Abstract
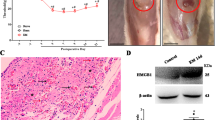
Pelvic organ cross-sensitization, also termed as viscero-visceral referred hyperalgesia, is a major contributor to painful endometriosis. Its underlying mechanism is poorly understood. Clinical and basic studies have shown that gabapentin, a drug that binds to the α2δ−1 subunit of voltage-dependent calcium channels (Cavα2δ−1), is effective in treating chronic visceral pain. Accordingly, we hypothesized that pelvic organ cross-sensitization in painful endometriosis is mediated by an upregulation of Cavα2δ−1 in the spinal cord. We examined if the dysregulation of spinal Cavα2δ−1 subunit may play an important role in the development of ectopic growths-to-colon cross-sensitization in a rat model of experimentally-induced endometriosis. Our findings suggest that there was an increased Cavα2δ−1 expression in the dorsal horn and an ectopic growths-to-colon cross-sensitization in female rats with established endometriosis. Intrathecal administration of gabapentin (300 μg) remarkably reduced the ectopic growths-to-colon cross-sensitization in rats with established endometriosis. Furthermore, intrathecal injection of Cavα2δ−1 antisense oligodeoxynucleotides reversed the ectopic growths-to-colon cross-sensitization and also normalized the upregulation of spinal Cavα2δ−1 expression in endometriosis rats. The current study suggests that the upregulation of Cavα2δ−1 in the spinal cord may contribute to pelvic organ cross-sensitization in painful endometriosis. Our study may provide a biological basis for selectively targeting this pathway to relieve viscero-visceral referred hyperalgesia in patients with painful endometriosis.






Similar content being viewed by others
References
Hansen KE, Kesmodel US, Baldursson EB, Schultz R, Forman A (2013) The influence of endometriosis-related symptoms on work life and work ability: a study of Danish endometriosis patients in employment. Eur J Obstet Gynecol Reprod Biol 169:331–339
Hansen KE, Kesmodel US, Baldursson EB, Kold M, Forman A (2014) Visceral syndrome in endometriosis patients. Eur J Obstet Gynecol Reprod Biol 179:198–203
Malykhina AP (2007) Neural mechanisms of pelvic organ cross-sensitization. Neuroscience 149:660–672
Pezzone MA, Liang R, Fraser MO (2005) A model of neural cross-talk and irritation in the pelvis: implications for the overlap of chronic pelvic pain disorders. Gastroenterology 128:1953–1964
Giamberardino MA, Berkley KJ, Affaitati G, Lerza R, Centurione L, Lapenna D, Vecchiet L (2002) Influence of endometriosis on pain behaviors and muscle hyperalgesia induced by a ureteral calculosis in female rats. Pain 95:247–257
Boroujerdi A, Zeng J, Sharp K, Kim D, Steward O, Luo ZD (2011) Calcium channel alpha-2-delta-1 protein upregulation in dorsal spinal cord mediates spinal cord injury-induced neuropathic pain states. Pain 152:649–655
Odrcich M, Bailey JM, Cahill CM, Gilron I (2006) Chronobiological characteristics of painful diabetic neuropathy and postherpetic neuralgia: diurnal pain variation and effects of analgesic therapy. Pain 120:207–212
Liao XZ, Zhou MT, Mao YF, Xu H, Chen H, Sun JH, **ong YC (2010) Analgesic effects of gabapentin on mechanical hypersensitivity in a rat model of chronic pancreatitis. Brain Res 1337:104–112
Abdel-Salam OM, Sleem AA (2009) Study of the analgesic, anti-inflammatory, and gastric effects of gabapentin. Drug Discov Ther 3:18–26
Stepanovic-Petrovic RM, Tomic MA, Vuckovic SM, Paranos S, Ugresic ND, Prostran MS, Milovanovic S, Boskovic B (2008) The antinociceptive effects of anticonvulsants in a mouse visceral pain model. Anesth Analg 106:1897–1903
Luo ZD, Chaplan SR, Higuera ES, Sorkin LS, Stauderman KA, Williams ME, Yaksh TL (2001) Upregulation of dorsal root ganglion (alpha)2(delta) calcium channel subunit and its correlation with allodynia in spinal nerve-injured rats. J Neurosci 21:1868–1875
Vernon MW, Wilson EA (1985) Studies on the surgical induction of endometriosis in the rat. Fertil Steril 44:684–694
Berkley KJ, Dmitrieva N, Curtis KS, Papka RE (2004) Innervation of ectopic endometrium in a rat model of endometriosis. Proc Natl Acad Sci USA 101:11094–11098
Berkley KJ, Cason A, Jacobs H, Bradshaw H, Wood E (2001) Vaginal hyperalgesia in a rat model of endometriosis. Neurosci Lett 306:185–188
Qu R, Tao J, Wang Y, Zhou Y, Wu G, **ao Y, Hu CY, Jiang X, Xu GY (2013) Neonatal colonic inflammation sensitizes voltage-gated Na(+) channels via upregulation of cystathionine beta-synthetase expression in rat primary sensory neurons. Am J Physiol Gastrointest Liver Physiol 304:G763–G772
Umezawa M, Sakata C, Tanaka N, Kudo S, Tabata M, Takeda K, Ihara T, Sugamata M (2008) Cytokine and chemokine expression in a rat endometriosis is similar to that in human endometriosis. Cytokine 43:105–109
Kalu E, Sumar N, Giannopoulos T, Patel P, Croucher C, Sherriff E, Bansal A (2007) Cytokine profiles in serum and peritoneal fluid from infertile women with and without endometriosis. J Obstet Gynaecol Res 33:490–495
Berkley KJ, McAllister SL, Accius BE, Winnard KP (2007) Endometriosis-induced vaginal hyperalgesia in the rat: effect of estropause, ovariectomy, and estradiol replacement. Pain 132(Suppl 1):S150–S159
Cason AM, Samuelsen CL, Berkley KJ (2003) Estrous changes in vaginal nociception in a rat model of endometriosis. Horm Behav 44:123–131
Luo ZD, Calcutt NA, Higuera ES, Valder CR, Song YH, Svensson CI, Myers RR (2002) Injury type-specific calcium channel alpha 2 delta-1 subunit up-regulation in rat neuropathic pain models correlates with antiallodynic effects of gabapentin. J Pharmacol Exp Ther 303:1199–1205
Li CY, Zhang XL, Matthews EA, Li KW, Kurwa A, Boroujerdi A, Gross J, Gold MS, Dickenson AH, Feng G, Luo ZD (2006) Calcium channel alpha2delta1 subunit mediates spinal hyperexcitability in pain modulation. Pain 125:20–34
Li KW, Yu YP, Zhou C, Kim DS, Lin B, Sharp K, Steward O, Luo ZD (2014) Calcium channel α2δ1 proteins mediate trigeminal neuropathic pain states associated with aberrant excitatory synaptogenesis. J Biol Chem 289:7025–7037
Smiley MM, Lu Y, Vera-Portocarrero LP, Zidan A, Westlund KN (2004) Intrathecal gabapentin enhances the analgesic effects of subtherapeutic dose morphine in a rat experimental pancreatitis model. Anesthesiology 101:759–765
McAllister SL, Dmitrieva N, Berkley KJ (2012) Sprouted innervation into uterine transplants contributes to the development of hyperalgesia in a rat model of endometriosis. PLoS One 7:e31758
Acknowledgments
This work was supported by National Natural Science Foundation of China (30801073, 81171055, 81428008), Bei**g Municipal Natural Science Foundation (7152056), New Century Excellent Talents Program from the Ministry of Education, China (NCET-10-0014), the Excellent Program for Scientific Activity of Returned Oversea Scholar, Bei**g, China (2013), and the Program for High Levels of Health Personnel in Bei**g City, China (2013).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, Y., Zhang, M., **e, F. et al. Upregulation of α2δ−1 Calcium Channel Subunit in the Spinal Cord Contributes to Pelvic Organ Cross-Sensitization in a Rat Model of Experimentally-Induced Endometriosis. Neurochem Res 40, 1267–1273 (2015). https://doi.org/10.1007/s11064-015-1592-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-015-1592-3