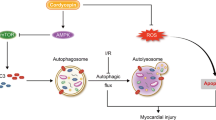
Abstract
Introduction: Autophagy is known as a conserved mechanism in order to preserve cell survival under various stress conditions via maintaining cellular homeostasis. Although autophagy is active in the heart at baseline and plays a critical role in cell survival, inappropriate activation of autophagy following ischemia/reperfusion (I/R) injury leads to cell death. Main text: The distinct functions of autophagy in myocardial I/R injury condition have been examined in numerous studies, however, contradicting results have been achieved in this field. These studies have documented that autophagy acts as a double-edged sword in myocardial I/R injury. Clarifying the exact role of autophagy in determining the health or death of cardiomyocytes under I/R injury is very helpful to achieve better cardioprotection in prospective clinical studies. Thus, autophagy may be an interesting target for the treatment or prevention of myocardial I/R injury. But before considering this matter, it is necessary to address the gaps in our knowledge about the complex role of autophagy in myocardial I/R injury. Conclusion: In this review, by providing updated data about the role of autophagy in the heart during ischemia and reperfusion, we tried to provide more insights in this context and encourage scientists to pay special attention towards manipulating autophagy as an intriguing and powerful approach in cardioprotection.


Similar content being viewed by others
References
Algoet M et al (2022) Myocardial ischemia-reperfusion injury and the influence of inflammation. Trends Cardiovasc Med
Davidson SM et al (2020) Mitochondrial and mitochondrial-independent pathways of myocardial cell death during ischaemia and reperfusion injury. J Cell Mol Med 24(7):3795–3806
Mokhtari B et al (2020) Human amniotic membrane mesenchymal stem cells-conditioned medium attenuates myocardial ischemia-reperfusion injury in rats by targeting oxidative stress. Iran J Basic Med Sci 23(11):1453
Dong Y et al (2010) Autophagy: definition, molecular machinery, and potential role in myocardial ischemia-reperfusion injury. J Cardiovasc Pharmacol Ther 15(3):220–230
Mokhtari B, Badalzadeh R (2021) The potentials of distinct functions of autophagy to be targeted for attenuation of myocardial ischemia/reperfusion injury in preclinical studies: an up-to-date review. J Physiol Biochem 77(3):377–404
Mokhtari B et al (2022) Alpha-lipoic acid preconditioning plus ischemic postconditioning provides additional protection against myocardial reperfusion injury of diabetic rats: modulation of autophagy and mitochondrial function. Mol Biol Rep 49(3):1773–1782
Aghaei M et al (2019) Targeting autophagy in cardiac ischemia/reperfusion injury: a novel therapeutic strategy. J Cell Physiol 234(10):16768–16778
Schiattarella GG, Hill JA (2016) Therapeutic targeting of autophagy in cardiovascular disease. J Mol Cell Cardiol 95:86–93
Przyklenk K et al (2012) Autophagy as a therapeutic target for ischaemia/reperfusion injury? Concepts, controversies, and challenges. Cardiovasc Res 94(2):197–205
Ma S et al (2015) The role of the autophagy in myocardial ischemia/reperfusion injury. Biochim Biophys Acta 1852(2):271–276.
Mokhtari B, Badalzadeh R, Aboutaleb N (2021) Modulation of autophagy as the target of mesenchymal stem cells-derived conditioned medium in rat model of myocardial ischemia/reperfusion injury. Mol Biol Rep 1–12.
Lavandero S et al (2015) Autophagy in cardiovascular biology. J Clin Investig 125(1):55–64
Chen WR et al (2018) Melatonin attenuates myocardial ischemia/reperfusion injury by inhibiting autophagy via an AMPK/mTOR signaling pathway. Cell Physiol Biochem 47(5):2067–2076
Egan D et al (2011) The autophagy initiating kinase ULK1 is regulated via opposing phosphorylation by AMPK and mTOR. Autophagy 7(6):643–644
Horman S et al (2003) Myocardial ischemia and increased heart work modulate the phosphorylation state of eukaryotic elongation factor-2. J Biol Chem 278(43):41970–41976
Matsui Y et al (2007) Distinct roles of autophagy in the heart during ischemia and reperfusion: roles of AMP-activated protein kinase and Beclin 1 in mediating autophagy. Circ Res 100(6):914–922
Matsui Y et al (2008) Molecular mechanisms and physiological significance of autophagy during myocardial ischemia and reperfusion. Autophagy 4(4):409–415
Zhang Y et al (2019) HIF-1α/BNIP3 signaling pathway-induced-autophagy plays protective role during myocardial ischemia-reperfusion injury. Biomed Pharmacother 120:109464
Takagi H, Matsui Y, Sadoshima J (2007) The role of autophagy in mediating cell survival and death during ischemia and reperfusion in the heart. Antioxid Redox Signal 9(9):1373–1382
Gustafsson ASB, Gottlieb RA (2009) Autophagy in ischemic heart disease. Circ Res 104(2):150–158.
Lin XL et al (2018) Molecular mechanisms of autophagy in cardiac ischemia/reperfusion injury. Mol Med Rep 18(1):675–683
Chen-Scarabelli C et al (2014) The role and modulation of autophagy in experimental models of myocardial ischemia-reperfusion injury. J Geriatr Cardiol 11(4):338
Powell SR (2006) The ubiquitin-proteasome system in cardiac physiology and pathology. Am J Physiol Heart Circ Physiol 291(1):H1–H19
Mizushima N (2007) Autophagy: process and function. Genes Dev 21(22):2861–2873
Bence NF, Sampat RM, Kopito RR (2001) Impairment of the ubiquitin-proteasome system by protein aggregation. Science 292(5521):1552–1555
Tannous P et al (2008) Intracellular protein aggregation is a proximal trigger of cardiomyocyte autophagy. Circulation 117(24):3070
Hamacher-Brady A et al (2007) Response to myocardial ischemia/reperfusion injury involves Bnip3 and autophagy. Cell Death Differ 14(1):146–157
Kim I, Rodriguez-Enriquez S, Lemasters JJ (2007) Selective degradation of mitochondria by mitophagy. Arch Biochem Biophys 462(2):245–253
Valentim L et al (2006) Urocortin inhibits Beclin1-mediated autophagic cell death in cardiac myocytes exposed to ischaemia/reperfusion injury. J Mol Cell Cardiol 40(6):846–852
Kihara A et al (2001) Two distinct Vps34 phosphatidylinositol 3–kinase complexes function in autophagy and carboxypeptidase Y sorting insaccharomyces cerevisiae. J Cell Biol 152(3):519–530
Itakura E et al (2008) Beclin 1 forms two distinct phosphatidylinositol 3-kinase complexes with mammalian Atg14 and UVRAG. Mol Biol Cell 19(12):5360–5372
Pattingre S et al (2005) Bcl-2 antiapoptotic proteins inhibit Beclin 1-dependent autophagy. Cell 122(6):927–939
Zhang H et al (2008) Mitochondrial autophagy is an HIF-1-dependent adaptive metabolic response to hypoxia. J Biol Chem 283(16):10892–10903
Li Y et al (2007) Bnip3 mediates the hypoxia-induced inhibition on mammalian target of rapamycin by interacting with Rheb. J Biol Chem 282(49):35803–35813
Haar L et al (2014) Acute consumption of a high-fat diet prior to ischemia-reperfusion results in cardioprotection through NF-κB-dependent regulation of autophagic pathways. Am J Physiol Heart Circ Physiol 307(12):H1705–H1713
Scherz-Shouval R et al (2007) Reactive oxygen species are essential for autophagy and specifically regulate the activity of Atg4. EMBO J 26(7):1749–1760
Scherz-Shouval R, Elazar Z (2007) ROS, mitochondria and the regulation of autophagy. Trends Cell Biol 17(9):422–427
Morales PE et al (2020) Emerging role of mitophagy in cardiovascular physiology and pathology. Mol Aspects Med 71:100822
Barsoum MJ et al (2006) Nitric oxide-induced mitochondrial fission is regulated by dynamin-related GTPases in neurons. EMBO J 25(16):3900–3911
Yorimitsu T et al (2006) Endoplasmic reticulum stress triggers autophagy. J Biol Chem 281(40):30299–30304
Ogata M et al (2006) Autophagy is activated for cell survival after endoplasmic reticulum stress. Mol Cell Biol 26(24):9220–9231
Brady NR et al (2007) The autophagic response to nutrient deprivation in the hl-1 cardiac myocyte is modulated by Bcl-2 and sarco/endoplasmic reticulum calcium stores. FEBS J 274(12):3184–3197
Li J et al (2008) The unfolded protein response regulator GRP78/BiP is required for endoplasmic reticulum integrity and stress-induced autophagy in mammalian cells. Cell Death Differ 15(9):1460–1471
Mayanskaya S et al (2000) Activity of lysosomal apparatus in rat myocardium during experimental coronary and noncoronary myocardial damage. Bull Exp Biol Med 129(6):530–532
Hamacher-Brady A et al (2006) Autophagy as a protective response to Bnip3-mediated apoptotic signaling in the heart. Autophagy 2(4):307–309
Grünenfelder JR et al (2001) Upregulation of Bcl-2 through caspase-3 inhibition ameliorates ischemia/reperfusion injury in rat cardiac allografts. Circulation 104(suppl_1): I-202–I-206.
Yousefi S et al (2006) Calpain-mediated cleavage of Atg5 switches autophagy to apoptosis. Nat Cell Biol 8(10):1124–1132
Pyo J-O et al (2005) Essential roles of Atg5 and FADD in autophagic cell death dissection of autophagic cell death into vacuole formation and cell death. J Biol Chem 280(21):20722–20729
Ma X et al (2012) Autophagy is impaired in cardiac ischemia-reperfusion injury. Autophagy 8(9):1394–1396
Maejima Y, Isobe M, Sadoshima J (2016) Regulation of autophagy by Beclin 1 in the heart. J Mol Cell Cardiol 95:19–25
Li Y et al (2020) CARD9 promotes autophagy in cardiomyocytes in myocardial ischemia/reperfusion injury via interacting with Rubicon directly. Basic Res Cardiol 115(29):29
Meng Z et al (2017) shRNA interference of NLRP3 inflammasome alleviate ischemia reperfusion-induced myocardial damage through autophagy activation. Biochem Biophys Res Commun 494(3–4):728–735
Hao M et al (2017) Myocardial ischemic postconditioning promotes autophagy against ischemia reperfusion injury via the activation of the nNOS/AMPK/mTOR pathway. Int J Mol Sci 18(3):614
Wei C et al (2013) Activation of autophagy in ischemic postconditioning contributes to cardioprotective effects against ischemia/reperfusion injury in rat hearts. J Cardiovasc Pharmacol 61(5):416–422
Huang C et al (2010) Autophagy induced by ischemic preconditioning is essential for cardioprotection. J Cardiovasc Transl Res 3(4):365–373
Qian J et al (2009) Blockade of Hsp20 phosphorylation exacerbates cardiac ischemia/reperfusion injury by suppressed autophagy and increased cell death. Circ Res 105(12):1223–1231
Hsu C-P et al (2009) Nicotinamide phosphoribosyltransferase regulates cell survival through NAD+ synthesis in cardiac myocytes. Circ Res 105(5):481–491
Gurusamy N et al (2009) Cardioprotection by adaptation to ischaemia augments autophagy in association with BAG-1 protein. J Cell Mol Med 13(2):373–387
Dosenko VE et al (2006) Proteasomal proteolysis in anoxia-reoxygenation, preconditioning and postconditioning of isolated cardiomyocytes. Pathophysiology 13(2):119–125
Dosenko V, Zagoriĭ V, Moĭbenko A (2005) Effect of proteasomal proteolysis on NO-synthase activity in isolated platelets. Ukrains' kyi biokhimichnyi zhurnal 77(3):39–43.
Tsang A et al (2004) Postconditioning: a form of “modified reperfusion” protects the myocardium by activating the phosphatidylinositol 3-kinase-Akt pathway. Circ Res 95(3):230–232
Breitschopf K, Zeiher AM, Dimmeler S (2000) Ubiquitin-mediated degradation of the proapoptotic active form of bid a functional consequence on apoptosis induction. J Biol Chem 275(28):21648–21652
Dosenko VE et al (2006) Protective effect of autophagy in anoxia-reoxygenation of isolated cardiomyocyte? Autophagy 2(4):305–306
Tong G et al (2019) Long non-coding RNA FOXD3-AS1 aggravates ischemia/reperfusion injury of cardiomyocytes through promoting autophagy. Am J Transl Res 11(9):5634
Gao C et al (2020) TXNIP/Redd1 signalling and excessive autophagy: a novel mechanism of myocardial ischaemia/reperfusion injury in mice. Cardiovasc Res 116(3):645–657
Chen C et al (2017) Hyperbaric oxygen protects against myocardial reperfusion injury via the inhibition of inflammation and the modulation of autophagy. Oncotarget 8(67):111522
Chien C-Y, Chien C-T, Wang S-S (2014) Progressive thermopreconditioning attenuates rat cardiac ischemia/reperfusion injury by mitochondria-mediated antioxidant and antiapoptotic mechanisms. J Thorac Cardiovasc Surg 148(2):705–713
Cheng B-C et al (2013) Hypothermia may attenuate ischemia/reperfusion-induced cardiomyocyte death by reducing autophagy. Int J Cardiol 168(3):2064–2069
Du J, Li Y, Zhao W (2020) Autophagy and myocardial ischemia. Autophagy 217–222.
Yan L et al (2005) Autophagy in chronically ischemic myocardium. Proc Natl Acad Sci 102(39):13807–13812
Sciarretta S et al (2011) Is autophagy in response to ischemia and reperfusion protective or detrimental for the heart? Pediatr Cardiol 32(3):275–281
Acknowledgements
Not applicable.
Funding
Financial support was received from Drug Applied Research Center at Tabriz University of Medical Sciences, Tabriz-Iran (IR.TBZMED.VCR.REC.1400.468).
Author information
Authors and Affiliations
Contributions
BM had the idea for the article. BM performed the literature search and data collecting, wrote the manuscript, and designed the figures and table. BM and RB edited the language and critically revised the manuscript. All gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mokhtari, B., Badalzadeh, R. Protective and deleterious effects of autophagy in the setting of myocardial ischemia/reperfusion injury: an overview. Mol Biol Rep 49, 11081–11099 (2022). https://doi.org/10.1007/s11033-022-07837-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-022-07837-9