2017) Investigation of cold atmospheric plasma-activated water for the dental unit waterline system contamination and safety evaluation in vitro. Plasma Chem Plasma Process 37(4):1091–1103
Chen T-P, Su T-L, Liang J (2016) Plasma-activated solutions for bacteria and biofilm inactivation. Curr Bioact Compd 13:59–65
Nicol MJ et al (2020) Antibacterial effects of low-temperature plasma generated by atmospheric-pressure plasma jet are mediated by reactive oxygen species. Sci Rep 10(1):3066
Han L et al (2016) Assessing stress responses to atmospheric cold plasma exposure using Escherichia coli knock-out mutants. J Appl Microbiol 121(2):352–363
Yang Y et al (2020) Inactivation of antibiotic resistant Escherichia coli and degradation of its resistance genes by glow discharge plasma in an aqueous solution. Chemosphere 252:126476
Thai KH, Thathireddy A, Hsieh MH (2010) Transurethral induction of mouse urinary tract infection. J Vis Exp 42:20. https://doi.org/10.3791/2070
Olson PD, Hruska KA, Hunstad DA (2016) Androgens enhance male urinary tract infection severity in a new model. J Am Soc Nephrol 27(6):1625–1634
Lin WC, Lai TY, Wu YE (2000) Scavenging of reactive oxygen species by a urinary preparation. Am J Chin Med 28(2):251–258
Crestale L et al (2018) Cold atmospheric pressure plasma treatment modulates human monocytes/macrophages responsiveness. Plasma 1(2):261–276
Sabbatini M, Magnelli V, Reno F (2021) NETosis in wound healing: when enough is enough. Cells 10(3):494
Funding
This study was supported by the grants funded by the Slovak Research and Development Agency (Grant Number APVV-18-0287 and APVV-17-0382) and by the Grant Agency of Ministry of Education, Science, Research and Sport of the Slovak Republic VEGA 1/0234/18.
Author information
Authors and Affiliations
Contributions
Conceptualization: Peter Celec, Zdenko Machala; Methodology: Michal Pastorek, Slavomír Pásztor, Nadja Ivašková, Jakub Petrus; Formal analysis and investigation: Barbora Konečná, Mária Suchoňová, Ľubomíra Tóthová; Writing—original draft preparation: Michal Pastorek, Ľubomíra Tóthová; Writing—review and editing: Roman Gardlík; Funding acquisition: Zdenko Machala, Ľubomíra Tóthová; Resources: Zdenko Machala; Supervision: Peter Celec.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Pastorek, M., Suchoňová, M., Konečná, B. et al. The Effect of Air Plasma Activated Liquid on Uropathogenic Bacteria. Plasma Chem Plasma Process 42, 561–574 (2022). https://doi.org/10.1007/s11090-022-10239-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11090-022-10239-1