Abstract
Background
The emergence and spread of drug resistance in Vibrio cholerae are mainly attributed to horizontal gene transfer of mobile genetic elements, especially the SXT (sulfamethoxazole and trimethoprim) element, an integrative conjugative element carrying multiple drug resistance genes. SOS (save our souls) bacterial stress response in Vibrio cholerae plays a pivotal role in inducing the SXT element transfer and induction of the CTX prophage, carrying the important virulence factor cholera toxin encoded by the ctxAB gene.
Methods
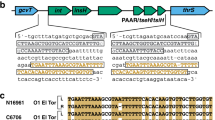
This study investigated whether the subinhibitory concentration of antibiotics like ciprofloxacin, tetracycline, and azithromycin induce SOS response by detecting the expression of recA and lexA, the key genes of SOS response by reverse transcriptase real time PCR (RT-qPCR). We also studied the transfer of SXT element in response to these three antibiotics by bacterial conjugation. Transfer of SXT elements was confirmed by detecting the SXT element-specific conserved genes.
Results
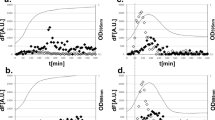
The results of the real-time PCR showed that all three antibiotics induced SOS response with more robust induction by tetracycline and azithromycin relative to ciprofloxacin. We observed a higher frequency of transfer of SXT elements in cultures exposed to these antibiotics and the control mitomycin C compared to unexposed cultures.
Conclusion
Our study indicates that antibiotics including azithromycin, ciprofloxacin, and tetracycline activate SOS response in Vibrio cholerae and demonstrates a robust mechanism for wide dissemination of drug resistance.



Similar content being viewed by others
Data availability
All data generated or analysed during this study are included in this published article and its supplementary information files (ESF1).
Code availability
Not applicable.
Abbreviations
- SXT:
-
Sulfamethoxazole and trimethoprim
- SOS:
-
Save our souls/ship
- MIC:
-
Minimum inhibitory concentration
- HGT:
-
Horizontal gene transfer
References
Lindenbaum J, Greenough WB, Islam MR (1967) Antibiotic therapy of cholera. Bull WHO 36:871–883
Garg P, Chakraborty S, Basu I, Datta S, Rajendran K, Bhattacharya T et al (2000) Expanding multiple antibiotic resistance among clinical strains of Vibrio cholerae isolated from 1992–7 in Calcutta. India Epidemiol Infect 124(3):393–399. https://doi.org/10.1017/s0950268899003957
Bhattacharya D, Sayi DS, Bhattacharjee H et al (2012) Emergence of multidrug-resistant vibrio cholerae O1 biotype El Tor in port blair. India Am J Trop Med Hyg 86(6):1015–1017. https://doi.org/10.4269/ajtmh.2012.11-0327
Mandal J, KP D, Parija SC, (2012) Increasing antimicrobial resistance of vibrio cholerae O1 Biotype El Tor strains isolated in a tertiary-care centre in India. J Health Popul Nutr. https://doi.org/10.3329/jhpn.v30i1.11270
Dalsgaard A, Forslund A, Sandvang D, Arntzen L, Keddy K (2001) Vibrio cholerae O1 outbreak isolates in Mozambique and South Africa in 1998 are multiple-drug resistant, contain the SXT element and the aadA2 gene located on class 1 integrons. J Antimicrob Chemother 48(6):827–838. https://doi.org/10.1093/jac/48.6.827
Das B, Verma J, Kumar P, Ghosh A, Ramamurthy T (2020) Antibiotic resistance in Vibrio cholerae: understanding the ecology of resistance genes and mechanisms. Vaccine 29(38 Suppl 1):A83–A92. https://doi.org/10.1016/j.vaccine.2019.06.031
Hochhut B, Waldor MK (1999) Site-specific integration of the conjugal Vibrio cholerae SXT element into prfC. Mol Microbiol 32(1):99–110. https://doi.org/10.1046/j.1365-2958.1999.01330.x
Burrus V, Pavlovic G, Decaris B, Guédon G (2002) Conjugative transposons: the tip of the iceberg. Mol Microbiol 46(3):601–610. https://doi.org/10.1046/j.1365-2958.2002.03191.x
Waldor MK, Tschäpe H, Mekalanos JJ (1996) A new type of conjugative transposon encodes resistance to sulfamethoxazole, trimethoprim, and streptomycin in Vibrio cholerae O139. J Bacteriol 178(14):4157–4165. https://doi.org/10.1128/jb.178.14.4157-4165.1996
Mohapatra H, Mohapatra SS, Mantri CK, Colwell RR, Singh DV (2008) Vibrio cholerae non-O1, non-O139 strains isolated before 1992 from Varanasi, India are multiple drug-resistant, contain intSXT, dfr18 and aadA5 genes. Environ Microbiol 10(4):866–873. https://doi.org/10.1111/j.1462-2920.2007.01502.x
Iwanaga M, Iwanaga M, Toma C, Miyazato T, Insisiengmay S, Nakasone N, Ehara M (2004) Antibiotic resistance conferred by a class I integron and SXT constin in Vibrio cholerae O1 strains isolated in Laos. Antimicrob Agents Chemother 48(7):2364–2369. https://doi.org/10.1128/AAC.48.7.2364-2369.2004
Wang R, Yu D, Yue J, Kan B (2016) Variations in SXT elements in epidemic Vibrio cholerae O1 El Tor strains in China. Sci Rep 6:22733. https://doi.org/10.1038/srep22733
Beaber JW, Hochhut B, Waldor MK (2004) SOS response promotes horizontal dissemination of antibiotic resistance genes. Nature 427(6969):72–74. https://doi.org/10.1038/nature02241
Radman M (1975) SOS repair hypothesis: phenomenology of an inducible DNA repair which is accompanied by mutagenesis. Basic Life Sci 5A:355–367. https://doi.org/10.1007/978-1-4684-2895-7_48
Walker GC (1984) Mutagenesis and inducible responses to deoxyribonucleic acid damage in Escherichia coli. Microbiol Rev 48(1):60–93. https://doi.org/10.1128/mmbr.48.1.60-93.1984
Janion C (2008) Inducible SOS response system of DNA repair and mutagenesis in Escherichia coli. Int J Biol Sci 4(6):338–344. https://doi.org/10.7150/ijbs.4.338
Baharoglu Z, Mazel D (2011) Vibrio cholerae triggers SOS and mutagenesis in response to a wide range of antibiotics: a route towards multiresistance. Antimicrob Agents Chemother 55(5):2438–2441. https://doi.org/10.1128/AAC.01549-10
Sarkar A, Morita D, Ghosh A, Chowdhury G, Mukhopadhyay AK, Okamoto K et al (2019) Altered integrative and conjugative elements (ICEs) in recent vibrio cholerae O1 isolated from cholera cases, Kolkata. India Front Microbiol 6(10):2072. https://doi.org/10.3389/fmicb.2019.02072
Davies J, Spiegelman GB, Yim G (2006) The world of subinhibitory antibiotic concentrations. Curr Opin Microbiol 9(5):445–453. https://doi.org/10.1016/j.mib.2006.08.006
Jara Song LY, Goff M, Davidian C, Mao Z, London M, Lam K et al (2016) Mutational consequences of ciprofloxacin in Escherichia coli. Antimicrob Agents Chemother 60(10):6165–6172
Mohanraj RS, Samanta P, Mukhopadhyay AK, Mandal J (2020) Haitian-like genetic traits with cree** MIC of azithromycin in vibrio cholerae O1 isolates from Puducherry. India J Med Microbiol 69(3):372–378. https://doi.org/10.1099/jmm.0.001131
Clinical and Laboratory Standards Institute [CLSI] (2015). Methods for Antimicrobial Dilution and Disk Susceptibility Testing of Infrequently Isolated or Fastidious Bacteria: 3rd Edition, CLSI Document M45. Wayne, PA: Clinical and Laboratory Standards Institute.
Laviad-Shitrit S, Sharaby Y, Izhaki I, Peretz A, Halpern M (2018) Antimicrobial susceptibility of environmental non-O1/Non-O139 vibrio cholerae isolates. Front Microbiol 9:1726. https://doi.org/10.3389/fmicb.2018.01726
Yildiz FH, Liu XS, Heydorn A, Schoolnik GK (2004) Molecular analysis of rugosity in a Vibrio cholerae O1 El Tor phase variant. Mol Microbiol 53(2):497–515. https://doi.org/10.1111/j.1365-2958.2004.04154.x
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25(4):402–408. https://doi.org/10.1006/meth.2001.1262
O’Sullivan DM, Hinds J, Butcher PD, Gillespie SH, McHugh TD (2008) Mycobacterium tuberculosis DNA repair in response to subinhibitory concentrations of ciprofloxacin. J Antimicrob Chemother 62(6):1199–1202. https://doi.org/10.1093/jac/dkn387
Walter BM, Cartman ST, Minton NP, Butala M, Rupnik M (2015) The SOS Response master regulator lexi is associated with sporulation, motility and biofilm formation in clostridium difficile. PLoS ONE 10(12):e0144763. https://doi.org/10.1371/journal.pone.0144763
Pande K, Mendiratta DK, Vijayashri D, Thamke DC, Narang P (2011) SXT constant among Vibrio cholerae isolates from a tertiary care hospital. Indian J Med Res 135(3):346–350
Sambe-Ba B, Diallo MH, Seck A et al (2017) Identification of Atypical El Tor V. cholerae O1 Ogawa Hosting SXT Element in Senegal, Africa. Front Microbiol. https://doi.org/10.3389/fmicb.2017.00748
Hochhut B, Marrero J, Waldor MK (2000) Mobilisation of Plasmids and Chromosomal DNA Mediated by the SXT element, a Constin Found in Vibrio cholerae O139. J Bacteriol 182(7):2043–2047. https://doi.org/10.1128/jb.182.7.2043-2047.2000
Poulin-Laprade D, Burrus V (2015) A λ Cro-like repressor is essential for the induction of conjugative transfer of SXT/R391 elements in response to DNA damage. J Bacteriol 197(24):3822–3833. https://doi.org/10.1128/jb.00638-15
Bordeleau E, Brouillette E, Robichaud N, Burrus V (2010) Beyond antibiotic resistance: integrating conjugative elements of the SXT/R391 family that encode novel diguanylate cyclases participate to c-di-GMP signalling in Vibrio cholerae. Environ Microbiol 12(2):510–523. https://doi.org/10.1111/j.1462-2920.2009.02094.x
Taviani E, Ceccarelli D, Lazaro N, Bani S, Cappuccinelli P, Colwell RR et al (2008) Environmental Vibrio spp., isolated in Mozambique, contain a polymorphic group of integrative conjugative elements and class 1 integrons. FEMS Microbiol Ecol 64(1):45–54. https://doi.org/10.1111/j.1574-6941.2008.00455.x
Khan WA, Saha D, Rahman A, Salam MA, Bogaerts J, Bennish ML (2002) Comparison of single-dose azithromycin and 12-dose, 3-day erythromycin for childhood cholera: a randomised, double-blind trial. The Lancet 360(9347):1722–1727. https://doi.org/10.1016/s0140-6736(02)11680-1
Saha D, Karim MM, Khan WA, Ahmed S, Salam MA, Bennish ML (2006) Single-dose azithromycin for the treatment of cholera in adults. N Engl J Med 354(23):2452–2462. https://doi.org/10.1056/nejmoa054493
Rashed SM, Hasan NA, Alam M, Sadique A, Sultana M, Hoq M et al (2017) Vibrio cholerae O1 with reduced susceptibility to ciprofloxacin and azithromycin isolated from a Rural Coastal Area of Bangladesh. Front Microbiol. https://doi.org/10.3389/fmicb.2017.00252
Kohanski MA, Dwyer DJ, Hayete B, Lawrence CA, Collins JJ (2007) A common mechanism of cellular death induced by bactericidal antibiotics. Cell 130(5):797–810. https://doi.org/10.1016/j.cell.2007.06.049
Ysern P, Clerch B, Castańo M, Gibert I, Barbé J, Llagostera M (1990) Induction of SOS genes in Escherichia coli and mutagenesis in Salmonella typhimurium by fluoroquinolones. Mutagenesis 5(1):63–66. https://doi.org/10.1093/mutage/5.1.63
Jara LM, Cortés P, Bou G, Barbé J, Aranda J (2015) Differential roles of antimicrobials in the acquisition of drug resistance through activation of the SOS response in acinetobacter baumannii. Antimicrob Agents Chemother 59(7):4318–4320. https://doi.org/10.1128/aac.04918-14
Maiques E, Ubeda C, Campoy S, Salvador N, Lasa I, Novick RP, Barbé J, Penadés JR (2006) beta-lactam antibiotics induce the SOS response and horizontal transfer of virulence factors in Staphylococcus aureus. J Bacteriol 188(7):2726–2729. https://doi.org/10.1128/JB.188.7.2726-2729.2006
Dörr T, Lewis K, Vulić M (2009) SOS response induces persistence to fluoroquinolones in Escherichia coli. PLoS Genet 5(12):e1000760. https://doi.org/10.1371/journal.pgen.1000760
Bielaszewska M, Idelevich EA, Zhang W, Bauwens A, Schaumburg F, Mellmann A et al (2012) Effects of antibiotics on shiga toxin 2 production and bacteriophage induction by epidemic Escherichia coli O104:H4 strain. Antimicrob Agents Chemother 56(6):3277–3282
Quinones M, Kimsey HH, Waldor MK (2004) LexA cleavage is required for CTX prophage induction. Mol Cell 17(2):291–300. https://doi.org/10.1016/j.molcel.2004.11.046
Guerin E, Cambray G, Sanchez-Alberola N, Campoy S, Erill I, Re Da et al (2009) The SOS response controls integron recombination. Science 324(5930):1034–1034. https://doi.org/10.1126/science.1172914
Acknowledgements
We thank Dr. Asish K Muhkopadhyay, Scientist F (Sr. Deputy Director), Division of Bacteriology, NICED, Kolkata, for kindly providing us the standard strains; classical (O395), El Tor (N16961), and variant (IDH 03709) and E coli J53 strain. We thank Dr. Sudhakar Baluchamy, Professor of Biotechnology Pondicherry University, Pondicherry, for his valuable comments and suggestion given throughout the study. We are also thankful to Mr. Mailan Natrajan, Ms. Ankita Das and Ms. Kamali Doss research scholars, for their time, help and support throughout the project.
Funding
The Council of Scientific and Industrial Research, (CSIR), New Delhi, supported this research, Grant No. Ref: 27(0336)/18/EMR-II.
Author information
Authors and Affiliations
Contributions
JM conceived and designed the experiments. RM performed the experiments. RM and JM analyzed the data. JM & RM discussed the results and reviewed them, RM wrote the manuscript, and JM edited it.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This study was reviewed and approved by the Institute Ethics Committee at Jawaharlal Institute of Postgraduate Medical Education and Research (JIPMER), Pondicherry (Approval No. JIP/IEC/2017/0130) and by the Institute Bio-Safety Committee (Approval No. JIP/IBSC/2017/01) for performing the genetic transfer.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mohanraj, R.S., Mandal, J. Azithromycin can induce SOS response and horizontal gene transfer of SXT element in Vibrio cholerae. Mol Biol Rep 49, 4737–4748 (2022). https://doi.org/10.1007/s11033-022-07323-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-022-07323-2